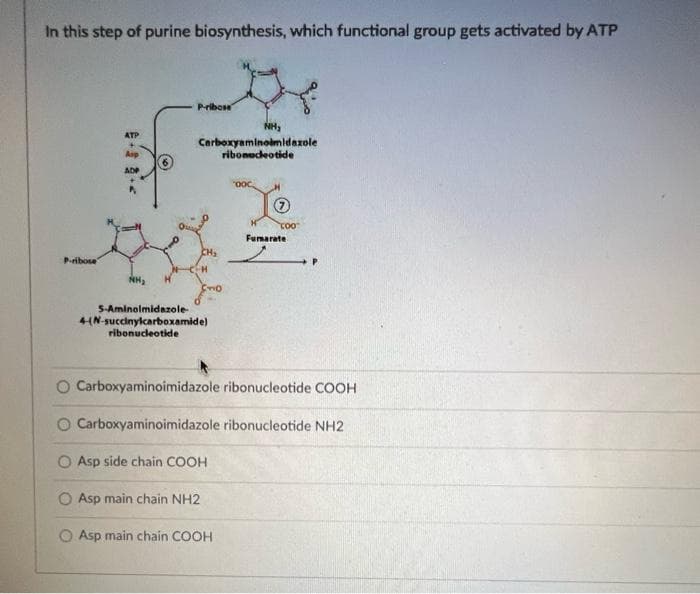
In this step of purine biosynthesis, which functional group gets activated by ATP Pribose NH3 ATP Carboxyaminomidazole ribonedeotide Asp ADP Fumarate Pribose S-Aminolmidazole- 4(N-succinylcarboxamide) ribonucdeotide O Carboxyaminoimidazole ribonucleotide COOH O Carboxyaminoimidazole ribonucleotide NH2 O Asp side chain COOH O Asp main chain NH2 O Asp main chain COOH
Q: abbreviations please answer al|
A: The solubility of proteins is affected by a number of intrinsic as well as extrinsic factors. The…
Q: An acidic amino acid has a side chain that contains O a methyl group O an alcohol group O a carboxyl…
A: The amino acids can be classified as acidic basic, polar and nonpolar based on the side chains of…
Q: An interaction from a protein structure is shown below. This interaction is commonly referred to as…
A: An amino acid-chain molecule has a three-dimensional structure made up of atoms arranged in rows…
Q: If OAT takes ornithine and alpha-ketoglutarate as (a) substrates, draw the structures of the…
A: Aminotransferases are group of enzymes that catalyzes the transamination reaction between amino acid…
Q: Discuss how adsorption, covalent binding, entrapment and encapsulation are used to immobilize…
A: Enzymes are biocatalyst that increases the speed of reaction by lowering the activation…
Q: 1. =0 Н—с —он Н—с —он Н—с —он CH2OH 2. нно н нонн онно I III TIT H-N-C-Ĉ-N-Ć-Ĉ-N-C-Ĉ-N--Ĉ-OH H-C-H…
A: Biological macromolecules can be classified into nucleic acids, proteins, lipids and carbohydrates.
Q: f. Total number of net cytosolic ATP molecules produced right after glycolysis: g. Total number of…
A: The starch molecules obtained through the diet are degraded to form dextrins, maltose, and…
Q: What modifications can be done to streptomycin's structure to enhance its pharmacokinetics and…
A: Streptomycin is the primary found aminoglycoside antibiotic, initially isolated from the micro…
Q: 1. Is the reagent used to cut peptide bonds during amino acid analysis. 2. Is the name of the amino…
A: Proteins are biomolecules composed of amino acids. The amino acids in a protein are joined together…
Q: Briefly explain how ester hydrolysis experiments helped to provide evidence for the mechanism of…
A: Carboxylic acid and free alcohol are formed by an ester hydrolysis reaction that breaks an ester…
Q: how essential is water in our body? Please expand your answer and give examples
A: Water is the solvent of life. It is the most abundant substance in the living systems. It makes up…
Q: iven the following information, calculate the catalytic efficiency of the enzyme. Step by step…
A: The substrate binds to the enzyme's active site and is converted to the product. Multiple…
Q: Alterations in our genome can lead to a variety of genetic diseases including cancers. These can be…
A: Genetic diseases occurs when some harmful variation occurs in the genetic makeup or genes of any…
Q: draw 3 allosteric enzyme curves for glycogen synthesis in the presence of high AMP, high NADH, and…
A: An allosteric enzyme is one that comprises a very small region that helps them in adapting to…
Q: PSI ROS phosphorylated and repr es the 24 hol period in the absence of any light cues. In VIVO in…
A: An alternative pathway for glucose oxidation is the pentose phosphate pathway (PPP). In…
Q: When reacted, how does the use of potassium (K+) vs sodium (Na+) change the soap product?
A: Saponification is a biochemical process which deals with the conversion of oil, lipid, and fats…
Q: all dehydrogenase reactions are variations upon a theme. Within that set of reactions, all reactions…
A: Dehydrogenases is an enzyme belongs to the class of oxidoreductase enzyme. In oxidoreductase the…
Q: 1 What is the dominant biochemical activity or molecular interaction that your protein is predicted…
A: Amyloid-beta precursor protein is single pass transmembrane protein with large extracellular domain…
Q: fatty acid be converted into glucose?
A: ''Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Why is fumarate hydrated? To enable it to interact with an enzyme O To form a double bond To reduce…
A: Introduction: The citric acid cycle is the final common pathway for the oxidation of various…
Q: What is the net ATP production for the complete degradation of a C20 fatty acid molecule to CO2 and…
A: Fatty acids are long hydrocarbon chain carboxylic acids. Fatty acid breakdown occurs in the…
Q: Which of the following fatty acids is/are most likely responsible for the liquid state of oils? COOH…
A: Fats and oils are the composed of triglycerides in which the glycerol is esterified with fatty…
Q: A receptor antagonist is used to O elicit a response by binding to a receptor. O increase a response…
A: Agonists and antagonists give the opposite response on binding to the receptor. a similar…
Q: 7. Which of the following is true of water in the hydration layer of proteins? a. It has a lower AS…
A: Note : Hi! Thank you for the question. We are authorized to answer one question at a time. Since you…
Q: EN Below. Prepare / stabs in big tubes and 8 slants npute the amount of MgCl2.3H2O (grams) needed to…
A: Stab culture is a type of culture in which organisms are introduced into the media by inserting a…
Q: The enzymes responsible for forming the final phosphoiester bond between two DNA fragments during…
A: The phosphodiester bonds constitute the backbone of the strands of DNA.
Q: During glucagon signalling, one goal is to promote export of glucose from liver, In liver cells a…
A: Glucagon is a hormone secreted by the alpha cells in pancreas. It is a principle hormone that…
Q: first half of the Q cycle results in
A: The mechanism of Q-cycle is proposed by Peter Mitchell. Q-cycle terms as proton motive Q cycle. It…
Q: and artificial sweeteners. a) Draw a Haworth projection of the following a-D-glucose, B-D-fructose,…
A: The glucose has a six-membered pyran ring, and the fructose has a five-membered furanose ring. The…
Q: Compare splicing to alternative splicing, focusing on the purpose of alternative splicing, and…
A: Alternative splicing is an alternative splicing process of pre-mRNA during gene expression that…
Q: na ligation and sticky-ehd produced from blunt-end iigation and sticky-end ligation (Hint: observe…
A: Biotechnology is a branch of science in which the life processes has been indulged with technology…
Q: Identify the binding interactions that might be possible for the drug reported below. Rank these…
A: Negative interaction with the Amino acid residue of the target, positive interactions with the Amino…
Q: antibiotic
A: Antibiotics, also known as antibacterials, are medications that destroy or slow down the growth of…
Q: Just answer the items with no answer yet, thank you!
