lonization energy (E;) is the amount of energy required to remove an electron from a neutral gaseous atom or gaseous ion. Electrons are attracted to the positively charged nucleus; therefore removing an electron requires energy. The process is endothermic, and so ionization energies have a positive value. The first ionization energy (Ei1) is the energy associated with the removal of an electron from the neutral gaseous atom. The reaction is represented for the generalized atom X as Part A Based on position in the periodic table and electron configuration, arrange these elements in order of decreasing Rank the elements from highest to lowest ionization energy. To rank items as equivalent, overlap them. • View Available Hint(s) X-X+ +e Reset Help The amount of energy required to remove an electron is related to the effective nuclear charge and the stability of the electron configuration of the atom. It therefore shows periodic variation generally increasing from left to right in a period and from bottom to top of a group. In general, metals have lower E values than nonmetals. Exceptions to this general trend from left to right occur when a completely filled s subshell or half-filled p subshell is encountered. These stable configurations have larger than expected E values. OPSAI Na K Highest ionization energy Lowest ionization energy
lonization energy (E;) is the amount of energy required to remove an electron from a neutral gaseous atom or gaseous ion. Electrons are attracted to the positively charged nucleus; therefore removing an electron requires energy. The process is endothermic, and so ionization energies have a positive value. The first ionization energy (Ei1) is the energy associated with the removal of an electron from the neutral gaseous atom. The reaction is represented for the generalized atom X as Part A Based on position in the periodic table and electron configuration, arrange these elements in order of decreasing Rank the elements from highest to lowest ionization energy. To rank items as equivalent, overlap them. • View Available Hint(s) X-X+ +e Reset Help The amount of energy required to remove an electron is related to the effective nuclear charge and the stability of the electron configuration of the atom. It therefore shows periodic variation generally increasing from left to right in a period and from bottom to top of a group. In general, metals have lower E values than nonmetals. Exceptions to this general trend from left to right occur when a completely filled s subshell or half-filled p subshell is encountered. These stable configurations have larger than expected E values. OPSAI Na K Highest ionization energy Lowest ionization energy
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter6: An Introduction To Spectrometric Methods
Section: Chapter Questions
Problem 6.9QAP: Cesium is used extensively in photocells and in television cameras because it has the lowest...
Related questions
Question
Please answer question 16 part A and B
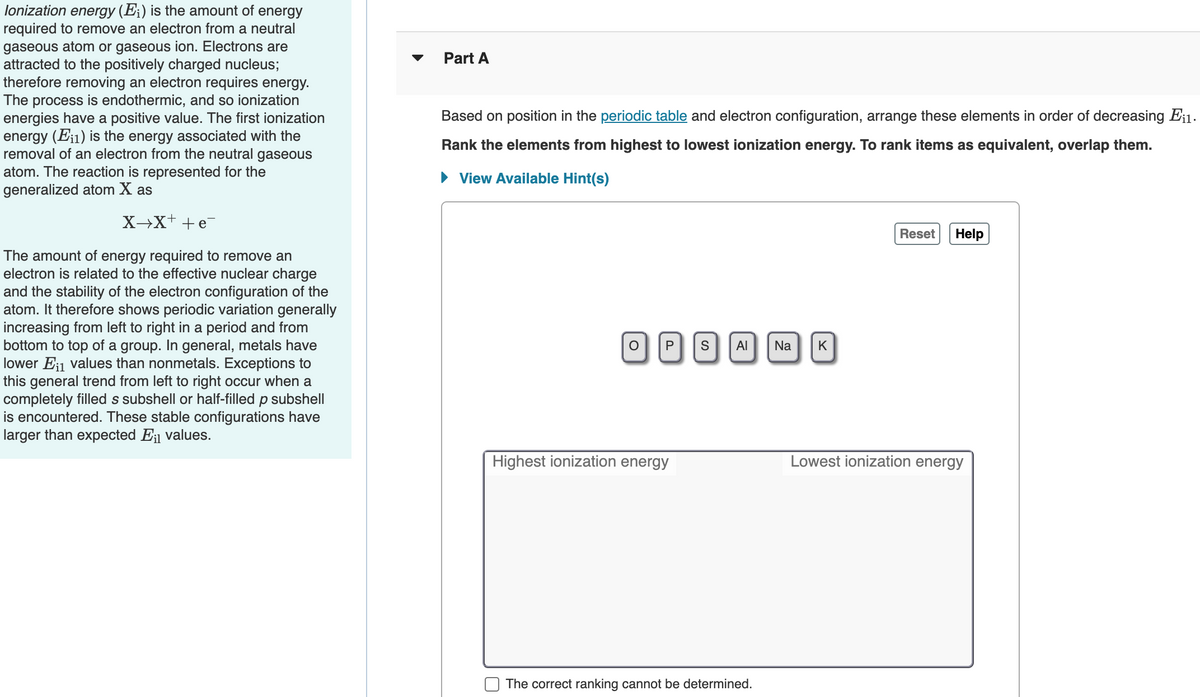
Transcribed Image Text:lonization energy (E;) is the amount of energy
required to remove an electron from a neutral
gaseous atom or gaseous ion. Electrons are
attracted to the positively charged nucleus;
therefore removing an electron requires energy.
