Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter34: Particle Size Determination
Section: Chapter Questions
Problem 34.12QAP
Related questions
Question
Interpret The IR graph identifying peaks for Nitration

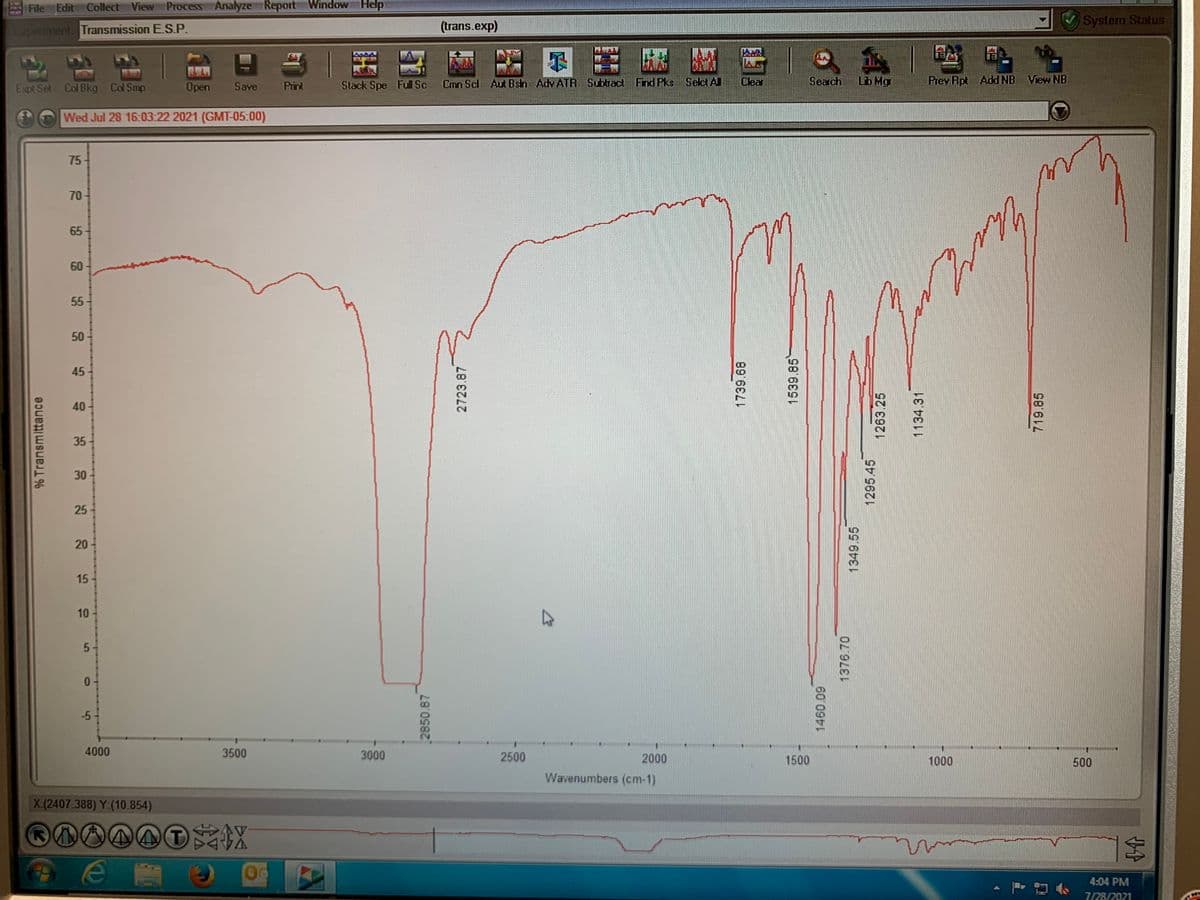
Transcribed Image Text:File Edit Collect View Process Analyze Report Window Help
(trans.exp)
System Status
Lxperiment Transmission ES.P.
Stack Spe Ful Sc
Cmn Scl Aut Bsn Adv ATR Subtract Fod Pks SelctAI
Clear
Search
Lb Mg
Prev Rpt Add NB View NB
Expl Set Col Bkg Col Smp
Open
Save
Print
Wed Jul 28 16:03:22 2021 (GMT-05:00)
75
70
65
60
55
50
45
40
35
25
20
15
10-
-5
4000
3500
3000
2500
2000
1500
1000
500
Wavenumbers (cm-1)
X:(2407.388) Y:(10.854)
V
%24
4:04 PM
7/28/2021
DET
Transmittance
30
2850.87
2723.87
89 6
1539.85
60 09
1376.70
1349.55
1295.45
1263.25
1134.31
719.85
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

