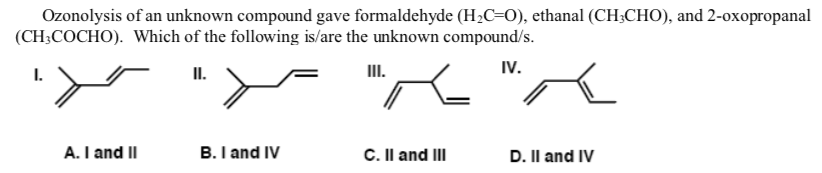
Ozonolysis of an unknown compound gave formaldehyde (H;C=0), ethanal (CH3CHO), and 2-oxopropanal (CH;COCHO). Which of the following is/are the unknown compound/s. IV. - "K "A I. II. II. A. I and II B. I and IV C. Il and III D. Il and IV
Q: tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid…
A: The answer is as follows:
Q: Compound Mol. Wt (g/mol) Density (g/mL) b.p. (°C) m.p. (°C) 2-Methyl-2-propanol 74.12 0.786…
A:
Q: 1. Ferric Chloride Test Add 5 drops of FeCl3 solution to 4 different test tubes containing 1 mL each…
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: In the acylation of ferrocene experiment, some diacetylferrocene was formed as a biproduct. Where…
A: This is Friedel craft acetylation reaction. The second acyl group will add on 4 position. Second…
Q: Select the reagent(s) expected to accomplish the reaction shown below. CH2CH3 C=c H3CH2C…
A: Choose the suitable reagent of the following reaction ---
Q: Provide a plausible arrow pushing mechanism for the reaction below. O-OtBu s-OtBu AHN. CO,Me 1. LDA…
A:
Q: Which of the following compounds will give a positive visible result with the Belstein test? Rich…
A: Positive Beilstein test is given by halides (except fluoride). When Cu wire is heated then a coating…
Q: event is not an oxidation reaction? A. Product formation as a result of the reaction of benzoine…
A: Answer The event is not an oxidation reaction All events are…
Q: 1. HNO3, H,SO, 2. Fe, HCI 3. NANO,, HCI 4. HBF, 5. KMN04, A CH(CHS2
A:
Q: NaH ? LOH HO PBr3 D в A (d) Propose the structure of the organic compound D formed by reaction with…
A:
Q: CF2Cl2 can be prepared from the nucleophilic substitution of another alkyl halide CCl4 in the…
A: Given-> CCl4 + 2HF ------> CF2Cl2 + 2HCl. Rate = K[CCl4] Rate constant (K) = 1.5 × 10-4 s-1…
Q: 4) Benzene, CH, can be described as an aromatic substance and is a starting material for many…
A:
Q: provide appropriate product 9. Br 1. Pd Ln, EtsN, MECN, Me Me 2. MCPBA, CH2Cl2 3. НСЮ, ЕОН
A:
Q: Organotin compounds play a significant role in diverse industrial applications. They have been used…
A:
Q: Consider the following reaction: КОН + KBr Br OH a) Does the reaction proceed via an SN1 or SN2…
A:
Q: Rank and explain your ranking of the following chlorides of group 15 in the order of increasing iv)…
A: Ionic compounds have positively charged cation and negatively charged anion due to which a dipole…
Q: For the successful synthesis of triphenylmethanol , 0.05 moles of methyl benzoate would require…
A:
Q: Illustrate the Electrophilic Addition of H2O to an Alkene—Hydration ?
A: The electrophilic addition reaction is a type of organic reaction in which the electrophile attacks…
Q: The diagrams 1 to 3 below can be arranged to show the mechanism involved in a familiar reaction. The…
A:
Q: For question below the separation scheme, please indicate the compound that best fits in the blank…
A: When two soluble salts are present in an aqueous solution, their ions can be displaced by one…
Q: N' 'N' CH3 CH3 II
A: I is more favorable enamine
Q: II (a) I= a, B-diketone, II = a, ß-diester, III = B-keto nitrile (b) I = B-diketone, II = B-diester,…
A: In this question, we will Identify the class of all three Compounds. You can see details Solution…
Q: Why are solutions of KMnO4 and Na2S2O3 generally stored in dark reagent bottles?
A: The reason why solutions of KMnO4 and Na2S2O3 generally stored in dark reagent bottles have to be…
Q: Provide a plausible arrow pushing mechanism for the reaction below. 1. ABuLi ŅME2 2. MgCl HO,C. 3.…
A:
Q: A chemist wanted to synthesize the anesthetic 2-ethoxy-2-methylpropane. He used ethoxide ion and…
A:
Q: Pentachlorophosphine is used in Beckmann Rearrangement. 4. 5. Metachloro-peroxybenzoic acid is used…
A: Beckmann rearrangement reaction. As per our guidelines we have to answer first question only.
Q: Question 3
A: Organic compounds that contain a carbon atom directly bonded with metals such as magnesium, lithium…
Q: a. Write two reaction paths for the production of monochlorodecane (C10H21CI) by reaction of Decane…
A: In the presence of sunlight, a radical substitution reaction takes place and monochlorodecane is…
Q: explain some of the hazards (name at least three) involved in the Friedel-Crafts Acylation:…
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you.…
Q: 3. Determine the amount, in of a (2.20 M s olution of dichloromethane needed to completely react…
A: Given: Bromine(Br2) reacts with Cyclohexene to give 1,2-dibromocyclohexane.
Q: nost" und nder the one acetaldehyd e "most" under the one which t" under the one which would be le…
A:
Q: The conversion of 1-butanol to the given compound can be performed with some combination of the…
A:
Q: tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid…
A:
Q: (a) (b) (b) The following reactions as written will NOT give the indicated product. For each of the…
A: a. (i) and (iii)Reaction does not give the given product if the given reagents are used as per…
Q: The most common reducing agent used for the conversion of ester into aldehyde. * a-H2/Ni, Heat…
A: esters are converted into aldehyde by DIBAL-H Option"d" is correct.
Q: 1.The reduction of butanoic acid can form butanal and butan-1-ol. The boiling points of these…
A: Reduction of carboxylic acid produces alcohol and aldehyde. C=O converts to aldehyde and and when…
Q: In the lab for the synthesis of methyl m-nitrobenzoate from methyl benzoate using HNO3 and H2SO4…
A: Formation of m-nitrobenzoate can be done from the reaction of methyl benzoate with nitric acid and…
Q: 4- Adding HCI in the preparation of aniline from nitrobenzene. 5- When acetanilide reacts with…
A: A question based on amines that is to be accomplished.
Q: 4. For the Sn2 reaction below: Give the mechanism with curved arrows and products for the reaction…
A: Given:
Q: ation assignment.pdf 1/ 2 308% ОН KMNO4 H3C- -CH3 H,SO4, H2O C. 4 O v i 1:17 ТАС
A:
Q: A greener alternative to bromination with elemental bromine is the reaction of the acetanilide with…
A: Ammonium ceric nitrate is an inorganic compound. It's molecular formula is (NH4)2Ce(NO3)6. It is…
Q: write the reaction of the action of methyl-2, bromo-2-propane with hot KOH solution. a) Name the…
A:
Q: NaH ? HO LOH PBR3 D в A (d) Propose the structure of the organic compound D formed by reaction with…
A: This reaction proceeds via SN2 mechanism. OH is converted to leaving group which is displaced by…
Q: tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid…
A:
Q: Functional groups such as alkynes react the same in complex molecules as they do in simpler…
A:
Q: Why rearrangements are sometimes beneficial in designing synthetic routes, 3 reasons
A: As per the bartleby guidelines, an expert is allowed to answer first one question at a time. Please…
Q: 1.) Why are functional group interconversions controversial? (double bonds into epoxide) 2.) Explain…
A: A double bond (alkene) converts into an epoxides as : Functional group modification or…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- For butenafine, develop a synthesis that starts with napthalene and benzene. (aromatic methyl groups can be converted to alkyl bromides using Br2/light, and aldehydes by heating the alkyl bromide in DMSO, eg. ArCH3 to ArCH2Br then to ArCHO).Describe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…E. TESTS FOR PHENOLIC COMPOUNDSPhenolic compounds such as phenol, salicylic acid, etc., give characteristiccolors with FeCl3 and Millon’s Reagent.1. Ferric Chloride TestAdd 5 drops of FeCl3 solution to 4 different test tubes containing 1 mL each ofdilute solutions of the following compounds and note the color obtained.a. Phenol _______________________________________________________________b. Salicyclic acid ___________________________________________________________c. Resorcinol _______________________________________________________________d. Picric acid _______________________________________________________________2. Millon’s TestPrepare another set of 4 test tubes containing 1 mL each of the solutions listedbelow. Add 3 drops of Millon’s reagent to each and place all the test tubes in aboiling water bath. Note the color formed.a. Phenol _______________________________________________________________b. Salicylic acid ___________________________________________________________c.…
- In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…
- A mixture of 0.10 mol benzene and 0.10 mol p-xylene was allowed to react with 0.10 mol nitronium ion until all the nitronium ion was gone. Two products were obtained: 0.002 mol of one and 0.098 mol of the other. a. What was the major product? b. Why was more of one product obtained than of the other?What is reaction 1 and 2 called? Choices: A. Williamson ether synthesis, B. Reduction with Grignard reagent, C. Acidic ether cleavage, D. FC Alkylation What are the reagents for reaction 1 and 2? Choices: A. methanol in acidic medium, B. sodium hydride and bromomethane, C. methyl bromide and aluminum bromide, D. methanoyl bromideFill in the blanks with appropriate compounds via Wittig reaction.
- Acid Halide Preparation reaction mechanism: Please use HCl and SOCl2 as reagentsOrganotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4AI(C2H5)3 →3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d = 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3): d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257 L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Which or which of the statements given below is correct. I) Maleic anhydride is a carboxylic acid derivative and its reaction with water is a reduction reaction. II) Fumaric acid and maleic acid are stereoisomers of each other III) Since fumaric acid has a more stable structure than maleic acid, its boiling point is higher. A. Solo I B. I and III C. II and III D. I, II, III E. Solo III

