Jse the following table of bond energies to calculate the molar enthalpy of combustion of acetylene (C2H2) gas in oxygen, based on the following chemical equation: C2H2(g) + 2.502(g)→2CO2(g) + H2O(g) Single Bond CNOo H H 432 411 346 386 305 167 459 358 201 142 C=C 602 C=0 799 Multiple C=C 835 C=0 1072 Bonds C=N 615 O=0 494 C=N 887 N=N 942 "All values in kJ/mol** Z O
Jse the following table of bond energies to calculate the molar enthalpy of combustion of acetylene (C2H2) gas in oxygen, based on the following chemical equation: C2H2(g) + 2.502(g)→2CO2(g) + H2O(g) Single Bond CNOo H H 432 411 346 386 305 167 459 358 201 142 C=C 602 C=0 799 Multiple C=C 835 C=0 1072 Bonds C=N 615 O=0 494 C=N 887 N=N 942 "All values in kJ/mol** Z O
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter3: Atomic Shells And Classical Models Of Chemical Bonding
Section: Chapter Questions
Problem 38P: The percent ionic character of the bonds in several interhalogen Molecules (as estimated from their...
Related questions
Question

Transcribed Image Text:Use the following table of bond energies to calculate the molar
enthalpy of combustion of acetylene (C2H2) gas in oxygen, based on
the following chemical equation:
C2H2(g) + 2.502(g) → 2CO2(g) + H2O(g)
->
Single
H.
C
N
kJ/mol
Bond
H
432
C
411
346
386
305
167
1
459
358
201
142
C=C
602
C=O
799
4
6.
C
Multiple
C=C
835
C=0
1072
Bonds
C=N
615
O=0
494
C=N
887
N=N
942
8.
9.
**All values in kJ/mol**
+/-
X 100
2.
7.
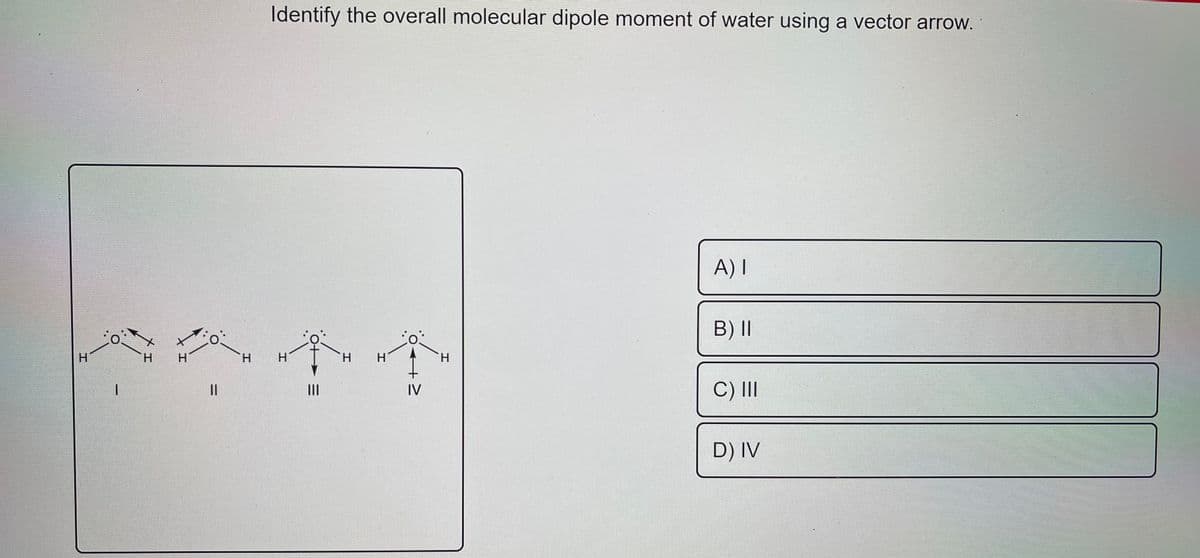
Transcribed Image Text:Identify the overall molecular dipole moment of water using a vector arrow.
A) I
B)II
H.
H.
H
H.
H.
C) II
II
II
IV
D) IV
エ
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning