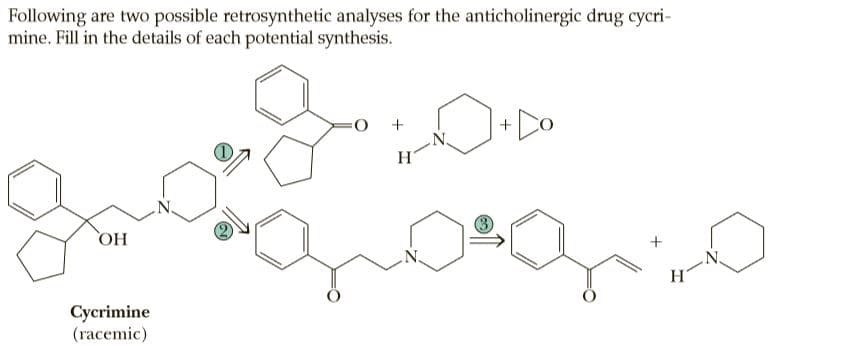
Following are two possible retrosynthetic analyses for the anticholinergic drug cycri- mine. Fill in the details of each potential synthesis. O: OH + H. Cycrimine (гасemic)
Q: Q2. Complete the following base hydrolysis reaction of ester. (1) NaOH (2) (H3O*) Q3. Write down the…
A:
Q: 2. Phenobarbital NH C2H5 Synthesis: LOEt EtO - Co OEt OEt NaOEt OEt OEt Eto H3C `Br NaOEt H2N NH2…
A:
Q: Following are the final steps in one industrial synthesis of vitamin A acetate. H,SO, ÓH (1) (2) (3)…
A: (a)
Q: 11. The amine below can be prepared from the ketone shown and what reagents? i ? (A) 1. NaCN 2.…
A: Ketons are organic compounds that contain only carbonyl group.
Q: Propose a mechanism for formation of 2-carbethoxy-4-butanolactone and 4-butanolactone…
A: 2-carbethoxy-4-butanolactone
Q: Which carboxylic compound of each pair would you expect to be stronger acid? Indicate (a) or (b) on…
A: In case 1) Me3N+ is an electron withdrawing group. It with draws electron from O-H bond through…
Q: Which of the following is a reasonable retrosynthetic approach to the target compound? он MgBr OH Br…
A: Free alcohol may hamper a Grignard reaction. So during retrosynthesis we have to very cautious…
Q: Draw the structure(s) of the major organic product(s) obtained after workup of the following…
A: The natural product is nicotine which is found in tobacco.
Q: Which nitrogen atom in each compound is more basic?
A:
Q: In the Gabriel synthesis of primary amines, N-potassiophthalimide is used as a source of the…
A: This is a Gabriel phthalamide synthesis reaction. Through this reaction 1° amine is produced by the…
Q: Which of the following bases will convert acetophenone to its conjugate base with greater than 99%…
A:
Q: Given this retrosynthetic analysis, propose a synthesis for the antidepressant moclobemide. CI H CI…
A:
Q: gas, Cl¬CH2CH2¬S¬CH2CH2¬Cl, was used as a poisonous chemical agentin World War I. Mustard gas is…
A: Mustered is highly alkylating agent due to the presence of two chlorine atom this chlorine and this…
Q: 1 The reaction of an a-diketone with concentrated sodium or potassium hydroxide to give the salt of…
A:
Q: Cancer of the prostate is the second leading cause of cancer deaths among American males, exceeded…
A:
Q: Following is a retrosynthetic analysis for the anthelmintic (against worms) diethylcarbamazine. N.…
A: Diethylcarbamazine is an organic compound used to treat Filariasis. It is formed y treating…
Q: Propose a synthesis for compound 1 (in the box I show how the molecule should be disconnected as a…
A: Multi step synthesis:
Q: Propose a synthetic route for the following conversion: N. toluene benzyl (methyl) amine
A: Stepwise reaction answered below.
Q: B1) Suggest a brief retrosynthetic analysis for 4-7 until you arrive at the starting material shown…
A: Since you have asked multiple questions, we will answer the first one for you. To get the remaining…
Q: 5. The acyl group is a protecting group for amines which can be deprotected by treatment with sodium…
A:
Q: How many signals would the product of the following reaction show in a. its 1H NMR spectrum?…
A: The expected product for the given reaction is shown below.
Q: 1. НаNNH2, mild acid 2. КОН, Н20, heat 1. LDA 2. 3. Н,О, н* HCN KCN
A: Organic synthesis involves the process of preparation, isolation and characterization of organic…
Q: N,N-Diethyl-m-toluamide (DEET) is the active ingredient in several common insect repellents. Propose…
A: Introduction: DEET is N,N-Diethyl-m-toluamide. It is commonly used in insect repellent. It can be…
Q: NH2 HO Atenolol Tenormin® Below is a two-step reaction sequence. What are the steps in the…
A:
Q: Propose a synthesis for the antihistamine histapyrrodine. NH2 ÇOOH Do -NH Histapyrrodine Pyrrolidine…
A: Body shows allergic reactions when some harmful foreign substance enters into it. These allergic…
Q: 11.6. The following pair of compounds produces different products depending on the base and order of…
A: Given are organic reactions.
Q: which of the following compounds would be the most basic? A. p-methoxyaniline B. p-nitroaniline C.…
A: Basicity means donate the lone pair of electron Basicity depends on 1) + I and + R
Q: 2-Propylpentanoic acid (valproic acid) is an effective drug for treatment of several types of…
A: Given: 2-Propylpentanoic acid (valproic acid) is an effective drug for treatment of several types of…
Q: 4. Here's a figure-it-out mechanism, resulting in the formation of p-aminophenol from an aromatic…
A: Introduction: The formation of para-aminophenol takes place through N-phenyl hydroxylamine. The…
Q: VII. Chemical Reactions and Mechanisms: Use the space below for reactions and mechanisms.| NH, он о…
A: Note : 1-Phenylethanamine reacts with tartaric acid gives diastereomeric salts. These diastereomeric…
Q: 1) How would you prepare phenylacetic acid (PHCH2CO2H) from benzyl bromide (PHCH2B1)?
A: Conversion of one compound to another desired compound is to be done by using specific reagent or…
Q: Which of the following compounds is the most basic? A. p-nitroaniline B. p-chloroaniline C.…
A: Basicity depends on the stability of conjugate acid forms after accepting the proton.
Q: Rank the attached three carboxylic acids in order of increasing acidity ?
A: Acidic behavior of compounds is explained on their tendency to give H+ ions. In organic molecules,…
Q: Given this retrosynthetic analysis, propose a synthesis for the local anesthetic hexylcaine. N. + HO…
A:
Q: 4. Show how you might carry out the following transformations using any needed inorganic reagents.…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: OH CH₂CNHCH3 CH3 C d OH CHCHNHCH₂ CH3 Ephedrine can be synthesized via reductive amination plus…
A:
Q: 3 Treatment of 1-aminoadamantane, C„H„N, with methyl 2,4-dibromobutanoate in the presence of a…
A: In the given SN2 reaction, 1-aminoadamantane act as a nucleophile and it make a bond with terminal…
Q: Propose a mechanism for the conversion of F to toremifene
A:
Q: Phenacetin is an analgesic compound having molecular formula C10H13NO2. Once a common component in…
A: Molecular formula of the compound is C10H13NO2 Since the compound contains Nitrogen atom in the…
Q: Rank the following compounds in order of increasing basicity: I. p-nitroaniline…
A: The basicity of a compound is the tendency of the atom to donate its lone pair. More the electron…
Q: Rank the following compounds in order of increasing basicity: 1.p-nitroaniline I p-aminobenzaldehyde…
A:
Q: Propose a synthesis of the topical anesthetic cyclomethycaine from 4-hydroxybenzoic acid,…
A:
Q: On the basis of retrosynthetic analysis, identify starting materials and synthons for the following…
A: The retrosynthesis is generally a process of disconnecting the target molecules into synthetic…
Q: In the mid-1930s a substance was isolated from a fungus that is a parasite of ryes and other…
A: Given that, lysergic acid diethylamide (LCD, C20H25N3O). It is a weak base, having Kb = 7.6×10-7.…
Q: I. Propose an efficient synthesis for the following transformation. OH -Ph Ph
A: Reaction
Q: In the Gabriel synthesis of primary amines, N-potassiophthalimide is used as a source of the…
A: Here we have to write products formed in step-1 and step-2 in the following given Gabriel synthesis.…