The process is endothermic, and so ionization
energies have a positive value. The first ionization
energy (Ei1) is the energy associated with the
removal of an electron from the neutral gaseous
atom. The reaction is represented for the
generalized atom X as
Part A
Based on position in the periodic table and electron configuration, arrange these elements in order of decreasing E¡1.
Rank the elements from highest to lowest ionization energy. To rank items as equivalent, overlap them.
• View Available Hint(s)
X→X+ +e
Reset
Help
The amount of energy required to remove an
electron is related to the effective nuclear charge
and the stability of the electron configuration of the
atom. It therefore shows periodic variation generally
increasing from left to right in a period and from
bottom to top of a group. In general, metals have
lower Ei1 values than nonmetals. Exceptions to
this general trend from left to right occur when a
completely filled s subshell or half-filled p subshell
is encountered. These stable configurations have
larger than expected E; values.
P
Al
Na
K
Highest ionization energy
Lowest ionization energy
The correct ranking cannot be determined.
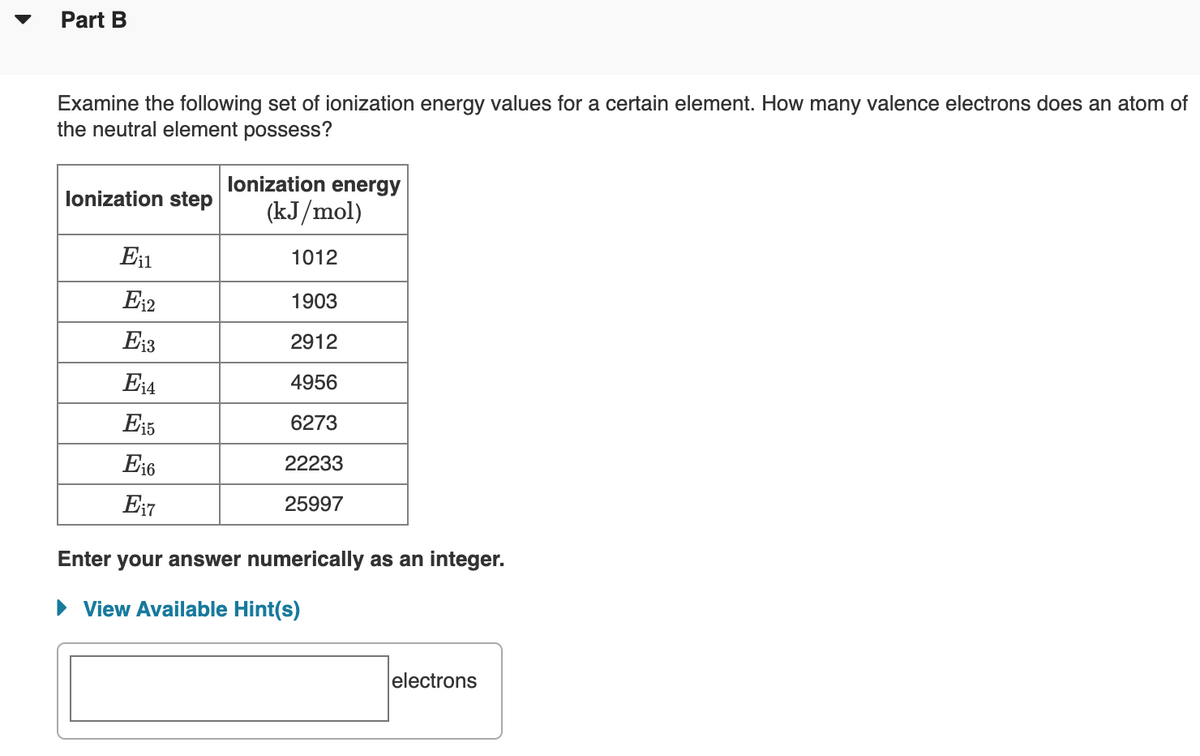
Transcribed Image Text:Part B
Examine the following set of ionization energy values for a certain element. How many valence electrons does an atom of
the neutral element possess?
lonization energy
lonization step
(kJ/mol)
En
1012
E12
1903
E13
2912
E14
4956
Ei5
6273
E16
22233
E17
25997
Enter your answer numerically as an integer.
• View Available Hint(s)
electrons
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning