Q: The element lithium has two stable isotopes, lithium-7 with an atomic mass of u and lithium-6 with a...
A: Hi.. In the given question, numerical value for atomic mass of both the isotopes are not available.S...
Q: How many gold atoms are in an 0.214-ounce, 18 K gold bracelet? (18 K gold is 75% gold by mass.) Exp...
A: Given,A 0.214 ounce gold bracelet which contains 75% gold by mass.First convert the given weight of ...
Q: Calculate the boiling and freezing points of 0.22m glycerol in ethanol. Normal BP ethanol: 78.4 Kb ...
A: Given:Normal boiling point of ethanol (Tbo) = 78.4 ℃ = 351.55 K.Kb of ethanol = 1.22 K Kg/mol.Normal...
Q: How would you prepare 10 mL of a 0.25% m/v HCl solution if 1% m/v HCl was available? How much 1% m/v...
A: It is given that:Volume (V1) of solution = 10 mLConcentration of HCl solution (M1) = 0.25 %Concentra...
Q: the world's total petroleum reserve is estimated at 2.0x10^22 joules. At the present rate of 1.8x10^...
A:
Q: Give the chemical symbol for the element with the ground-state electron configuration [Ne]3s23p2. sy...
A:
Q: If a sample of gallium contains 1.56×104 atoms of gallium-69 and 1.02×104 atoms of gallium-71, what ...
A: Given,1.56×104 atoms of gallium 69.1.02×104 atoms of gallium 71.
Q: Whats the difference between organic and inorganic chemistry?
A: The major difference is organic chemistry deals with organic compounds, that is compounds containing...
Q: Under certain conditions, the substance bromine trifluoride can be broken down to form bromine and f...
A: It is given that:Mass of bromine trifluoride = 28.5 g Mass of bromine = 16.6...
Q: Naturally occurring bromine is composed of two isotopes: 50.69 % Br-79 (mass of 78.9183 amu) and 49....
A: Given,Naturally occurring bromine is composed of two isotopes:50.69 % Br-79 (mass of 78.9183 amu) an...
Q: The activation energy for the gas phase isomerization of methyl cis-cinnamate is 174 kJ/mol.cis-C6H5...
A: The rate constant of the reaction can be calculated by using the Arrhenius equation.
Q: So I’m taking organic chemistry and I’m doing my first lab. It’s on crystallization of sulfanilamide...
A: The techniques used for the purification of solid compounds is known as crystallization. The princip...
Q: You wish to make 10.0L of phosphate buffer with a pH of 7.4 from NaH2PO4 (molecular weight, 119.98g/...
A: We have to calculate the mass of NaH2PO4 (molecular weight, 119.98g/mol) and Na2HPO4 (molecular weig...
Q: . Gold has a density of 19.6 g/cm. What is the mass of 9.5 cm3 of gold?
A: Given, The density of gold is 19.6 g/cm3.Mass of gold is 9.5 cm3. Density is mass divided by volume ...
Q: #6 (a) How to add a amine functional group to a benzene ring?
A: Electrophilic aromatic substitution takes place when an electrophile displaces a hydrogen atom of an...
Q: An alloy of Sn, Pb, and Cd has a mole ratio of Sn to Pb of 2.73:1.00, and the mass ratio of Pb to Cd...
A: Given,An alloy of Sn, Pb, and Cd has a mole ratio of Sn to Pb of 2.73:1.00, and the mass ratio of Pb...
Q: Suppose you are trying to find the volume of a box based on the given measurements for the length, w...
A: Given,length, l = 2.20 inwidth, w = 3.71 inheight, h = 10.4 in + 1 in
Q: a 22.5 grams sample of ammonium carbonate contains_____________moles of ammonium ions.
A: Conversion of mass to moles of ammonium carbonate:
Q: If a gas occupies 47.6 dm3 at SATP, what is its volume at 1.0 bar and 500 K? The acronym for new sta...
A:
Q: Gaseous methane CH4 will react with gaseous oxygen O2 to produce gaseous carbon dioxide CO2 and gase...
A: Given,Mass of methane = 7.1 gMass of oxygen = 16.3 gFirst write balanced equation for the given reac...
Q: please answer the questions given. thank you
A: The Rydberg equation for determining the wavelength of light is given by
Q: Look up the density of liquid water at 25.0 degrees C. What mass of water will occupy 250L at 25.0 d...
A: The term density is defined as the ratio of mass to the volume of given substance. The mathematical ...
Q: 4. Copper is found in nature as two isotopes, 63Cu (69.0%) with a mass of 62.93 amu and 65Cu (31.0%)...
A: The average atomic mass of two isotopes can be calculated using the following expression:
Q: If 7.99 ml of a 0.058 M solution of oxalic acid is mixed with 11.6ml of water and 5.2ml of potassium...
A: 7.99 ml of a 0.058 M solution of oxalic acid is mixed with 11.6 ml of water and 5.2 ml of potassium ...
Q: What are some enviromental, social, and economic risks of polystyrene watse. Refrences included woul...
A: Polystyrene is a versatile plastic used to make a wide variety of consumer products. But along with ...
Q: Balmer observed a hydrogen violet emission line from n=6 to n=2, but not for transition from n=7 to ...
A: The wave length of the emission from n=6 to n=2 is calculated as
Q: What is isotopic notation example and definitions please?
A: Isotope Notation is also known as nuclear notation. It is written form of an element that describes ...
Q: Unit Analysis 15. The white blood cell concentration in normal blood is approximately12,000 cells/mm...
A: Given:Number of white blood cell in 1 mm3 = 12,000.Normal adult blood volume = 5 L.
Q: Explain the bicarbonate buffer system
A: Buffer solution is defined as a solution which resist the change in pH of the solution on the additi...
Q: I am poor at physics chem, so please explain as much as possible.
A: The equation for the formation of diborane is given below.2 B(s) + 3 H2(g) ---> B2H6 (g)
Q: When sulfuric acid reacts with zinc hydroxide, zinc sulfate and water are produced. The balanced equ...
A: Here, we have to determine number of moles of zinc hydroxide, zinc sulfate and water.
Q: X For You - Xfinity Stream b Answered: Microsoft PowerPoint x Smartwork5 X Digital Resources for Che...
A: Entropy means dis-orderness here the ions disorderness (moments).The number of ions increase means t...
Q: Fill in the compound formulas in the table below. Cation Formula Anion Formula Compound Formula ...
A: Cation formula Anion formula Formula of the compoundMn2+ CN- ...
Q: Calculate the wavenumber (cm^-1) and excitation energy (kj/mol) for each of the following max wavel...
A: Electromagnetic spectrum involves different radiations from the lowest to the highest frequency valu...
Q: Carry out the division and enter the quotient with the proper number of significant figures in scien...
A: This is a division operation and in division, the number of significant figures are equal to the lea...
Q: The element X forms the compound XOCl2 containing 59.6% Cl. What is element X?
A: The empirical formula is the chemical formula of any compound that represents the proportions of eac...
Q: Table salt contains 39.33 g of sodium per 100 g of salt. The U.S. Food and Drug Administration (FDA)...
A: Mass percentage of sodium in salt can be calculated as follows:
Q: The density of mercury is 13.6g/ cm^3. What volume (in quarts) is occupied by 101 g of Hg?
A: As density is the ratio of mass and volume of the substance which is represented by equation 1 in wh...
Q: Sa 1 points An electron completes a transition from the n = 1 energy level where it has energy E1 =-...
A: Given that,Energy in n = 1 state is E1 = -2.18 × 10-18 JEnergy in n = 3 state is E3 = -2.42 × 10-19 ...
Q: Using an integrated rate law for a first-order reaction Consider this reaction: 2H,PO (ag) P2O, (ag)...
A: Given,Rate constant (k) = 0.0189. s-1
Q: Which of the following set of quantum numbers (ordered n, l, m) are possible for an electron in an a...
A: An atom is mainly composed of three sub-particles; electrons, protons and neutrons. These particles ...
Q: Convert the following condensed formula to a Lewis structure. Be sure to include all lone pair elect...
A: The condensed formula of the compound is given as follows:
Q: O KINETICS AND EQUILIBRIUM Darlana Using a second-order integrated rate law to find concentration......
A: Chemical kinetics is the branch of chemistry that mainly deals with the rate of reaction and change ...
Q: (0.0041×20000.0)+(2817×14). Express your answer to the appropriate number of significant digits.
A: The given calculation consists of multiplication and addition.The first part is multiplication. One...
Q: Using the correct number of significant figures, calculate the perimeter of a small, rectangular mir...
A: In addition or subtraction involving significant figures, the final answer should be reported as hav...
Q: Calculate the mass of 72.1 mmol SO2
A: It is given that:Number of moles(n) of SO2 = 72.1 mmol
Q: Convert 7.57×10−3 m7.57×10−3 m to the equivalent length in nanometers
A: Given,7.57×10−3
Q: A solution containing 3.30 g of an unknown nonelectrolyte liquid and 10.00 g water has a freezing ...
A: When the non-electrolytes liquid is added in solution there is the change in freezing point which is...
Q: 1.65 g H2 is allowed to react with 10.0 g N2, producing 1.79 g NH3. What is the theoretical yield in...
A: The reaction between H2 and N2 is as follows,
Q: How do I make a line structure for (CH3)3CCO2CH(CH2CH3)2? I’m confused on how you should angle the l...
A: Lone pairs present on the central atom affects the bond angle. The lone pair electrons repel the bo...
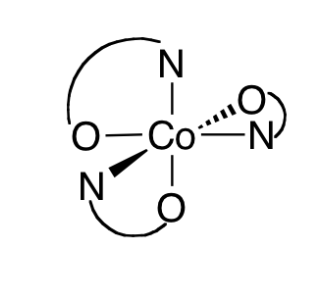
For selected molecule identify:
a) all the symmetry elements present
b) appropriate point group
c) which of the symmetry elements listed in (a) are sufficient to determine the point group.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images