Q: Following is a retrosynthetic analysis for the synthesis of the herbicide (S)-Metolachlor from…
A: The retrosynthetic method is used to predict the starting material from product structure by…
Q: Following are two possible retrosynthetic analyses for the anticholinergic drug cycrimine. N govor…
A: Retro-synthetic analysis is process by which one can get the possibility synthons / synthetic…
Q: 1. Predict the products or the reaction of (i) phenylacetaldehyde and (ii) acetophenone with the…
A: Organic reactions are those in which organic reactant react to form organic products.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 4 images

- Treatment of 1-aminoadamantane, C10H17N, with methyl 2,4-dibromobutanoate in the presence of a nonnucleophilic base, R3N, involves two successive SN2 reactions and gives compound A. Propose a structural formula for compound A.Following is an outline of a synthesis of the bronchodilator carbuterol, a beta-2 adrenergic blocker with high selectivity for airway smooth muscle receptors. Q.Why is it necessary to add the benzyl group, PhCH2—, as a blocking group in Step 1?Which of the following reactions represents the most likely reaction of naproxen with water? a.) C14H14O3(aq) + H2O(l) ⇄C14H13O3-(aq) + OH-(aq) b.) C14H14O3(aq) + H2O(l) ⇄C14H15O3+(aq) + OH-(aq) c.) C14H14O3(aq) + H2O(l) ⇄ C14H13O3-(aq) + H3O+(aq) d.) C14H14O3(aq) + H2O(l) ⇄C14H15O3+(aq) + H3O+(aq)
- When an α-hydroxy amide is treated with Br2 in aqueous NaOH under Hofmann rearrangement conditions, loss of CO2 occurs and a chain-shortened aldehyde is formed. The mechanism involves the following steps: Base abstracts an acidic amide proton, yielding amide anion 1; The amide anion reacts with bromine in an α-substitution reaction to give N-bromoamide 2. Abstraction of the remaining amide proton by base gives a resonance-stabilized bromoamide anion 3; Rearrangement occurs to yield isocyanate 4; Water adds to the isocyanate to yield carbamic acid 5; Elimination of CO2 yields carbinolamine 6; Following proton transfer, expulsion of ammonia yields the final product aldehyde. Write out the mechanism and then draw the structures of bromoamide anion 3 and amide anion 1.gas, Cl¬CH2CH2¬S¬CH2CH2¬Cl, was used as a poisonous chemical agentin World War I. Mustard gas is much more toxic than a typical primary alkyl chloride. Itstoxicity stems from its ability to alkylate amino groups on important metabolic enzymes,rendering the enzymes inactive.(a) Propose a mechanism to explain why mustard gas is an exceptionally potent alkylatingagent.(b) Bleach (sodium hypochlorite, NaOCl, a strong oxidizing agent) neutralizes and inactivates mustard gas. Bleach is also effective on organic stains because it oxidizes coloredcompounds to colorless compounds. Propose products that might be formed by thereaction of mustard gas with bleach.Mustard gas, Cl¬CH2CH2¬S¬CH2CH2¬Cl, was used as a poisonous chemical agentin World War I. Mustard gas is much more toxic than a typical primary alkyl chloride. Itstoxicity stems from its ability to alkylate amino groups on important metabolic enzymes,rendering the enzymes inactive.(a) Propose a mechanism to explain why mustard gas is an exceptionally potent alkylatingagent
- One step in the urea cycle for ridding the body of ammonia is the conversion of argininosuccinate to the amino acid arginine plus fumarate. Propose a mechanism for the reaction, and show the structure of arginine.Following is a synthesis for toremifene, a nonsteroidal estrogen antagonist whose structure is closely related to that of tamoxifen. (a) This synthesis makes use of two blocking groups, the benzyl (Bn) group and the tetrahydropyranyl (THP) group. Draw a structural formula of each group and describe the experimental conditions under which it is attached and removed. (b) Discuss the chemical logic behind the use of each blocking group in this synthesis. (c) Propose a mechanism for the conversion of D to E. (d) Propose a mechanism for the conversion of F to toremifene. (e) Is toremifene chiral? If so, which of the possible stereoisomers are formed in this synthesis?



