NaBr H,0 solution Use these sketches to answer the questions in the table below. The enthalpy of solution AHolo is negative when NaBr dissolves in water. Use this information to list the stages in order of increasing enthalpy. ? absorbed Would heat be absorbed or released if the system moved from Stage C to D? released neither absorbed nor released none ionic bonding force covalent bonding force metal bonding force What force would oppose or favor the system moving from Stage C to D? Check all that apply. hydrogen-bonding force dipole force ion-dipole force dispersion force
NaBr H,0 solution Use these sketches to answer the questions in the table below. The enthalpy of solution AHolo is negative when NaBr dissolves in water. Use this information to list the stages in order of increasing enthalpy. ? absorbed Would heat be absorbed or released if the system moved from Stage C to D? released neither absorbed nor released none ionic bonding force covalent bonding force metal bonding force What force would oppose or favor the system moving from Stage C to D? Check all that apply. hydrogen-bonding force dipole force ion-dipole force dispersion force
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter8: Thermochemistry
Section: Chapter Questions
Problem 8QAP: Sodium chloride is added in cooking to enhance the flavor of food. When 10.00 g of NaCl are...
Related questions
Question
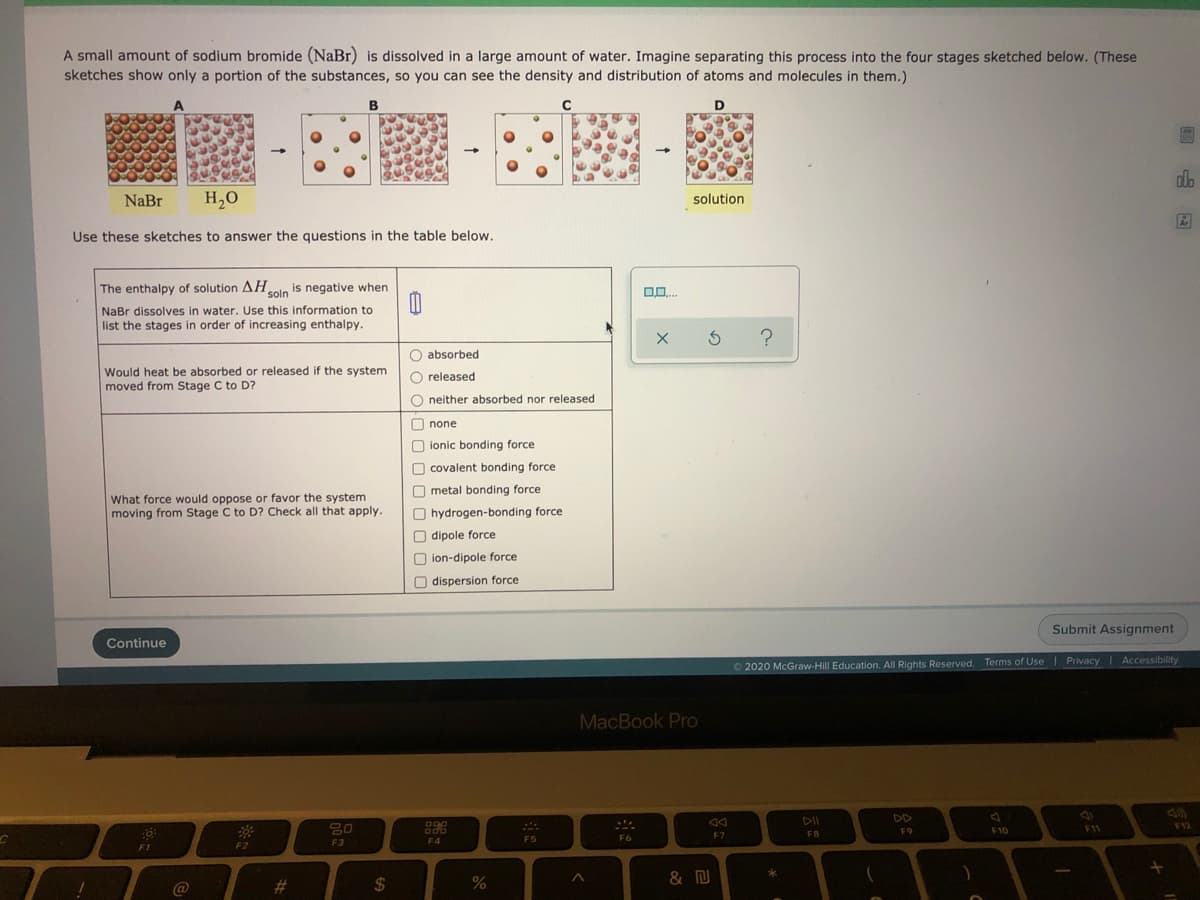
Transcribed Image Text:NaBr
H,0
solution
Use these sketches to answer the questions in the table below.
The enthalpy of solution AHolo is negative when
NaBr dissolves in water. Use this information to
list the stages in order of increasing enthalpy.
?
absorbed
Would heat be absorbed or released if the system
moved from Stage C to D?
released
neither absorbed nor released
none
ionic bonding force
covalent bonding force
metal bonding force
What force would oppose or favor the system
moving from Stage C to D? Check all that apply.
hydrogen-bonding force
dipole force
ion-dipole force
dispersion force
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning