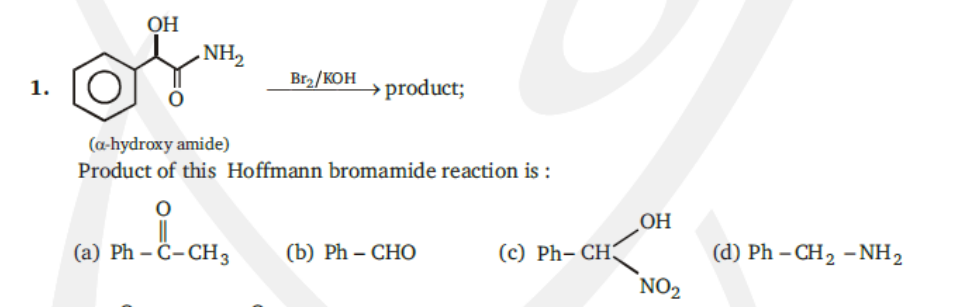
OH - NH, Br2/KOH →product; 1. (a-hydroxy amide) Product of this Hoffmann bromamide reaction is : OH (a) Ph – Ĉ-CH3 (Ъ) Ph — CHO (c) Ph- CH3 (d) Ph – CH2 -NH2 NO2
Q: clopropanation of allylic ethers as shown in the scheme below. Ph Et₂Zn + CICH₂l Toluene 3 h, -20°C…
A:
Q: (b) Suggest a plausible mechanism for the following transformation : Ph * LDA/Mel Ph Me Ph `Me + -78…
A:
Q: а) Consider the reaction scheme below. -[•] PhLi HONH,, HCI A ELOH B D (i) Deduce the structures of…
A: We have to give the structures of the compounds A, D, and the intermediates C. We also have to give…
Q: Heating compound X with aqueous formaldehyde forms Y (C17H23NO), which has been converted to a…
A:
Q: 2. Examine the syothetic scheme below and identify CO Reagenta und Conditions ()-() ( Structures A-E…
A: The key component associated with the molecule that provides the characteristic chemical reaction is…
Q: The scheme below shows the acid catalysed reaction of ethanoic acid with methanol. он H CH;--OH…
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only.…
Q: (c) Write the product(s) for the following reaction with supportive mechanism : ŞiMez KH →? Bu, Нех…
A: Here the given reaction is an example of Peterson Olefination. So, in this reaction alkene is…
Q: Q17. Compound 1 can undergo an intramolecular reaction to give cyclic product 2. Using curly arrows,…
A: Cyclic product is formed , which is cyclic amide , ester functional group act as electrophile and…
Q: Draw the organic product(s) of the following reaction. aqueous H,SO4 CH,0 NaCN Draw curved arrows to…
A:
Q: KMNO4, hot -CH2-CH2-C-H H*
A: In this question, we want to draw the major products. You can see details Solution below.
Q: Steroids are a class of organic compounds containing a specific tetracyclic skeleton (four rings),…
A: Given : We have to draw second step for the given reaction.
Q: ОН + H2O
A:
Q: C. Arrange the following compounds in order of decreasing reactivity towards hydrolysis. H2N, H3CO…
A: The reactivity of the acid derivatives towards the hydrolysis reaction is KAH>BCS>NAI>IYK
Q: Benzenediazonium carboxylate decomposes when heated to yield N2, CO2, and a reactive substance that…
A: The N2 and CO2 present in given diazonium carboxylate compound gets eliminated and results to form…
Q: cess CHCH-C 1 KMKO, NaOH HO AICI, H$O. 2. H0
A:
Q: C. Arrange the following compounds in order of decreasing reactivity towards hydrolysis. H2N, H;CO…
A: A multiple choice question based on hydrolysis, which is to be accomplished.
Q: 6B. Write a reaction scheme to selectively synthesize the following: OH OMe H3C OMe
A: 6B We have to write a reaction scheme selectively synthesise the following given molecules as…
Q: 2. (a) Explain the reactivity of dimethyl- sulphoxonium methylide and dimethyl- sulphoxium…
A:
Q: 3. When compound 11 is treated with two equivalents of methansulfonic acid (MSOH) in…
A: The reaction involves 2 molecules of 11 as reactant. The reaction mechanism is drawn below.
Q: Rank the reactivity of the following carboxylic acid derivatives towards hydrolysis. Assign '1' for…
A: The answer is given as follows
Q: The Claisen condensation of two molecules of ethyl phenylacetate (shown below) in the presence of…
A:
Q: Write the complete stepwise mechanism for the acid-catalyzed hydrolysis of the following amide to…
A:
Q: Oc-OH Which of the following can be hydrolyzed with acid to give benzoic acid, NO2 CN HN-NH2 OCCH,…
A:
Q: The following presented reaction scheme lead to the synthesis of which major product from benzene,…
A: The reaction of benzene with SO2 in presence of H2SO4 will give product benzene sulfonic acid. The…
Q: -NH2 N CI CI A B C Aniline Imine Aminal Stan and Kyle have observed a reaction between the aniline…
A:
Q: Suggest a plausible-mechanism-for the synthesis of the compound shown below H30 OH
A: This synthesis is bassed on Dienone-phenol rearrangement followed by reduction of side ring.
Q: Write the reaction CH3 CHz Ċ - OH to methyl propylamine 2.) CH3 CH2 C - CI CH3 NH CH3 3-) CH3 CH =…
A: GIVEN:-
Q: Me H2N NH2 acetaldehyde Chemical 1 methanamine Chemical 2 S-methyl ethanethioate Chemical 3…
A: The nitrogen atom of amine has lone pair of electrons therefore it can act as a nucleophile. The…
Q: Q3\ The pKa of p-cyclopropylbenzoic acid is 4.45. Is cyclopropylbenzene likely to be more reactive…
A:
Q: The following group are ortho-para director.Draw a contributing structure for the…
A: If the electrophile is attack at ortho position, the formed carbocation is stabilized by donating of…
Q: 1. H2SO4 Br +] H20 HO, NaBr 2. H3C-O H,C-O-Li Li f= Sn1 Nucleophilic substitution g= SN2…
A: The reactions given are,
Q: (e) DMP, CH2CI2 РСС, СН2CI2 Pyridinium chlorochromate in Aco OAc -OAc methylene chloride solvent OAc…
A:
Q: Q6 Butryic Acid + Trimethylamine The following equation describes the reaction of butyric acid with…
A:
Q: (i) Compare the biological activities of morphine and apomorphine (structures shown below). (ii)…
A: Biological activity of morphine and apomorphine- Morphine is an opioid agonist used for the relief…
Q: The following group are ortho-para director.Draw a contributing structure for the…
A:
Q: Acetonitrile (CH3C‚N) is deprotonated by a very strong base. Write resonance forms to show the…
A:
Q: Explain this
A: Nitration indicates the adding of -NO2 group. Mononitration of a benzene ring results in the…
Q: When diethyl succinate (1) is treated with NaOEt in EtOH, the major product is diethyl…
A:
Q: Give the major organic products for the following reaction: a) 1. CH23CH—с- A В Trace H* 2. HCI, H2O…
A: The given reactions can be completed as follow:
Q: b) Listed below are several hypothetical nucleophilic substitution reactions. None is synthetically…
A:
Q: 4. Tryptophan can also form adducts with arenediazonium ions, although the reaction is not…
A: Here the chemical reaction is:
Q: C. Arrange the following compounds in order of decreasing reactivity towards hydrolysis. H2N. H,CO…
A: In this question, we will arranged all compounds in decreasing order of their reactivity. You can…
Q: The following group are ortho-para director.Draw a contributing structure for the…
A: When an electrophilic aromatic substitution takes place, carbocation is formed on the aromatic ring…
Q: What is the major organic product to the following reaction with LIAIH, followed b acid hydrolysis?…
A:
Q: Q1) suggest a suitable mechanisms for the following organic Ton a) penta-1-cne H₂O/* sec-pentyl…
A:
Q: Following are two possible retrosynthetic analyses for the anticholinergic drug cycri- mine. Fill in…
A: Reaction of Piperidine with epoxide ring initiates the ring opening of epoxide and the product on…
Q: Benzamide, CaHsCONH2 1. is an amino acid 2. can be converted to phenylamine (aniline) on warming…
A: Amino acids: Amino acids are monomers of a protein. Amino acids through peptide bonds protein…
Q: Triethanolamine, (HOCH2CH2)3N, is a widely used biological buffer, with maximum buffering capacity…
A: The synthesis of triethanolamine, from ethylene oxide and ammonia has to be given.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Enamines formed from the cyclic secondary amine pyrrolidine are important intermediates in the synthesis of 1,5-diketones. (1st pic) On the structures provided below, draw arrows showing electron flow for the reaction mechanism for the acetic acid-catalyzed formation of an enamine from cyclohexanone and pyrrolidine. (2nd pic)Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Propose structural formulas for compounds (2) and (3).Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Name the types of functional groups in estrone
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.How many chiral centers are present in estrone?Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Show how your proposals for compounds (2) and (3) can be converted to compound (1). (Note: In the course of developing this synthesis, Tietze discovered that vinylic bromides and iodides are more reactive in Heck reactions than are aryl bromides and iodides.)Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q. In the course of the double Heck reactions, two new chiral centers are created. Assume in compound (3), the precursor to rings C and D of estrone, that the fusion of rings C and D is trans and that the angular methyl group is above the plane of the ring. Given this stereochemistry, predict the stereochemistry of…
- Electrostatic potential maps of anisole and thioanisole are shown. Which do you think is the stronger acid, p-methoxybenzoic acid or p-(methylthio)benzoic acid? Explain.Define the Mechanism - Conversion of Acid Chlorides to CarboxylicAcids ?Benzoic acid, Ph-COOH (C6H5CO2H), is not soluble in water while it dissolves in ether (diethyl ether), (CH3CH2)2O. Yet upon treatment with sodium hydroxide, benzoic acid turns hydrophilic and dissolves in water. Provide chemical explanation of this observation.
- Explain why a much better yield of primary amine is obtained from the reaction of an alkyl halide with azide ion (-N3), followed by catalytic hydrogenation. (Hint: An alkyl azide is not nucleophilic.)Synthesize the following compound from benzonitrile (C6H5CN):? Please don't provide hand written solution....Rank each of the following sets of nitrogen bases in terms of basicity and explain your answer



