In the oxidation reduction reactions identify what is being oxidized and what is being reduced.
Q: Why does biological Fe2+ oxidation under oxic conditions occurmainly at acidic pH?
A: Oxic condition refers to the process or environment in which oxygen is involved.
Q: Terminal electron acceptors in anaerobic respiration include all of the following except A) O…
A: Anaerobic respiration is respiration which uses terminal electron acceptors other than oxygen.…
Q: Draw the Fischer projection of glucose and show its catabolism until it releases carbon dioxide via…
A: In order to track where every carbon of glucose went we use isotropic tracers experiments. Carbon of…
Q: Example: Oxidation of ethanol by NAD+ in the presence catalyzed by alcohol dehydrogenase Calculate…
A: The actual change in free energy (∆G) during a reaction is influenced by temperature, pressure and…
Q: Outline the reactions in the a-oxidation pathway
A: The process of α-oxidation results in the removal of the first carbon atom. The carboxyl group of…
Q: explain in quantitative terms the circumstances under which the following reaction can porceed;…
A:
Q: Cytochrome oxidase and succinate-CoQ oxidoreductase are isolated from mitochondria and are incubated…
A: The metabolic pathway Kreb cycle is connected with the electron transport chain, in which the high…
Q: In cellular respiration, carbon dioxide is formed from the oxidation of which of the following?
A: Cellular respiration is a set of metabolic processes that occur within a cell, in which biochemical…
Q: Using the symbols X-H2 and Y, draw a coupled oxidation-reduction reaction. Designate the molecule…
A: An increase in the rate of a reaction without changing itself and the final product, Catalyst is…
Q: Identify any coupled reactions and what processes are coupled together. a. oxidation/reduction…
A: Biochemical reactions are of different types. Different type of reactions are catalyzed by…
Q: Certain tissues, such as the heart muscle, can use lactate as an energy substrate. How much ATP is…
A: Lactate generated by RBC is used by heart, it is oxidized to carbon di oxide and water, following…
Q: Are there any coupled reactions. If so, which ones are coupled. In the oxidation reduction reactions…
A: Biochemical reactions are of different types. Different type of reactions are catalyzed by…
Q: Explain whether the statement, "If an oxidation occurs in a reaction, it must be accompanied by a…
A: Introduction :- Redox reactions include a change in the oxidation state of the substrate. Loss of…
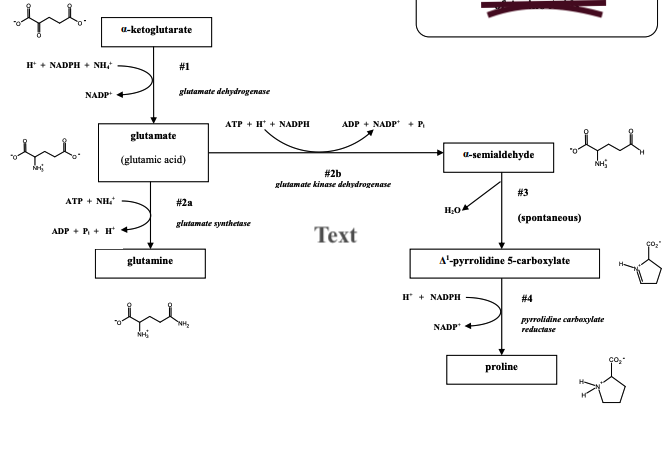
Q: Among the choices, I. alpha-ketoglutarate II. Glutamine III. NADPH IV. H+ which among here is the…
A: Citric acid cycle is also called as Krebs cycle.
Q: H. C=0+NADH + H* H-C-OH + NAD+ CH3 Acetaldehyde CH3 Ethanol 7. Using the reaction above, describe…
A: Fermentation is a type of anaerobic respiration in which Glucose is converted into the ethyl…
Q: Consider the following biochemical reaction. pyruvate + CoA-SH Acetyl-CoA + CO2 AG = -175 kJ Select…
A: The change in free energy of a reaction ΔG is calculated by the equation: ΔG=ΔH −TΔS where ΔG is the…
Q: Why are oxidation–reduction reactions important?
A: A redox reaction or an oxidation-reduction reaction can be described as a type of chemical reaction…
Q: NAD+ coenzyme is required for the reaction of ethanol in the liver to ethanal and NADH. Write down…
A: In liver, ethanol is converted into acetaldehyde, an ethanal and a carcinogen by the enzyme alcohol…
Q: What are the reactions of key oxidation–reduction coenzymes?
A: All biological processes are enhanced by control proteins known as enzymes in which they act as…
Q: Examine the reaction: B-Hydroxybutyrate + NAD* → Acetoacetate+ NADH + H* What is the AEº' of this…
A: The change in internal energy can be calculated by using the formula : ∆E0 = Eo reduction - Eo…
Q: Show the carbon atom that changes oxidation state during the reaction catalyzed by…
A: Glyceraldehyde-3-phosphate dehydrogenase is an enzyme that catalyzes the sixth step of glycolysis in…
Q: Identify the substrates for the nine-reaction Krebs cycle and the overall products.
A: The citric acid cycle or the Krebs cycle is the major cycle that forms ATP. It is a closed cyclic…
Q: Write the overall reaction for the conversion of acetylCoA and oxaloacetate to succinate by citric…
A: The citric acid cycle is the sequential chemical reaction that takes place in aerobic organisms…
Q: The reaction is reversible. Check all that apply. acetyl CoA lactate ethanol
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Malonate is a competitive inhibitor of succinate dehydrogenase. What citric acid cycle…
A: Aerobic metabolism is a set of three basic metabolic processes that occur in cells to generate…
Q: Given the following reaction, identify the class and subclass of the enzyme involved. CH,OPO,2-…
A: Enzymes are biocatalysts that speed up biochemical reactions. These are mainly three dimensional…
Q: Explain how to increase the yield of a TEMPO-mediated oxidation?
A: TEMPO is a chemical compound having the chemical name (2,2,6,6-tetramethyl piperidine-1-yl)oxidanyl…
Q: Vitamin B12 has long been known to be involved in biological radical reactions, cycling between what…
A: Vitamin B12 It is mostly found in animal products such as meat, it is normally not found in plant…
Q: Oxygen is not a reactant in the β oxidation of fatty acids. Can β oxidation occur under anaerobic…
A: Beta-oxidation is the metabolic process which involves multiple steps by which fatty acid molecules…
Q: Select the following enzymes that utlize a mechanism involving covalent catalysis. Select ALL that…
A: Covalent catalysis: The process of covalent catalysis involves the formation of covalent bonds…
Q: Explain How arsenic affects the pyruvate dehydrogenase reaction
A: Pyruvate dehydrogenase reaction is a reaction in which pyruvate is converted into acetyl-CoA.
Q: in which of the reactions in the electron transport chain, coenzyme Q transfers electrons to complex…
A: Most of the energy released during the steps of aerobic respiration is retained by the reduced…
Q: a With which of the possible fates of pyruvate – acetyl CoA, lactate, and ethanol – is each of the…
A: Cellular respiration is the process by which glucose is metabolized and produce energy in the form…
Q: In the energy recovery phase of glycolysis, Glyceraldehyde 3-p reacts with NAD+ In this reaction,…
A: BASIC INFORMATION GLYCOLYSIS it is a pathway through which the glucose is broken down into a…
Q: Complete the following diagram, using arrows to show the flow of electrons, for this reaction…
A: GAP dehydrogenase or Glyceraldehyde-3-phosphate dehydrogenase (GADPH) is an enzyme found in the…
Q: Most biochemists agree that the most accurate number of H+ needed to generate one ASP is blank.…
A: Two ATP were formed as a result of glycolysis, two NAD+ were reduced to two NADH + H+, and glucose…
Q: What is the reduction potential (DE) for the reaction catalyzed by isocitrate dehydrogenase if the…
A: Enzymes that catalyze redox reactions in the living systems are usually categorized under the enzyme…
Q: What does oxidation mean? What does reduction mean?Which of them is endergonic and which is…
A: Loss of electrons is known as "Oxidation" For Example - In oxidation, loss of hydrogen or addition…
Q: List the following substances in order of their increasing oxidizing power: (a) acetoacetate, (b)…
A: Oxidizing power - Oxidizing power is defined as the ability to gain electrons. Oxidation is define…
Q: Complete the following reactions: NADH, H+ oxidoreductase O2 oxidoreductase
A: Introduction This reaction is involved in the linolenic acid metabolism in the presence of…
Q: Oxidative phosphorylation has three reaction products.(a) What is the energy-carrying product?(b)…
A: Oxidative phosphorylation is the metabolic process in which cells use enzymes to oxidize nutrients…
Q: In tricarboxylic acid cycle, what are the reactions that involve the following : (write down…
A: Krebs cycle is the cyclic process, occuring in the mitochondrial matrix of living cells, where…
Q: What are the products of β-oxidation?
A: β-oxidation: It is the long-chain fatty acid degradation process to produce energy. Long fatty…
Q: Explain why rotenone inhibits oxidative phosphorylationwhen the substrate is pyruvate, but not when…
A: Site-specific inhibitors of electron transport have been identified. These compounds prevent the…
In the oxidation reduction reactions identify what is being oxidized and what is being reduced.
Step by step
Solved in 2 steps

- One treatment for shock is to administer dichloroacetate (DCA), which inhibits the kinase associated with the pyruvate dehydrogenase complex. What is the biochemical rationale for this treatment?Although phosphoesters are generally not energetic enough to donate the phosphate to make a high energy bond (e.g., a phosphoanhydride), the phosphate in PEP is used to convert ADP to ATP in the pyruvate kinase reaction. Carefully explain the ‘quirk’ of this reaction that makes it energetically feasible.Fructose-2,6-bisphosphate is an activator of and an inhibitor of O phosphofructokinase; hexokinase O fructose bisphosphatase; phosphofructokinase O phosphofructokinase; fructose bisphosphatase O glucose-6-phosphatase; pyruvate kinase O pyruvate kinase; phosphoenolpyruvate carboxykinase
- The drug troglitazone was used to treat diabetes but was withdrawn from the market when patientswho took the drug suffered from severe side effects. The data below show the activity of an enzymein the steroid biosynthetic pathway in the presence and absence of 10 µM troglitazone.[S] (µM) v0 (pmol min-1)Create a plot of 1/v0 vs. 1/[S]. Calculate KM and vmax in presence and absence of inhibitor. Whattype of inhibitor is troglitazone? Clearly support your answer.The most commonly used biochemical reducing agent is: a) NADH b) NADPH c) FADH2 d) LiALH4 Hexokinase catalyzes: a) conversion of glucose to glucose-6-phosphate b) conversion of glucose-6-phosphate to fructose-6-phosphate c) conversion of fructose-6-phosphate to fructose-1,6-biphosphate d) cleavage of fructose-1,6-biphosphate to glyceraldehyde-3-phosphate and dihydroxyacetone phosphate Which one of these molecules is not found in glycolysis? a) 1,3-biphosphoglycerate b) phosphoenolpyruvate c) pyruvate d) oxaloacetate The major regulatory enzyme of glycolysis is: a) hexokinase b) phosphofructokinase c) aldolase d) triose isomerase NADH is produced at which step in glycolysis? a) conversion of fructose-6-phosphate to fructose-1,6-biphosphate b) cleavage of fructose-1,6-biphosphate to glyceraldehyde-3-phosphate and dihydroxyacetone phosphate c) conversion of glyceraldehyde-3-phosphate to 1,3-biphosphoglycerate d) conversion of phosphoenolpyruvate to…Because dichloroacetate inhibits the enzyme pyruvatedehydrogenase kinase, this compound has been used,with limited results, to treat lactic acidosis. The phosphorylation of the a-subunit of the pyruvate dehydrogenasecomponent of the pyruvate dehydrogenase complex bypyruvate dehydrogenase kinase causes complete loss ofenzymatic activity. Describe the theory behind the clinicaluse of dichloroacetate.
- ATP is both an inhibitor and substrate of adenyl cyclase. As a substrate, the enzyme converts ATP to cyclic AMP only when the energy sources of cells are low. cAMP mediates the synthesis of a kinase that adds a phosphate to glycogen synthase and phosphorylase. The Km value of the active site is 3.5 x 10-5M while the Km for the allosteric site is 1.7 x 10-3M. What is the implication of the difference in Km value of the active and allosteric site in terms of the utilization of the stored sources of energy of cells? Explain.ATP is a (+) allosteric effector, and CTP is a (-) allosteric effector of theenzyme ATCase. Both of these heterotropic effectors bind to the regulatorysubunits on ATCase. The substrates of ATCase, aspartate and carbamoylphosphate, bind the enzyme active site with positive cooperativity (i.e.,they exert a “+” homotropic effect on activity). As the concentrations ofthe substrates change from values where [S] ≪ KM to values where [S] issaturating ([S]≫ KM), how will the binding constants for each of the twoallosteric effectors change? In other words, does ATP bind ATCase withhigher affinity when [S] is low or high? Does CTP bind ATCase with higheraffinity when [S] is low or high?Describe the effects of (1) oligomycin and (2) uncouplers of oxidative phosphorylation, e.g., dinitrophenol (DNP), carbonyl cyanide-p- trifluoromethoxyphenylhydrazone (FCCP), on respiration and ATP synthesis when added to a suspension of mitochondria with excess malate, ADP, and inorganic phosphate (Pi).
- Upon the addition of diazonium salts, ketoacids like glutamate and pyruvate forms in the presence of hydroxide as a color developer in Reitman Frankel Method. At pH 6.8, Pyruvate kinase catalyzes the transfer of phosphate from phosphoenolpyruvate to ADP to become ATP a. both false b. both are true c.. only second is true d. only first is true 2. constriction of the sphincter of oddi may cause falsely ______ in ____ activity a. none of the choices b. decresed, lipase c. increased, ck-mb d. increaased, amylase e. decreased, LDH 3. The amyloclastic method of amylase activity determination refers to the measurement of the disappearance of the starch substrate when mixed with amylase in the sample. The addition of L-tartrate measurement of CP activity increases the sensitivuty of the substrate used to the prostatic ACP isoenzyme a. both are true b. both are false c. only second is true d. only first is trueWhy is it important that the hexokinase reaction have a large negative deltaG? Would the cell be able to generate/store energy if the deltaG were close to zero? The GAPDH reaction uses an active site cysteine to catalyze the reaction. How would replacing the cysteine by serine affect the reaction? Why do kinases require magnesium to function well?The enzyme β-methylaspartase catalyzes the deamination of β-methylaspartate. For this aspartate reaction in the presence of the inhibitor hydroxymethylaspartate (3.8 M), determine KM and whether the inhibition is competitive or noncompetitive (KI = 1.0 M). [S], M V w/o inhibitor, M/s V w/ inhibitor, M/s 1x10-4 0.0259 0.0098 5x10-4 0.0917 0.040 1.5x10-3 0.136 0.086 2.5x10-3 0.150 0.120 5x10-3 0.165 0.142 In the ABSENCE of inhibitor: The Lineweaver-Burke equation is 1V=1V= __________ (1[S])(1[S]) + __________, and the KM is __________ M. In the PRESENCE of inhibitor: The Lineweaver-Burke equation is 1V=1V= ____________ (1[S])(1[S]) + ___________, and the KM is ___________ M. The type of inhibition is ____________. Round-off all answers to two (2) significant figures.

