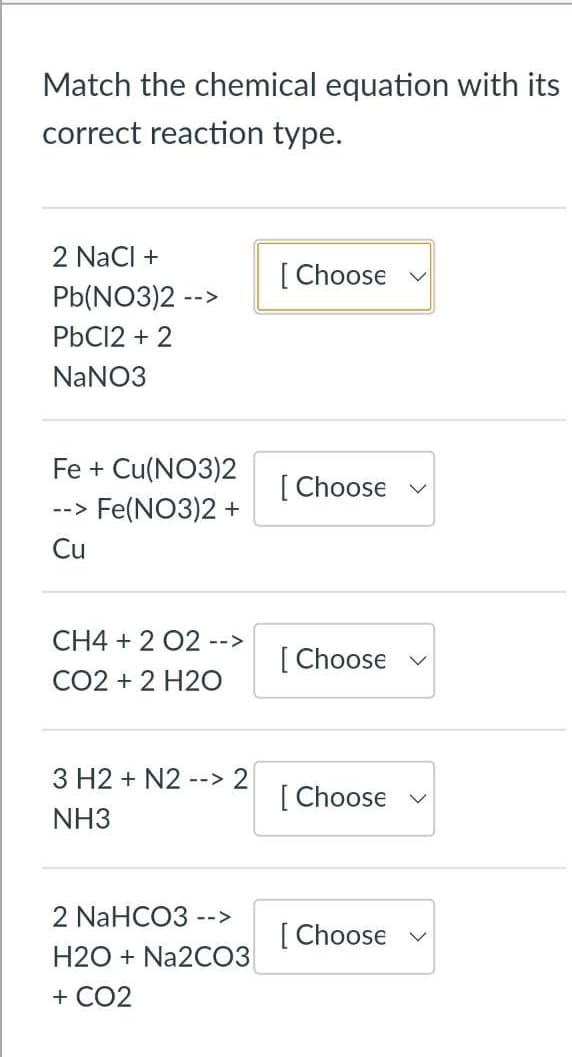
Match the chemical equation with its correct reaction type. 2 Naci + [ Choose Pb(NO3)2 -- PBC12 + 2 NaNO3 Fe + Cu(NO3)2 [ --> Fe(NO3)2 + [ Choose v Cu CH4 + 2 02 --> [ Choose v CO2 + 2 H2O 3 H2 + N2--> 2 [ Choose v NH3 2 NaHCO3 --> [ Choose v H2O + Na2C03 + CO2
Q: the determination of iron in limonite, 0.5166 g of minerals dissolve in acid and Fe2+ is titrated…
A: First find the moles of K2Cr2O7 utilized. Then using reaction stoichiometry finally the moles of…
Q: 0.683 grams of iron mineral dissolves with acid. Fe2+ is titrated with Ce(SO4)2 solution.…
A: Answer is explained below.
Q: A feldspar sample was analyzed. In the analysis of 0.4270-g sample, a mixture of KCl and NaCl is…
A:
Q: I + Cr,0,- Cr" + I, + H2O (acidic)
A: The given redox reaction is I-+Cr2O72-→Cr3++I2+H2O Oxidation half reaction I-→I2(as oxidation…
Q: Hydroxyapatite, Cao H),, has a solubility constant of Rp according to Ca(PO,)OH),(s) 10C2 (aq) + 6…
A: We have given that Ca10(PO4)6(OH)2(s) ⇌ 10Ca2+(aq) + 6PO3-4(aq) + 2OH-(aq) Ksp = 2.34×10-59…
Q: 3. For 52 Te, write: a. the valence electron configuration b. the oxidation states (underline the…
A: The answer to the following question is
Q: KĄFE(CN)6?
A: Correct answer is (b) +2.
Q: Balance the reaction between PhOz and Cu to form P and Cu* in acidic solution. When you have…
A: When the reaction is balanced in the acidic medium then it must include H+ ions in the balanced…
Q: Determine the oxidation number for each of the transition metal atoms or ions. a.) [ V(OH2)6 ](NO3)a…
A: Applying definition of coordination no. i.e. no.of co-ordination bonds is equal to coordination…
Q: Many transition metals and their compounds are used as catalysts. Given MnO2, FeCl3, Pt, and Ni,…
A: Catalyst are the compounds that lowers the activation energy of reaction to increase the rate of the…
Q: In the determination of iron in limonite, 0.5166 g of mineral is dissolved in acid and Fe2 + is…
A: This question is related to finding out the percentage of Fe2+ in the given iron ore sample. First…
Q: How to write a chemical equation for transition metal ions found in a solution? The ions found were…
A: Given : solution of Co2+ (aq) and Cu2+ (aq). 1) Adding NaOH : Since addition of NaOH will bring OH-…
Q: Which element will produce a new compound when added to a beaker containing an aqueous solution of…
A: The reactivity series is the arrangement of the elements from most reactive to least reactive.…
Q: أحد المعقدات التالية يظهر تشوه جان تیلر ؟ [Cr(OH2)6]3+ [Mn(OH2)6]3+ O [Mn(CN)6]3- C [Fe(CN)6]2-…
A:
Q: What is the oxidation state of Ti in TiO2? Group of answer choices 2- 2 4 0 4- 2,
A: "Since, you posted multiple questions . So, we will solve the first question for you. If you want…
Q: Determine the percentage of iron in a sample of limonite from the following data. Weight of sample…
A: Weight of sample = 0.5000 g Molar Mass of Iron = 55.85 g Molar mass of FeO = 71.84 g Volume of…
Q: draw the structure of the possible product forthe following reaction, and how does the oxidation…
A: We have to draw the structure of possible product .and identify how the oxidation state changeg.
Q: Identify the oxidizing agent. 2 Al3+(aq) + 2 Fe(s) →→ 2 Al(s) + 3 Fe2+(aq)
A: An oxidizing agent is the one that oxidizes the other ralumeactants. Hence we have to find the…
Q: Mass of KxFe(C2O4)y · zH2O : 4.70 g Mass of sample : 0.175 g Mass of FeCl3 used in preparation :…
A: The given compound first undergoes dissociation in its aqueous solution as,
Q: A 3.00-g sample of an alloy (containing only Pb and Sn) was dissolved in nitric acid (HNO 3).…
A: Given: Mass of alloy sample = 3.00 gMass of PbSO4 precipitated = 1.90 gMolar mass of PbSO4 = 303.3…
Q: Why is the oxidation state of Fe 2.5?
A: Iron is a transition metal. It can exists in more than one oxidation state in the bonded state.
Q: 5. Name following compounds and find the oxidation states of them. (a) [Co(NH3)6]Cl; (b)…
A: Here we are required to name the complex and also find the oxidation state of the central metal.
Q: Write a balanced equation for the reaction between nitrous acid and copper (II) hydroxide. O HNO3 +…
A:
Q: A 0.4560 g mixture of FeCl3 and FeBr3 was dissolved in water and reacted with excess NaOH. All of…
A: The balanced chemical equation for the reaction is: (FeCl3+FeBr3) (aq)+3NaOH(aq)-> Fe(OH)3(s) + 3…
Q: A 0.5962 g sample of iron ore is dissolved in perchloric acid (HCIO4). All iron present is oxidized…
A:
Q: Explain why solid CO2 is referred to as dry ice.
A: The rigid, incompressible objects present in the surrounding are classified as solid. The rigidity…
Q: Potassium dichromate is used to titrate a sample containing an unknown percentage of iron (AW.…
A: We have to tell which option is correct from given choices of options by telling percentage of iron…
Q: A 0.630 g sample of the ore is completely dissolved in concentrated HNO3(aq). The mixture is diluted…
A: Given data Mass of ore = 0.630 g Volume of solution = 50 ml = 0.05 L Absorbance of solution = 0.74 =…
Q: When Cr(III) is oxidized to Cr(VI), its solubility, mobility and toxicity are dramatically…
A: The solution of the question is given below:
Q: Ni content of the steel can be determined by the precipitation gravimetric analysis. For this…
A: The complex of Ni forming is Ni(DMG)2
Q: ion oxidation state of metal atom [Mac (NH).J [Fe(OH), (co)]
A: This question is related to coordination chemistry. A coordination complex consists of a central…
Q: A sample of iron ore weighing 0.6248 g is dissolved in acid, the iron reduced to Fe²⁺, and the…
A: Given data,Mass of iron sample=0.6248gVolume of K2Cr2O7=33.60mLNormality of K2Cr2O7=0.1502NFormula…
Q: A sample of magnetite (impure Fe3O4) weighing 0.5000 g is fused with oxidizing flux and the ferric…
A: Magnetite is an ore of Fe with the formula Fe3O4.
Q: 1,5419 g of magnetite (Fe3O4) ore; Fe+2 and Fe + 3 in concentrated HCL to form a mixture it's…
A:
Q: In a metallurgical process the mineral pyrite, FeS2, is roasted in air: FeS 2 +O 2 Fe 2 O 3 +SO 2…
A: Given reaction FeS2 + O2 -----> Fe2O3 + SO2 Balanced equation can be written as 4 FeS2 + 11…
Q: The black “smoke” that flows out of deep ocean hydrothermal vents is made of insoluble metal…
A: Given:- The black “smoke” that flows out of deep ocean hydrothermal vents is made of insoluble metal…
Q: In the alloy containing sn pb and cd, the mole ratio sn/pb is 2.73/1 and the mass ratio of pb/cd is…
A:
Q: In the laboratory you are given the task of separating Ca and Fe2+ ions in aqueous solution. For…
A: Separation of Ca2+ and Fe2+ ions in aqueous solution .Reagents used for separation : KCl, K2SO4…
Q: A volume of 2.5 ml was taken from a hypothetical sample containing (3 ) valent antimony and (3 )…
A:
Q: 22. Copper is obtained from sulfide ores through a two-step process consisting of roasting the ore…
A: Copper sulfide reacts with oxygen and carbon to give copper, sulfur dioxide, and carbon monoxide.…
Q: Ammoniacal nitrogen can be determined by treatment of the sample with chloroplatinic acid; the…
A: Stoichiometry of a balanced chemical gives the relationship between the amount of reactants and…
Q: Balance the equation for the following reaction under acidic aqueous conditions. Fe2+ + Cr20,2- →…
A: Balance equation: 6Fe2+ + Cr2O72- + 14H+ → 6Fe3+ + 2Cr3+ + 7H2O Correct answer is (d) 6.
Q: What is the balanced net ionic equation of NH3 reacting with Fe(NO3)3 and reacting with CuS
A:
Q: Write the chemical equation It got cut off (K4[Fe(CN)6](aq))
A:
Q: Determine the oxidation state of the coordinated metal atom in each of the following compounds:(a)…
A: (a) K[Co(NH3)2(CN)4] Let oxidation state of metal is y. +1 + y + ( 0 x 2) + (-1 x 4) = 0 y = +3…
![[ Choose]
synthesis
combustion
double replacement
single displacement
decomposition](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F03ba95c9-584b-483d-8f8a-8f0f9c4c55b7%2F60f039f1-d67a-4fba-8666-e64eb5e4c852%2Fece5gr7_processed.jpeg&w=3840&q=75)
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- The Ksp values of two solid carbonates, A2CO3 and BCO3, were determined by preparing 250.0-mL saturated aqueous solutions of both solids and then determining the concentration of carbonate by titrating it with 0.01023 M HCl up to methyl orange endpoint. Solutions of A2CO3 and BCO3 required 6.184 mL and 3.456 mL of the titrant, respectively.d. Instead of distilled water, 250.0 mL of 0.10 M ANO3 was used to prepare a saturatedsolution of A2CO3 while 250.0 mL of 0.10 M B(NO3)2 was used for BCO3. Which will have a higher solubility?The UV–visible spectrum of GLB-1 is given in Figure 2. i.What colour(s) will the deoxy and oxy forms of GLB-1 be? Note, an electromagnetic spectrum is reproduced in the Assessment area on the S315 website. ii.By comparing the bands on the right-hand side (around 550 nm) with those of oxy- and deoxymyoglobin, suggest what type of transition gives rise to these bands.Given that the Ksp for Ag2S is 6×10^-51, Kf=1.1×10^5 for AgCl2-, and Ka1 and Ka2 for H2S are 9.5×10^-8 and 1×10^-19, respectively, calculate the equilibrium constant for the following reaction .