A: Maltose is a disaccharide composed of α-D-Glucose molecules, which are linked together through α…
Q: Which of the following is NOT a fat soluble vitammin? O A O E D
A: Vitamins are the organic compounds required in small amounts for the maintenance of normal growth…
Q: Use drawings to explain how an enzyme (such as hexokinase, mentioned in the text) can distinguish…
A: In order to answer the folowing quastion one should have a clear idea about the emzyme specificity…
Q: The enzyme that catalyzes the conversion of glucose to mannose is classified as a a. Isomerase b.…
A: Enzymes are classified as oxidoreductases, transferases, hydrolases, lyases, isomerases, and ligases…
Q: A polypeptide contains 36 amino acids. How many nucleotides should be found in the open reading…
A: A codon is a sequence of three consecutive nucleotides in a DNA or RNA that codes for a specific…
Q: True or false. If false, changed the underlined words. Ceramides consist of a fatty acyl group…
A: Ceramide is a type of lipid which is commonly found in shampoo. mRNA is the transcribed or the…
Q: Which of the following would be the correct Haworth projection for a-D-gulose, whose Fischer…
A: Glucose is a monosaccharide. It is aldohexose. In Haworth projection, it is represented in a…
Q: Collagen is a fibrous protein which involves interactions between multiple helixes. Ohas a highly…
A: Collagen is a structural protein found naturally in humans and animals. It constitutes upto 30% of…
Q: NH, SH NH, он HN C. H,N HN PEPTIDE RNGCSN NH, PEPTIDE AHIKP
A: Trypsin is a serine protease that is found in the digestive system of vertebrates where it…
Q: why is LDHA an attractive target for a cancer therapeutic?
A: Elevated glycolysis is still a common and fundamental feature of cancer metabolism, which is heavily…
Q: amino acids can be polar neutral, polar folded protein in the aquEous cellular environment, is…
A: The four non-covalent interactions that stabilize the structure of a protein are: hydrophobic…
Q: You have been provided with the results of a sequence analysis of the gene for a fish with a small…
A: Chromatograms are depicted in colourful peaks which are used to represent the sequence of bases in…
Q: 1. You wish to develop a fluorescent sensor to monitor glucose concentrations in human serum.…
A: Fluorescent sensors are very useful and are used to study chlorophyll. It is also used for measuring…
Q: If the carbonyl carbon of acetyl-CoA were marked with 14C, where would that carbon be located within…
A: Metabolism includes biosynthesis/ reduction (an anabolic process) and oxidation (catabolic…
Q: Using skeletal structures, draw the structure of a simple triacylglyceride containing 1…
A: A triglyceride is an ester derived from glycerol and three fatty acids.
Q: Which of the following is an anomer of a-D-galactopyranose? CHOH OH H. он он CH,OH OH он H он H H он…
A: Galactose is aldohexose. It is a C-4 epimer of glucose. The cyclic six-membered ring is called…
Q: How many moles of ATP can be obtained from palmitoleic acid? Show your solution.
