Q: Answer the following questions. What are the reasons for determining the ash content of a drug?…
A:
Q: Table 6. Baeyer,s Test (cold, dilute, neutral KMNO4) Description reagent: The reagent has a purple…
A: Baeyer's reagent is alkaline KMnO4. It is a mild oxidizing agent and is used to detect carbon-carbon…
Q: Shown below is the structure of one of the methyl ester biodiesel products together with its 13c NMR…
A: We have to determine the 13C NMR peaks of A, B, C, and D.
Q: 1. Aromatic compounds are metabolized via transformation of the aromatic ring to a reactive…
A: Arene oxides are getting metabolized via: Rearrangement to produce phenols Hydration GSH…
Q: Explain the following statement. Although 2-methoxyacetic acid (CH3OCH2COOH) is a stronger acid than…
A:
Q: Pease indicate the compound that best fits in the blank of the separation scheme below. OH HO. H2N'…
A:
Q: Draw the structure of sodium tetradecyl sulfate and ciprofloxacin and state how the activity of the…
A: The key components associated with a molecule that is responsible for providing the characteristic…
Q: Which of the following reagents can be used in the reaction in the photo? A.) pyridinium…
A: Given, the following conversion as follows,CH3CH2CH2OH → CH3CH2COOHwe are asked which reagent is…
Q: the pKa values of acetone and 1-phenyle-2-propanone are 26.5 amd 19.8 respectively. use electronic…
A: Since in 1-phenyle-2-propanone we have a phenyle group which is having -ve inductive effect on the…
Q: Why is it easier to see all three pKa values in a tripolic acid but not all 3 equivalence points?
A:
Q: Propranolol binds to beta-adrenergic receptors with a Kj of 8.9 x 10-9M. What concentration of…
A:
Q: 9-Aminofluorene has applications in the structural analysis of proteins and carbohydrates. Write a…
A: The given compounds are 9-aminofluorene and fluorene. In this question, a mixture of 9-aminofluorene…
Q: Pease indicate the compound that best fits in the blank of the separation scheme below. 0000 OH OH…
A: The organic compounds present in the mixture are benzocaine, phenol, biphenyl and benzoic acid.
Q: Assume that 2 mL of aniline and 3 mL of acetic anhydride are used in the preparation of acetanilide.…
A:
Q: 3. When butanoic acid (7.0 mL) is dissolved in methanol (20 mL) and heated with a catalytic amount…
A: Volume of Butanoic acid = 7.0 mL Volume of methanol = 20.0 mL Volume of methyl butanoate = 6.5 mL
Q: 12. Colour of cold CoCl2 (aq): 13. Colour of hot [CoCl4]- (aq: 14. Is this reaction exothermic or…
A: 1
Q: MCQ 159: Bromine (Brg) is relcased during reactions A. reddish brown gas B. blue gas C. white gas D.…
A: The question is based on physical properties of gases. We have were to identify the physical…
Q: Compound EE, CSH100 gives a positive result (formation of silver mirror) when reacted with Tollen's…
A:
Q: Sulfuric acid (H2SO4) is a strong acid in aqueous solution, yet the pKa of sulfuric acid is 7.6 in…
A:
Q: Which of the compounds below form water soluble salts (quantitatively) when added to a saturated…
A: In the given set of molecule only compound a will form slat with sodium bicarbonate as it contains…
Q: он OH OH он OCH3 CI CH3 10.0 10.2 9.2 7.7
A: Give an explanation, why the acidity of these phenolic derivatives give these pKa value?
