N но What is the chemical formula for the limiting reactant in the reaction shown? chemical formula: NO Write the balanced chemical equation for the reaction, using lowest whole-number coefficients. chemical equation: 4NO + SH, → 4H,0 + 2N, + H, - Incorrect
N но What is the chemical formula for the limiting reactant in the reaction shown? chemical formula: NO Write the balanced chemical equation for the reaction, using lowest whole-number coefficients. chemical equation: 4NO + SH, → 4H,0 + 2N, + H, - Incorrect
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter4: Stoichiometry
Section: Chapter Questions
Problem 4.70PAE: 4.70 The particulate scale drawing shown depicts the products of a reaction between H2 and O2...
Related questions
Question
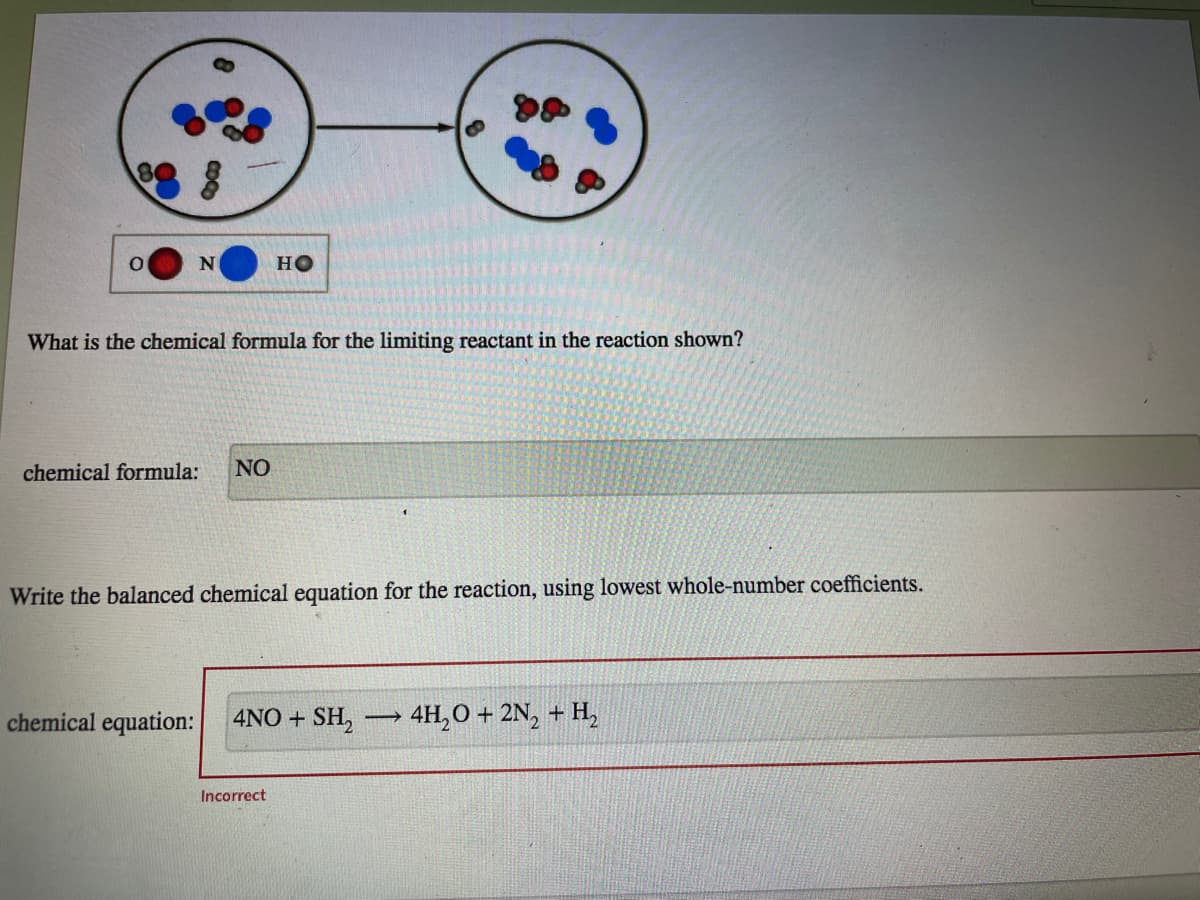
Transcribed Image Text:HO
What is the chemical formula for the limiting reactant in the reaction shown?
chemical formula:
NO
Write the balanced chemical equation for the reaction, using lowest whole-number coefficients.
chemical equation:
4NO + SH,
4H,O + 2N, + H,
Incorrect
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning