Part A How many grams of glucose are required to form 115 g of ethano? Express your answer with the appropriate units Value Submit Part B "A Submit Value Request Answer PA 0 Units How many grams of ethanol would be formed from the reaction of 0.280 kg of glucose? Express your answer with the appropriate unit Request An 0 Units ? ? Review
Part A How many grams of glucose are required to form 115 g of ethano? Express your answer with the appropriate units Value Submit Part B "A Submit Value Request Answer PA 0 Units How many grams of ethanol would be formed from the reaction of 0.280 kg of glucose? Express your answer with the appropriate unit Request An 0 Units ? ? Review
Chapter14: Chromatography
Section: Chapter Questions
Problem 9P
Related questions
Question
Give both solution just with small explanation
Asap
Thanks
Transcribed Image Text:‹6
A
R
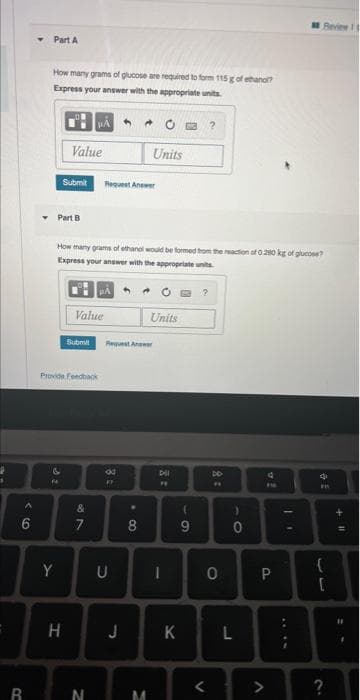
Part A
How many grams of glucose are required to form 115 g of ethano?
Express your answer with the appropriate units
Y
Submit
Part B
F4
Value
H
Provide Feedback
Value
Submit
How many grams of ethanol would be formed from the reaction of 0.280 kg of glucose?
Express your answer with the appropriate units.
μA
87
&
7
N
Request Answer
4 ★
8 =
FF
Request Answer
U
Units
.
8
Units
M
Dil
FF
JK
(
9
?
DO
0
<
)
0
L
v!
V
4
P
Review 1
LE IN
E
{
?
+11
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

