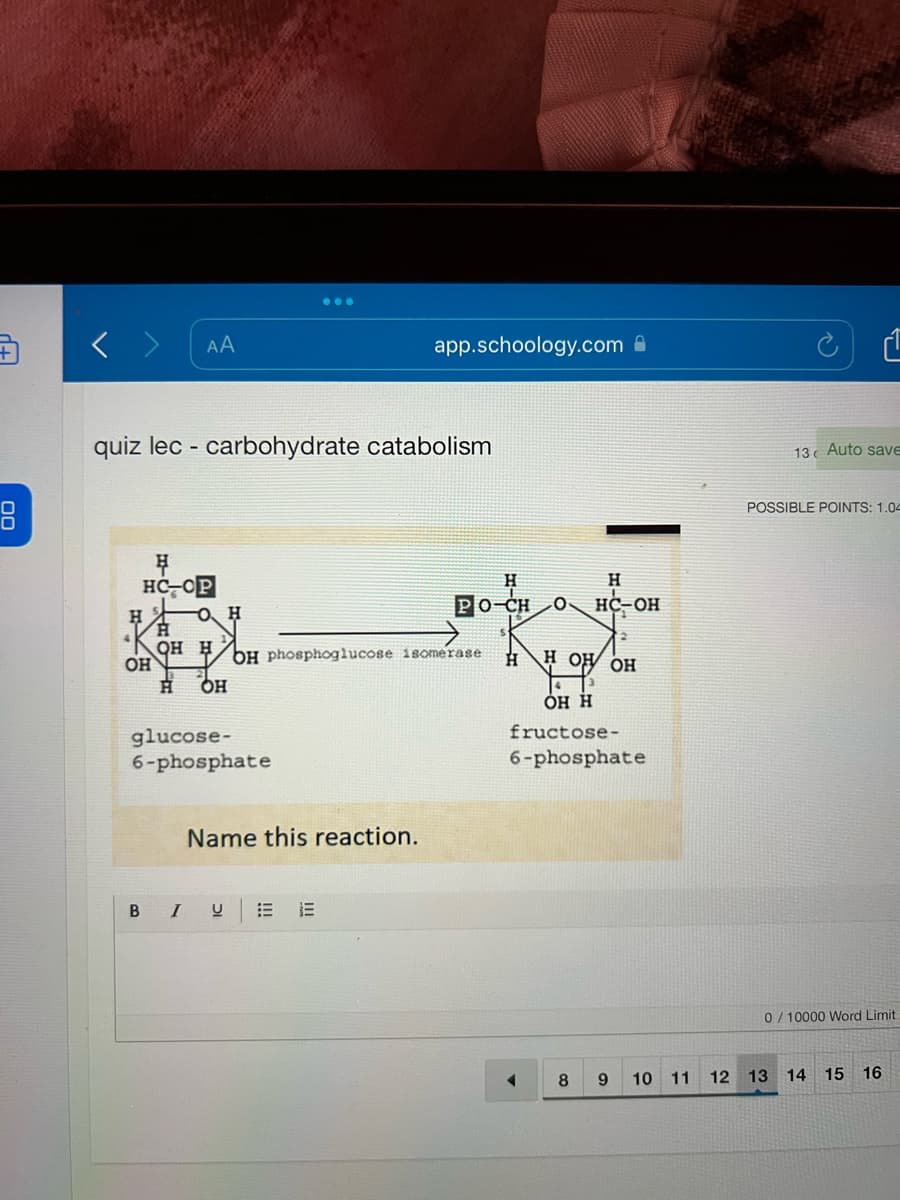
name the reaction of the carbohydrate catabolism
Q: The reaction catalyzed by citrate syn- thase, shown on the right, is the first step of the TCA…
A: In glycolysis, a 6-carbon molecule of glucose-6-phosphate is broken down into 3-carbon pyruvate. It…
Q: Which of the following describes the interaction between the amino acid last eluted and the anion…
A: In Ion exchange chromatography the molecules are separated according to their charge. The matrix…
Q: If there are still contaminating proteins with your LDH fraction after going through 3 different…
A: Proteins are high molecular weight biomolecules made up of amino acid residues linked via peptide…
Q: n a table, write down the enzyme that acts on proteins and also mention the final product of…
A: Introduction: Enzymes are proteins that act as biological catalysts to fasten the rate of the…
Q: Which of these is NOT true of nucleosomes? A. Some post-translational modifications to histone…
A: Nucleosome is the basic subunit of chromatin. It is the basic unit of DNA packaging. Nucleosomes…
Q: Draw the structure of a triacylglycerol containing stearic acid, palmitic acid, and oleic acid.
