lactate 0 NAD+ НО 0 НО ОН он н О ОН N NH₂ NH₂ lactate dehydrogenase НО H- НО он н 0 ОН H H+ pyruvate NH₂ N NH₂ NADH Н
Q: Explain why eating celery is considered ‘negative calories’ or negative net energy. Be sure to…
A: Celery is a leafy vegetable that is commonly consumed to burn calories. Most of the diets humans…
Q: When people are talking about ways to lose weight, cutting out carbohydrates is always on the…
A: General myths are: 1. Dieting (Less eating or skipping meals) to reduce weight 2. Skipping just the…
Q: Complete the pathway below by writing the intermediate (I) name, pathway name (P) and enzyme (E)…
A: Pyruvate molecules produced as the end product of the glycolytic pathway enter into the citric acid…
Q: The key to biopolymer self-replication is: O Natural selection O Intramolecular complimentarity O…
A: Biomolecules are organic compounds produced by living organisms. They play important function as…
Q: What are the three photochemical and the food they contained in
A: There is one correction in the question. These are phytochemicals which are present in foods.
Q: Item: Statement: a) Active site b) Induced fit c) Enzymes d) Enzyme-substrate complex 1. Decreases…
A: Metabolic activity is constant in living things. All live cells are constantly undergoing thousands…
Q: (i) (.. From the diagram to the right of the trp repressor in its approximate binding relationship…
A: Tryptophan (trp) repressor: It's a transcription factor that regulates amino acid metabolism. The…
Q: Compare the process of facilitated diffusion to the process of osmosis.
A: The cell membrane is a living, crucial part of the cell. It acts as a physical barrier, enclosing…
Q: Compartmentation occurs through biomolecule self-assembly in order to: isolate from the bulk…
A: A, b and C
Q: write true if the statement if correct and change the bold word/phrase to make it correct addition…
A: Hydrolysis of glycogen is done by the enzyme Glycogen phosphorylase. The activity of this enzyme is…
Q: Information for Part 2 The following table shows the concentration of ATP, ADP and phosphate into…
A: In the above question the change in free energy can be calculated by Δ?′ = Δ?0 + ?? ln ([???][??]/…
Q: Using the regional barometric pressure info. below, and being careful to consider the vapor pressure…
A: The alveolar gas equation is : PAO2=FiO2 (PB-PH2O)- (PaCO2/R), where PAO2 is alveolar oxygen…
Q: pMDawn is digested with EcoR1, and BamHI. Resulting in fragments shown below: EcoRI: 20 kb BamHI:…
A: Agarose gel electrophoresis is a method of gel electrophoresis that is used to separate a mixed…
Q: What is the total number of hydrogen bonds that exist between the DNA strand 5’-TTCAGAG-3’ and its…
A: Adenine and guanine are the purine bases which occur in the nucleic acids. And thymine, cytosine,…
Q: do butter, shortening and vegetable oil have in common? Based on these common features why do you…
A: Fats and oils are esters of glycerol and three fatty acids . Butter is saturated fat, oil are…
Q: The three-dimensional structure of DNA is said to be maintained by the presence of many hydrogen…
A: The biochemical basis of heredity is DNA. It is widely recognized as the genetic data reserve bank.…
Q: 3. Write out the net balanced reaction for the production of cytoplasmic malonyl-CoA starting with…
A: The enzyme Acetyl-CoA carboxylase (ACC) catalyzes the production of 1 molecule of malonyl-CoA from 1…
Q: QUESTION 14 COVID-19 has been shown to effect hemoglobin's ability to bind and transport oxygen…
A: Hemoglobin is an oxygen transport protein present in the RBCs. Hemoglobin transports oxygen from the…
Q: ubiquitin attaches to proteins via many biochemical reactions, please explain how this attachment…
A: Ubiquitin is a highly conserved 76-residue monomeric protein found in eukaryotes. It is found in…
Q: Which of the following regarding disulfide bonds is/are true?
A: Proteins are polymers of amino acids which are linked by peptide/amide bonds and form primary…
Q: polymers have a similar composition to that of common carbohydrates (predominantly composed of…
A: Carbohydrates are oraganic biomolecule consists of Carbon, Hydrogen and Oxygen atom with a chemical…
Q: Choose the secondinary messenger НО HO H3C. afafafa ОН ОН OH ОН ОН ОН NH₂ H₂N ОН ОН
A: A hormone receptor is a receptor molecule that binds to a specific hormone and elicits a cellular…
Q: Q/What are the Irreversible inhibitor ?