Q: Calculate the percentage yield: Preparation of N-(4-hydroxy-phenyl )-proponiamide ; To a suspension…
A: Synthetic organic chemistry involves the conversion of reactants into specific products by using the…
Q: How do you prepare this solution? 10ml of 20% (w/v) L-arabinose stock (weight/volume). How much of…
A:
Q: Calculate the weight of salicylic acid (FW: 138.12) needed to generate 5.00 g of aspirin…
A: In order to calculate the weight of the salicylic acid we need to know the theoretical yield as…
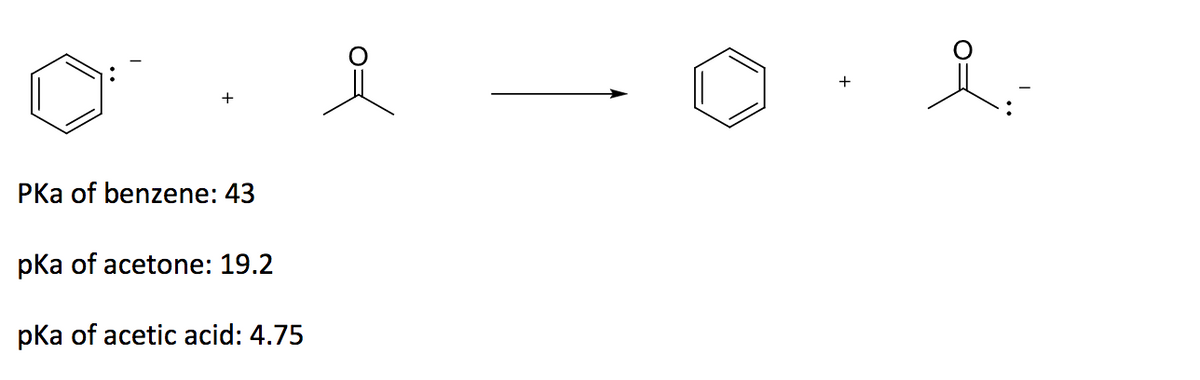
Q: Write an equation for the acid-base reaction between 2,4-pentanedione and sodium eth- oxide and…
A: An equation for the acid-base reaction of 2,4-Pentanedione and sodium ethoxide gives conjugate base…
Q: A student attempted to prepare 1-chlorobutane by mixing 1-butanol with NaCl in acetone. Was the…
A:
Q: Give the balanced equation of acetylene and the following reactions • Combustion • dilute KMnO4…
A: 1. Combustion of acetylene Combustion of acetylene produces CO2 and H2O…
Q: Which of the following sets of reagents would not be an acceptable method for the preparation of…
A:
Q: When 0.100 g of an unknown polyhydroxy organic substance was added to 50.0 mL of phenol, a decrease…
A: Given:- 0.100 g of an unknown polyhydroxy organic substance was added to 50.0 mL of phenol, A…
Q: Write the equilibrium-constant expressions and obtainnumerical values for each constant in. (a) the…
A:
Q: In acid-base equilibria, the presence of an electron withdrawing substituent on the R- group of the…
A: The nature of alcohols (R-OH) is acidic. After the removal of proton (deprotonation), alcohol forms…
Q: Calculate the number of moles of reactants and theoretical yield of each product in each reaction.…
A: Here structure-4 is the only final product. The structure and formula of both the reactants and…
Q: What is the suitable solvent to recrystallize azobenzene, p-nitroaniline and benzoic acid
A: Recrystallization is a technique used to purify solid compounds. During recrystallization, an impure…
Q: Show the electron-flow mechanism of amiline from benzoic acid, this involves predicting major and…
A:
Q: Here is a flowchart for the separation of a mixture of a primary aliphatic amine (RNH, pK, 10.8), a…
A: The given mixture contains amine, carboxylic acid and phenol. All these are soluble in diethyl ether…
Q: A 1. HNO, / H2SO, в 2. Br, / FeBr3 1. Fe/ HCI 2. NaNO, / HCI 3. CUCN 1. Large excess of LIAIH,…
A:
Q: What is the pKa of the imide proton of naphthalimide? To what do you attribute its acidity
A:
Q: Dimethyl disulfide, CH,S–SCH3, found in the vaginal secretions of female hamsters, acts as a sexual…
A:
Q: Propose a synthesis for esmolol from 4-hydroxycinnamic acid, epichlorohydrin, and isopropylamine
A: Reduction of 4-hydroxycinnamic acid using palladium produces a saturated acid. This reacts with…
Q: Would you expect 2-Chlorobutanoic acid to have higher or lower pka than 4-Chlorobutanoic acid?
A: Does 2-Chlorobutanoic acid to have higher or lower pka than 4-Chlorobutanoic acid?
Q: Barium chloride reagent is used to confirm the authenticity of a medicinal product: A) Bismuth…
A: Barium chloride is used to confirm sulfate ions.
Q: Reduction of ketones: How to calculate the theoretical yield in reduction of…
A: Theoretical yield calculation requires amount of reactant and product then using laboratory mathod…
Q: Synthesis of p-Bromoaniline A student starts with 1.15 g of acetanilide and isolates 1.55 g of the…
A:
Q: 4. Below is an acid-base extraction flow chart. The four compounds seen in the first box was mixed…
A: The above acid-base separation chart can be solved using some basic acid base concepts. these are-…
Q: Consider the nitration by electrophilic aromatic substitution of salicylamide to iodosalicylamide.…
A: Mass of salicylamide = Density of salicylamide × Volume of salicylamide Mass of salicylamide = 1.09…
Q: Show how you would use extractions with a separatory funnel to separate a mixture of the following…
A:
Q: Isoerythrogenic acid, C18H26O2, is an acetylic fatty acid that turns a vivid blue on exposure to UV…
A: Based on the data given above, the line structure is shown below- It is also known as isanic acid.
Q: Why are carboxylic acids more acidic than alcohols? The increased inductive effect of the alkyl…
A: Acidic strength of organic acids can be compared with their pKa value which is determined…
Q: Use the four compounds shown below to answer the following questions: a. Why are the…
A:
Consider the following reaction and decide whether acetic acid is a suitable solvent. Show your work by drawing the reaction of acetic acid with each reactant and determining the direction of equilibrium quantitatively.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- 1) Hydrolysis of Methyl Salicylate: Methyl Salicylate (C8H8O3) + sodium Hydroxide (NaOH) -> sodium Salicylate ( NaC7H5O3) + Methanol (CH3OH) 2) Acidification of Sodium Salicylate: Sodium salicylate (NaC7H5O3) + HCL -> Salicylic acid (C7H603) + sodium chloride (NaCL) Could you find the theoretical yield for me? 2.15g of methyl salicylate was used. 25ml of sodium hydroxide (2m solution) was used for hydrolysis and 2ml of hydrochloric acid was used for the acidification. This produced a yield of 1.53g of salicylic acid.Which 9.26 g of an unknown solid is dissolved in a coffee cup calorimeter that contains 62.0 mL of water. The measured temperature increased from 23.4 °C to 24.7 °C. The specific heat of water is 4.184Jg-1°C-1, and assume the density of the solution is 1.00 g mL-1. Calculate the molecular weight of the unknown solid if the ΔrH was determined to be -2.51kJmol-1 via another experiment.Calculate the percent yield and theoretical yield for the following reaction: oxidation of cyclododecanol to cyclododecanone. 0.2093 grams of Cyclododecanol 0.5 mL of acetone 0.20 mL of Glacial acetic acid 2.0 mL of bleach Final weight of cyclododecanone = 0.0578 g
- 1. Based from the given problem, What is the molecular weight of the tartaric acid (C4H6O6)? 2. Based from the given problem, What is the meq of the tartaric acid (C4H6O6)? 3. Based from the given problem, What is the percentage purity of the tartaric acid (C4H6O6)?Calculate the moles of the limiting reagent for the following reaction: oxidation of cyclododecanol to cyclododecanone. 0.2093 grams of Cyclododecanol 0.5 mL of acetone 0.20 mL of Glacial acetic acid 2.0 mL of bleach Final weight of cyclododecanone = 0.0578 gAcetanilide (C6H5NHCOCH3, M = 135.2) can beprepared from aniline (C6H5NH2, M = 93.1) and aceticanhydride ([CH3CO]2O, M = 102.1) according to thebalanced equation below. C6H5NH2 + (CH3CO)2O →C6H5NHCOCH3 + CH3COOH If 10.0 g acetanilide is isolated from the reaction of 10.0g aniline with 10.0 g acetic anhydride, what is the percentyield of the reaction?
- (a) benzene can potentially react with Lewis acid to form new carbon-carbon bond. Propose the starting material and stepwise mechanism to produce new chemical structure which consists of a formula molecule of C11H16.formation of tetraphenylnaphthalene Amount of tetraphenylcyclopentadienone: 0.530 g Volume of glyme: 3 mLVolume of isoamyl nitrite: 0.75 mL anthranlic acid: 0.253 g tetraphenylnaphthalene: 1.25 g theoretical yield: percent yield:Calculate the % yield of the following reaction. You have isolated 1.5g of benzil from the reaction of 1.95 of b enzoin and 1.0 g of ammonium nitrate A.75.5% B.77.6% C78.1 %
- The amount of tartaric acid is responsible for the tartness of wine and controls the acidity of the wine. Tartaric acid also plays a very significant role in the overall taste, feel and color of a wine. Tartaric acid is a diprotic organic acid The chemical formula for tartaric acid is C4H6O6 and its structural formula is HO2CCH(OH)CH(OH)CO2H. A 50.00 mL sample of a white dinner wine required 21.48 mL of 0.03776 M NaOH to achieve a faint pink color. Express the acidity of the wine in terms of grams of tartaric acid, H2C4H4O6 (M. M. = 150.10) per 100 mL of wine. Assume that the two acidic hydrogens are titrated at the end point. MM H2C4H4O6 = 150.10 MM NaOH = 40.00 Below is the balanced chemical equation for this titration.How many grams each of resorcinol and hexachloro-phene should be used in preparing 2 pounds of an acne ointment that is to contain 2% resorcinol and 0.25% of hexachlorophene?Methanogenic bacteria convert acetic acid (CH3COOH) to CO2(g) and CH4(g). ΔH°f, (kJ/mol) ΔG°f, (kJ/mol) CH3COOH -484.5 -389.9 CO2(g) -393.5 -394.4 CH4(g) -74.8 -50.8 Calculate ΔH°rxn. ΔH°rxn = ____ kJ Calculate ΔG°rxn. ΔG°rxn = ___kJ