A: Palmitoleic acid, also known as hexadec-9-enoic acid, is an omega-7 monounsaturated fatty acid with…
Step by step
Solved in 2 steps

- Give the corresponding class, subclass, sub-subclass, specific enzyme of the following E.C. codes. Give also the systematic name of the enzyme E.C. 2.7.4.3 E.C. 1.1.1.42 E.C. 5.3.1.8 E.C. 5.4.2.2 E.C. 3.4.13.18 Example:E.C. 2.7.1.1Class 2 = transferaseSubclass 7 = transfer of phosphateSub subclass 1 = an alcohol is the phosphate acceptorFourth number 1 = hexokinaseSystematic name = ATP:D-hexose 6-phosphotransferaseWhat product will accumulate if the there is a degradation defect involving the given amino acids? Choices: ?-ketoisovalerate ?-keto-β-methylvalerate ?-ketoisocaproate Phenylpyruvate 4-hydroxyphenylpyruvate dioxygenase Isoleucine Phenylalanine Leucine Valine Tyrosine Amino Acid Degradation Pathway (some choices can be selected more than once) Choices: Phenylalanine Tyrosine Cystine Cysteine Branched-Chain Amino Acids Aromatic Amino Acids Richner-Hanhart Syndrome Tyrosinemia Type III Cystinuria Maple-Syrup Urine Disease Tyrosinemia Type 1 Phenylketonuria Black-Urine Disease Management (some choices may be selected more than once). The number after the condition represents the number of answers required for that item. Choices: Sapropterin Pegvaliase Methionine restriction Vitamin A derivatives Emollients Nitisinone No treatment yet Vitamin B1 Vitamin C Acetazolamide, Hyperdiuresis L-CDME Phenylketonuria (2) Cystinuria (3) Black Urine Disease (2)…The key regulatory enzyme in pyrimidine synthesis is: a. Aspartate transcarbamoylase. b. Glutamine phosphoribosyl amidotransferase. c. Uridine monophosphate synthetase. d. Orotidylate decarboxylase
- With the aid of the simple generic diagram, identify and explain how the type of chemical bonding stabilizes a secondary structure present in 3GRS (glutathione reductase).Unbranched homopolymer of N-acetyl glucosamine is : 1.Cellulose 2.Chitin 3.Curcumin 4.Concanavalin AThe purinosome contains enzymes that convert the serine hydroxymethylgroup to the formyl group of 10-formyltetrahydrofolate. Write a balancedequation for each reaction in this conversion.
- Dihydrouracil and β-ureidopropionate (N-carbamoyl–β-alanine) are intermediates in the conversion of uracil to β-alanine. Provide the structures of the molecules in this pathway.Starting from glutamine ,glycine, aspartate, N-10 formal-ThF ,how many ATP equivalents are required for purine synthesis?Match each of the enzymes involved in de novo pyrimidine synthesis with the correct description: _______ Dihydroorotate dehydrogenase _______ UMP synthase_______ Carbamoyl phosphate synthetase II _______ TMP synthase _______ CTP synthetase_______ Aspartate transcarbamoylase _______ PRPP synthetase (A) attaches an aspartate to an activated carbamoyl molecule(B) bifunctional enzyme that combines a nitrogenase pyrimidine ring to an activated ribose(C) catalyzes the formation of the activated ribose sugar(D) uses glutamine to add an amine to a preformed nucleotide(E) produces the first cyclic nitrogenase ring during de novo pyrimidine synthesis(F) combines bicarbonate and nitrogen from glutamine in the first step of pyrimidine ring synthesis (G) uses a folate derivative to add a methyl group to a preformed nucleotide
- A pentapeptide was found to have the composition Ala Arg Gly Pro Trp. Reaction of the pentapeptide with Sanger’s reagent, followed by hydrolysis, gave the DNP derivative of proline. Treatment of the pentapeptide with carboxypeptidase initially produced alanine. Treatment of the pentapeptide with trypsin gave a tetrapeptide which, when treated with chymotrypsin, produced a tripeptide. What is the sequence of the pentapeptide? (note: trypsin cleaves after basic amino acids) *Lysine degradation requires removal of two amino groups. Removal of itsε-amino group gives a-aminoadipic semialdehyde. This product is thendegraded to acetoacetate by the same chemical strategy used to degrade thebranched-chain amino acids. Draw the proposed intermediates in this pathway (Hint: see Figure 18.14).The highest energy point of the serine protease reaction is the formation of the tetrahedral oxyanion intermediate on the original carbonyl carbon of the scissile peptide bond. True or False? DIPF is an irreversible inhibitor of serine proteases. True or False? The acyl-enzyme intermediate is the transition state of the reaction. True or False? Serine proteases are classified as isomerases. True or False?