A: Animal fat and vegetable oil fall into the category of simple fats or triglycerides. Triglycerides…
Q: Which of the following sequences is less favorable to find in a folded beta? Select one:…
A: Two types of secondary structures are abundant in protein: alpha helix and beta sheets. The alpha…
Q: The ff: table showed data of enzyme catalytic reaction. The rate of reaction (v) decreased with the…
A: For a one-substrate enzyme-catalyzed reaction, the Michaelis-Menton equation shows the quantitative…
Q: Draw the fractional binding curve with protein that bind to a molecule of ligand L.
A: A ligand is a molecule that binds to a receptor. The specificity of the ligand varies. One ligand…
Q: Chemicals such as cyanide and carbon monoxide can be deadly. This is because they directly inhibit:…
A: CO is a colorless, odorless gas that is formed by incomplete combustion of carbon compounds. CO…
Q: Choose below the correct order of the oxygen binding proteins from lowest to highest affinity for…
A: Hemoglobin is a tetrameric oxygen transport protein found in erythrocytes, on the contrary,…
Q: True or False: Spectrophotometric assays always track the rate of reaction through a. True b. False…
A: Spectrophotometry is the method that uses light beam to measure the concentration of a chemical or a…
Q: For paper chromatography, the obtained Rf value of an amino acid with uncharged nonpolar R group is…
A: Chromatography is performed to separate a mixture of closely related substances. Paper…
Q: 1. Minor changes in primary structure can have pronounced effects on biological function. Tetrameric…
A: Hemoglobin is a tertameric protein made up of 4 subunits namely α2β2. The αβ form a dimer and 2 such…
Q: Which of the following single-stranded DNA sequences is most likely to form a stem-loop structure?…
A: DNA is also called deoxyribonucleic acid. DNA act as the genetic material in all organisms except…
Q: Consider the reaction: H H₂C-(CH₂)C=C—C—SCOA H ➤ H3C- 13C-(CH₂) What kind of reaction is being…
A: The given structure is a fatty acyl Coenzyme A. Fatty acids are charged with acetyl CoA which are…
Q: There are numerous methods for sequencing DNA, including classical Sanger sequencing, automated…
A: The sequencing of DNA can be done using various approaches ranging from conventional Sanger…
Q: For class I MHC, which of the following statements are correct? P. They are expressed on all…
A: MHC are proteins encoded by a huge complex of genes called the major histocompatibility complex…
Q: When performing cloning experiments, it is not always necessary to treat sources of DNA with the…
A: Isoschizomers are enzymes that can share the same recognition site in restriction digestions.
Q: The metabolic amino acid ornithine is a direct precursor of the polyamine putrescine, shown here. +…
A: Ornithischians is a metabolically active non proteogenic amino acid so that it would not be…
Q: Cells absorb water by osmosis, but plant cells and animal cells react differently to the influx of…
A: Introduction: The term osmosis refers to the movement of water molecules across a selectively…
Q: Give an overview of Tumor necrosis factor (TNF) signaling, and discuss the nature of the TNF ligand…
A: TNF signaling is an apoptotic signaling pathway that causes programmed cell death due to external…
Q: 9. The NUDT18 protein was purified by affinity chromatography (Hiss tagged) followed by gel…
A: Affinity chromatography is a method of separating a biomolecule from a mixture, based on its ability…
Q: 10) Determine bending rigidity of double stranded DNA molecule with persistence length of 45 nm (80%…
A: Deoxyribonucleic acid (DNA) is a double stranded polynucleotide coiled around a central axis to form…
Q: Identify the type of base and sugar group present in each nucleotide or nucleoside. HO. H₂C -CH₂ ОН…
A: A nucleotide consists of a base, a molecule of sugar and one of phosphoric acid. The sugar can be…
Q: ermination of translation occurs when the ribosome reaches one of _____ potential stop codons. At…
A: Introduction: The process of translation involves messenger RNA (mRNA) getting translated into…
Q: 2. The Km of a particular enzyme is 2.0 x 105 M. If the initial velocity (Vo) is 0.16 mmol/min for…
A: For a one-substrate enzyme-catalyzed reaction, the Michaelis-Menton equation shows the quantitative…
Q: IR spectrum spectral analysis of aniline and acetanilide. Please ALSO identify functional group.
A: For analysis of ir spectrum of aniline and acetanilide , we have to know the structure and…
Q: If glucose labeled with 14C in C-1 is metabolized to ethanol and CO2 via fermentation, which carbon…
A: Pyruvate is the end product of glycolysis. Under anaerobic conditions it can have two possible…
Q: CONNECT Explain why the proton gradient formed du chemiosmosis represents a state of low entropy.…
A: Oxidation of glucose into pyruvate followed by oxidation of pyruvate into acetyl CoA and then…
Q: How many ATP will be yielded in 2 moles of an 17:0 and 18:0 carbon chain?
A: Beta oxidation is a process of oxidation of fatty acids into acetyl CoA. The total amount of ATP…
Q: 4. An enzyme-substrate complex has a Kg = 100 nM. A competitive inhibitor with which of the…
A: KD (Dissociation constant) is the amount of ligand molecules that is required to fill the binding…
Q: The structure of purine is shown. 2 N-1 N-3 N-7 N-9 6 5 N 4 3 7 N -N9 ZI 8 Which atoms of the purine…
A: Purines: The two groups of nitrogenous bases, which also include the two groups of nucleotide…
Q: Question 9 Briefly discuss the discovery and development of selective inhibitors of sodium-glucose…
A: Sodium-glucose linked transporters (SGLTs) , or to be more specific Sodium-glucose linked…
Q: The steady-state kinetics of an enzyme are studied in the absence and presence of an inhibitor B…
A: Enzymes are protein molecules that increase the rate of reaction by decreasing the activation…
Q: Prepare 1.5L 2X Base stock in a media bottle: Fill out the last column of this table: Component…
A: A buffer is a solution that can resist pH change upon the addition of an acids or bases. Buffers…
Q: Structural and functional features of the lysosomes include I. internal pH is higher than the…
A: Lysosomes are cell organelles that help in cellular digestion of foreign particles, food particles…
Q: Questions: 1. What test tube will show positive starch hydrolysis and negative starch hydrolysis?…
A: Starch is a storage-type homopolysaccharide that contains two types of glucose polymers: amylose:…
Q: Identify the reasons why the DNA molecule you selected would lead to DNA synthesis. It is…
A: An important characteristic of DNA replication is that it is semiconservative. Two strands of DNA…
Q: HO. CH₂ OH NH₂ ОН N Molecule 3 O pyrimidine, deoxyribose pyrimidine, ribose O purine, deoxyribose O…
A: Nucleic acids are huge macromolecules that perform crucial functions in all organisms and viruses. A…
Q: 4. Blood glucose level, glucose transporter proteins (classification, localisation and biological…
A: Blood Glucose is the major sugar found in our blood.It is the main source of energy used by a cell.…
Q: The highest level of structural complexity completely retained by the molten globule state of a…
A: The term "molten globule state of a protein "refers to the diverse types of partially-folded protein…
Q: In 1958, Meselson and Stahl conducted an experiment to determine which of the three proposed methods…
A: Three important experiments contributed to our current understanding of DNA structure and function:…
Q: [1,6-(C-14)-2,5-(C-13)]glucose Trace the course of through glycolysis and the TCA cycle. You need…
A: Carbon tracing is the method by which we trace the path taken by each carbon atom in a substrate, as…
Q: multimeric protein contains 20 or greater different amino acid residues At least 2 or more…
A: Introduction Proteins are the most abundant macromolecules in our body. Proteins are made up of…
Q: 20. P2 and P4 are bacteriophages of E. coli. They have the following properties: (1) When P2 phage…
A: Bacteriophage (or Phage): The viruses that infect bacteria are referred to as bacteriophages or…
Q: Which of the choices are types of posttranslational modifications a newly synthesized protein may…
A: Posttranslational modifications are the amino acid side chain modification in some proteins…
Q: A competitive inhibitor diminishes the rate of catalysis: A) By reducing the proportion of enzyme…
A: The enzymes are biological catalysts that increases the rate of biological reactions. The enzymatic…
Q: not all polypeptides form a secondary helical structure, explain why polypeptides containing ser,…
A: Two types of secondary structures are abundant in protein: alpha helix and beta sheets. The alpha…
Q: discuss the excitatory NMDA receptor, the role of the Mg++ ion, and how NMDA activation can produce…
A: Hi! Thank you for the question. We are authorized to answer three subparts at a time, since you have…
Step by step
Solved in 2 steps

- Energetic of Fructose-1 ,6-bis P Hydrolysis (Integrates with Chapter 3.) The standard free energy change (G) for hydrolysis of fructose-1. 6-bisphosphate (FBP) to fructose-S-phosphate (F-6-P) and P: is -16.7 KJ/mol: FBP + H2O fructose-6-P + Pi The standard free energy change (G) for ATP hydrolysis is -30.5 KJ/mol: ATP + H2O ADP + Pj What is the standard free energy change for the phosphofructokinase reaction: ATP + fructose-6-P ADP + FBP b. What is the equilibrium constant for this reaction? c. Assuming the intracellular concentrations of [ATP] and (ADP] are maintained constant at 4 mM and 1.6 mM, respectively, in a rat liver cell, what will be the ratio of [FBP]/[fructose-6-P] when the phosphofructokinase reaction reaches equilibrium?Radiolabeling with 14C-Glutamate Describe the labeling pattern that would result from the introduction into the TCA cycle of glutamate labeled at Cy with 14C.Comparing Glycolysis Entry Points for Sucrose Sucrose can enter glycolysis by either of two routes: Sucrose phosphorylase: Sucrose + Pi fructose + glucose-1-phosphate Invertase: Sucrose + H20 fructose + glucose Would either of these reactions offer Jin advantage over the other In the preparation of hexoses fur entry into glycolysis?
- For myristic acid, C 13H 27CO 2H: (a) How many molecules of acetyl CoA are formed from complete β-oxidation? (b) How many cycles of β-oxidation are needed for complete oxidation?In healthy adults, the concentration of glucose in blood is approximately80 to 110 milligrams per deciliter (mg/dl). After a carbohydrate-richmeal, however, the concentration may spike to 140 mg/dl. Describe thehormonal action that returns blood glucose to normal.Will an amino acid be glucogenic or ketogenic if it is catabolized to the following molecules?(a) Phosphoenolpyruvate(b) -Ketoglutarate (c) Succinyl-CoA(d) Acetyl-CoA(e) Oxaloacetate(f) Acetoacetate
- Consider the docosanoic acid, C21H43CO2H a. Label the a and B carbons b. Draw the acyl CoA derived from this fatty acid c. How many acetyl CoA molecules are formed by complete B-oxidation? d. How many cycles of B-oxidation are needed for complete oxidation? e. How many molecules of ATP are formed from the complete catabolism of this fatty acid?Consider the fatty acids: (a) Arachidic acid (C20H40O2); molar mass = 312.5 g/mol) (b) Palmitoleic acid(C16H30O2); molar mass = 256.4 g/mol). i. How many cycles of β -oxidation are needed for complete oxidation?ii. How many molecules of acetyl CoA are formed from its complete catabolism?iii. Calculate the number of molecules (moles) of ATP formed (net) by the completecatabolism of each fatty acid (show your calculation).iv. Calculate number of moles of ATP formed per gram of each fatty acid metabolized.Consider docosanoic acid C12H43CO2H a. Label the alpha and beta Carbons. Show the beta-oxidation in an EXPANDED structure. b. Draw each acyl CoA derived from this fatty acid. c. How many acetyl Co A molecules are formed by complete beta-oxidation? d. How many cycles of beta-oxidation are needed for complete oxidation? e. How many molecules of ATP are formed from the complete catabolism of this fatty acid? Show the complete computation. f. How many moles of ATP per gram of fatty acid is formed from the complete catabolism of the given fatty acid? g. What is the molar mass of the given fatty acid? Solution: Show here the complete computations, [from a to e]
- Considering the fatty acids: (a) Arachidic acid (C20H40O2); molar mass = 312.5 g/mol) (b) Palmitoleic acid (C16H30O2); molar mass = 256.4 g/mol). How many cycles of β -oxidation are needed for complete oxidation? How many molecules of acetyl CoA are formed from its complete catabolism? How can you calculate the number of molecules (moles) of ATP formed (net) by the complete catabolism of each fatty acid? and the number of moles of ATP formed per gram of each fatty acid metabolized??. Describe the β-oxidation of the fatty acid palmitateAccount for the total no. of ATPs produced from β-oxidation for one mole of the given fatty acid (16C). How many acetyl-coA are formed?