- Mass of maleic anhydride used: 5.1296 gram Volume of furan used: 3.25 ml Volume of THF used: approx. 16ml The reaction mixture was allowed to stand undisturbed for one week, whereupon, gem-like, colorless to white, prismatic crystals were isolated from the solvent matrix and allowed to air dry. When dry, the mass of the crystals was determined to be: 4.6732 gram. One small crystal was selected and ground to a fine powder for melting point measurement. The powder was observed to melt at 117 – 118 deg C. About 1ml of a saturated solution of product dissolved in DMSO-d6 was prepared and placed in an NMR tube. Subsequently, proton, carbon-13 and DEPT NMR spectra were acquired. Analysis Write the reaction using structural formulas. Calculate percent yield (show work) What does the measured melting point suggest?Butan-1-ol can be oxidised by acidified potassium dichromate( VI) using two different methods. (a) In the first method, butan-1-ol is added dropwise to acidified potassium dichromate( VI) and the product is distilled off immediately. (i) Using the symbol [O] for the oxidising agent, write an equation for this oxidation of butan-1-ol, showing clearly the structure of the product.State what colour change you would observe. Equation ........................................................................................................... Colour change .................................................................................................. (ii) Butan-1-ol and butan-2-ol give different products on oxidation by this first method. By stating a reagent and the observation with each compound, give a simple test to distinguish between these two oxidation products. Reagent…which subsatomic particles contriubte to the chrage of Cu2+, which do not
- A 0.703 g sample of a commercial detergent was red-calcined to destroy its components. organic. Treatment of the residue with hot HCl gave phosphorus in solution as H3PO4. the phosphate was precipitated as MgNH4PO4-6H2O by the addition of Mg + 2 or followed by aqueous NH3. The precipitate was filtered, washed, and converted to Mg2P2O7 by calcination at 1000 C. The residue weighed 0.432 g. Calculate the percentage of phosphorus in the sample.(i) Tungsten hexacarbonyl can be prepared by reacting WCl6 with triethylaluminium in the presence of carbon monoxide. By-products of this reaction are aluminium trichloride and n-butane. Give the two relevant half-equations and then construct the full redox equation.(ii) Tetrarhodium dodecacarbonyl is formed upon treatment of Na+ [Rh(CO)2Cl2]- with H2O under a stream of CO. Give the two relevant half-equations and then construct the full redox equation for this reaction. Take into account that some of the CO acts as a reducing agent.Quickly plezz!! The levigating agent role in Suppositories manufacturing ??
- Suggest a method by which Ni2+ can be precipitated homogeneously as NiS. a. Add sodium sulfste to the solution to precipitate NiSO4. Heat the nickel(II) sulfate to liberate O2 and yield pure NiS. b. The slow hydrolysis of thioacetamide can be used to generate a source of hydrogen sulfide gas, which provides the S²- to complex with the Ni2+. c. Add thiosulfuric acid to the solution to react with Ni2+ and precipitate NiS. d. Heat S8 in the presence of Ni2+, in an inert atmosphere, to produce NiS.In the presence of 18-crown-6, potassium permanganate dissolves in benzene to give“purple benzene,” a useful reagent for oxidizing alkenes in an aprotic environment. Use adrawing of the complex to show why KMnO4 dissolves in benzene and why the reactivityof the permanganate ion is enhanced.you've been tasked to compound 30 grams of a 0.5% Zinc oxide ointment. you have a stock preparation of zinc oxide 5% strength to mix with petrolatum to dilute the ordered strength. In what (parts) ratio could this be compounded?