A: Inhibitor is an agent that slows or interferes with the chemical reaction. Enzyme inhibitor is a…
Q: Ketohexose sugars can form 8 different stereoisomers. How many of those isomers can be distinguished…
A: Carbohydrates are organic molecules arranged in form of aldehyde or ketones with multiple…
Q: In order to pass the outer mitochondrial membrane a fatty acid Reacts with carnitine Is activated…
A: Introduction: Fatty acid contains a long hydrocarbon chain and a carboxylate group on the end. The…
Q: What hydrogen carriers participate in pentose phosphate pathway? NAD NADP Cobolamin FAD
A: One of the catabolic fate of 6 carbon Glucose 6-phosphate is conversion to ribose 5-phosphate, a 5…
Q: 37.Assume that the phosphoryl group in the shown chemical (Phosphoenolpyruvate (PEP)) is labeled…
A: Gluconeogenesis is the process of glucose synthesis from sources like pyruvate, amino acids,…
Q: What are the advantages and disadvantages of using the Lieberman-Burchard Test for Lipids
A: Lieberman-Burchard Test : this is the specific test used for detection of cholesterol in which…
Q: Name and in a brief describe the process by which immunological diversity is generated.
A: The ability of multicellular creatures to withstand dangerous germs is referred to as immunity.…
Q: Serum concentrations of acetoacetate and ß-hydroxybutyrate increase dramatically after a 3-day fast…
A: Acetone, acetoacetate, and beta-hydroxybutyrate are called ketone bodies. Ketone bodies are…
Q: Assuming that all the carbon skeleton of alanine is completely oxidized and released as CO2. How…
A: Alanine is an amino acid that takes part in protein synthesis. Alanine is a hydrophobic amino acid…
Q: d) Consider the following disaccharide C. i. ii. iii. iv. OH HO OH OH HO Disaccharide C OH OH LOH…
A: A disaccharide is a two monosaccharide linked in a glycosidic bond. What is given to us is the chair…
Q: Understanding membranes: a) Describe the factors that influence fatty acid melting temperatures…
A: The fluidity of membrane depends on the fatty acids present in it. Fatty acids form an integral part…
Q: Which amino acid is predominantly used to add an NH2 moiety during the de novo biosynthesis of…
A: Denovo synthesis of Nucleotides referes to the process of Synthesis of nucleotides from simple…
Q: Vitamins that are necessary for the transfer of hydrogen atoms: CH3 CH3 N NH₂ 'N Но NH₂ SENH ОН МОН…
A: Vitamins are organic compounds that are required in the diet in small amounts. They perform various…
Q: pMDawn is digested with EcoR1, and BamHI. Resulting in fragments shown below: EcoRI: 20 kb BamHI:…
A: Given: Digestion of a plasmid PMDawn with EcoRI, BamHI, and EcoRI+BamHI are as follows: EcoRI: 20 kb…
Q: In RNA OH group is present at 2¹ position
A: RNA has 2 -OH group attached to its carbon backbone, In Ribonucleic acid (RNA) The hydroxyl group…
Q: Chemistry Draw the stable form of the peptide Ser-Trp-Glu-Asp-Cys-Asn at pH 10.40. Be sure to…
A: At pH 7 this is how a oligopetide of Ser-Trp-Glu-Asp-Cys-Asn looks.
Q: Now consider three sugars-glucose, glucuronic acid, and N-acetylglucosamine. Use shorthand (e.g.,…
A: A glycosidic bond is formed between 2 carbon atoms as the result of a dehydration reaction. The…
Q: How does Candida albicans identified in person with Candidiasis? Discuss specific tests being done…
A: Candidiasis is a fungal infection caused by the yeast Candida (a type of fungus). Candida albicans…
Q: Write the sequence of reverse compliment chain to this DNA sequence: CGTCCGCCCCGCGAGCACA…
A: Double stranded nucleic acids are formed through hydrogen bonding between complementary nitrogenous…
Q: what is the amount of cellulose in plant cell? describe the molecular structure of cellulose.
A: Introduction: Cellulose is the most abundant naturally occurring organic material on our Earth. It…
Q: What is the relation between GMO crops and the four of the principles of bioethics? What issues are…
A: A GMO, or genetically modified organism, whose genetic makeup has been modified using scientific…
Q: Which vitamin is needed for FADH formation? Group of answer choices Niacin Riboflavin Vitamin C…
A: A number of significant metabolic reactions require the redox cofactor FAD. It exists in the redox…
Q: The first step of the lysozyme reaction (catalytic mechanism) is shown in the figure below. Which…
A: Only some of the amino acids in the active site of an enzyme play significant roles in catalysis,…
Q: What types of bonds form the primary structure of nucleic acids? 0 hydrophobic peptide hydrogen…
A: The nucleic acids form the genetic material of an organism. The genetic information is stored in the…
Q: Reaction mechanism of proteins, lipids, carbohydrates, nucleic acids
A: Proteins, nucleic acids, lipids and carbohydrates are frequently found in nature as lengthy…
Q: Assume that glycolysis is completely inhibited and thus glucose cannot be converted to pyruvate.…
A:
Q: the maximum amount of ATP that could be generated by the full oxidation of the compound…
A: In the given question, the fatty acid mentioned is CH3(CH2)4COOH. Here full oxidation means…
Q: antibiotics that bind to 30S ribosomal subunit trimetjoprime chloramphenicol sulfonamide…
A: Antibiotics affect the growth of bacteria by inhibiting various metabolic pathways, replication,…
Match each component of the reaction in the figure with the corresponding relative energy level.
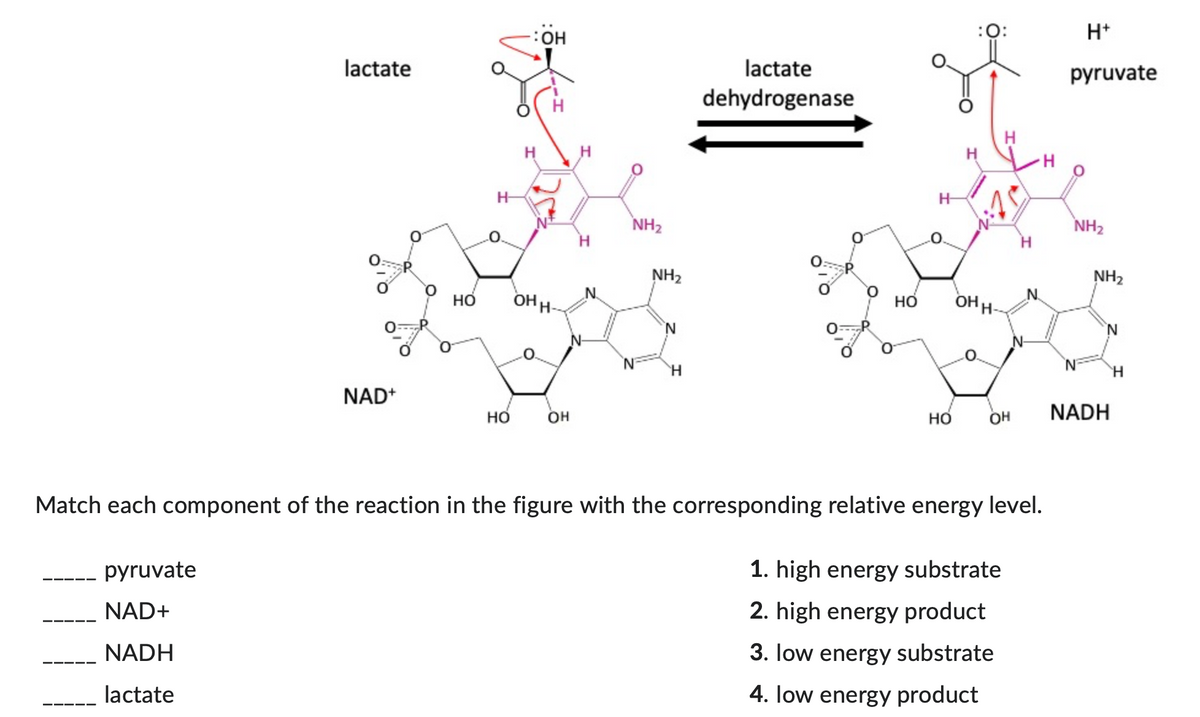
Step by step
Solved in 3 steps with 1 images

- Energetics of the Hexokinase Reaction The standard-state free energy change. Gfor the hexokinase reaction, is — 1 6.7 kJ/mol. Use the values in Table I to calculate the value of Gfor this reaction in the erythrocyte at 37°C.The Effect of lodoacetic Acid on the Glyceraldehyde-3-P Dehydrogenase Reaction (Integrates with Chapters 4 and 14.) How might iodoacetic acid affect the glyceraldehydes-3-phosphate dehydrogenase reaction in glycolysis? Justify your answer.Extending the Mechanism of Methylmalonyl-CoA Mutase to Similar Reactions Based on the mechanism for the methylmalonyl-CoA mutase (see problem 14), write reasonable mechanisms for the following reactions shown.
- Distinguishing the Mechanisms of Class I and Class I Aldolases Fructose bisphosphate aldolase in animal muscle is a class 1 aldolase, which forms a Schiff base intermediate between substrate (for example. fructose-1, 6-bisphosphate or dihydroxyacetone phosphate) and a lysine at the active site (see Figure I8.12). The chemical evidence for this intermediate conies from studies with aldolase and the reducing agent sodium borohydride, NaBH4. Incubation of the enzyme with dihydroxyacetone phosphate and NaBH4 inactivates the enzyme. Interestingly, no inactivation is observed if NabH4 is added to the enzyme in the absence of substrate. Write a mechanism that explains these observations and provides evidence for the formation of a Schiff base intermediate in the aldolase reaction.Effects of Changing Metabolite Concentrations on Glycolysis In an erythrocyte undergoing glycolysis what would be the effect of a sudden increase in the concentration of a. AΤP? b. AMP? c. fructose-1.6-bisphosphate? d. fructose-2, 6-bisphosphate? e. citrate? f. glucose-6-phospthate?Figure 27.3 illustrates the response of R (ATP-regenerating) and U (ATP-utilizing) enzymes to energy charge. a. Would hexokinase be an R enzyme or a U enzyme? Would glutamine: PRPP amidotransferase, the second enzyme in purine biosynthesis, be an R enzyme or a U enzyme? b. If energy charge = 0.5: Is the activity of hexokinase high or low? Is ribose-5-P pyrophosphokinase activity high or low? c. If energy charge = 0.95: Is the activity of hexokinase high or low? Is ribose-5-P pyrophosphokinase activity high or low?
- Radiolabeling with 14C-Glutamate Describe the labeling pattern that would result from the introduction into the TCA cycle of glutamate labeled at Cy with 14C.Ethanol as a Source of Metabolic Energy (Integrates with Chapters 19 and 20.) Acetate produced in ethanol metabolism can be transformed into acetyl-COA by the acetyl thiokinase reaction: Acetate+ATP+CoASHacetyleCoA+AMP+PPiAcetyle-CoA then can enter the citric acid cycle and undergo oxidation to 2 CO2by this route, assuming oxidative phosphorylation is part of the process? (Assume all reactions prior to acetyl-CoA entering the citric acid cycle occur outside the mitochondrion). Per carbon atom, which is a better metabolic fuel, ethanol or glucose? That is, how many ATP equivalents per carbon atom are generated by combustion of glucose versus ethanol to CO2?Understanding the Mechanisms of Reactions Related to Transketolase The mechanistic chemistry of the acetolactate synthase and phosphoketolase reactions (shown here) is similar to that of the transketolase reaction (Figure 22.30). Write suitable mechanisms for these reactions.
- Using the ActiveModel for enoyl-CoA dehydratase, give an example of a case in which conserved residues in slightly different positions can change the catalytic rate of reaction.The standard reduction potential for ubiquione (A or coenzyme Q) is .045 V, and the standard reduciton potential (E) for FAD is -0.219 V. Using these values, show that the oxidation for FADH2 by ubiquinone theoretically liberates enough energy to drive the synthesis of ATP. Faraday constant =96.48KJ/Vol delta G' standard for ATP Synthesis is +30.5 KJ/mol R=8.314 J/mol K=1.987 cal/mol KWrite out the balanced chemical equation for the FIRST round of oxidation of C16:cis-9.

