OH i ACP Suppose that a "snapshot" of FA synthase was taken and the above (drawn) intermediate (STRUCTURE X) is shown in the active site of FASI. What chemical equations (below) best explains what would happen in the NEXT 2 reactions to create C8:0-ACP? Assume that FA synthesis occurs in the proper sequential order. O Structure X + NADPH + H+ --> NADP+ + C6:0-ACP + H₂O O Structure X + NADPH + H+ --> NADP+ + C6:0-ACP Structure X + 2 NADPH + 2 H+ --> 2 NADP+ + C6:0-ACP Structure X + 2 NADPH + 2 H+ --> 2 NADP+ + C6:0-ACP + 1 CO₂ O Structure X + 2 NADPH + 2 H+ --> 2 NADP+ + C6:0-ACP + 1 CoA + H₂O
OH i ACP Suppose that a "snapshot" of FA synthase was taken and the above (drawn) intermediate (STRUCTURE X) is shown in the active site of FASI. What chemical equations (below) best explains what would happen in the NEXT 2 reactions to create C8:0-ACP? Assume that FA synthesis occurs in the proper sequential order. O Structure X + NADPH + H+ --> NADP+ + C6:0-ACP + H₂O O Structure X + NADPH + H+ --> NADP+ + C6:0-ACP Structure X + 2 NADPH + 2 H+ --> 2 NADP+ + C6:0-ACP Structure X + 2 NADPH + 2 H+ --> 2 NADP+ + C6:0-ACP + 1 CO₂ O Structure X + 2 NADPH + 2 H+ --> 2 NADP+ + C6:0-ACP + 1 CoA + H₂O
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter21: Photosynthesis
Section: Chapter Questions
Problem 8P
Related questions
Question
Hw.3.
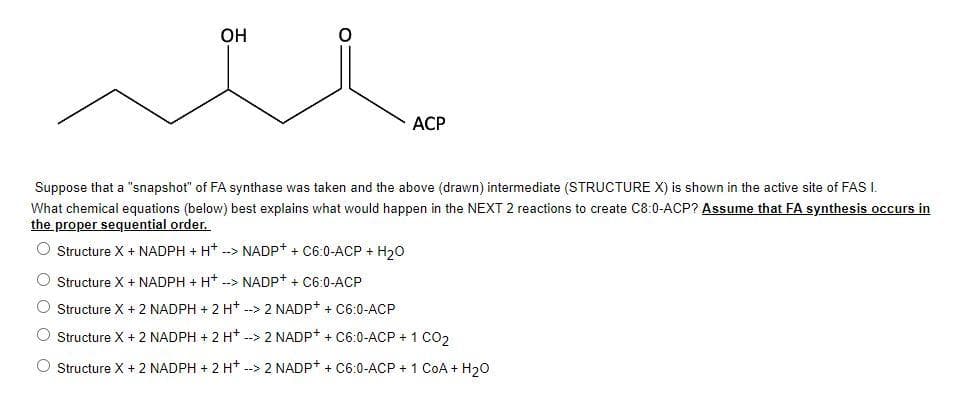
Transcribed Image Text:OH
ACP
Suppose that a "snapshot" of FA synthase was taken and the above (drawn) intermediate (STRUCTURE X) is shown in the active site of FAS I.
What chemical equations (below) best explains what would happen in the NEXT 2 reactions to create C8:0-ACP? Assume that FA synthesis occurs in
the proper sequential order.
O Structure X + NADPH + H+ --> NADP+ + C6:0-ACP + H₂O
O Structure X + NADPH + H+ --> NADP+ + C6:0-ACP
O Structure X + 2 NADPH + 2 H+ --> 2 NADP+ + C6:0-ACP
O Structure X + 2 NADPH + 2 H+ --> 2 NADP+ + C6:0-ACP + 1 CO2
O Structure X + 2 NADPH + 2 H+ --> 2 NADP+ + C6:0-ACP + 1 CoA + H₂O
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning