Please paraphrase how both experiments (from fig.2 and 3) are asking differing questions
Please paraphrase how both experiments (from fig.2 and 3) are asking differing questions
Basic Clinical Laboratory Techniques 6E
6th Edition
ISBN:9781133893943
Author:ESTRIDGE
Publisher:ESTRIDGE
Chapter6: Basic Clinical Chemistry
Section6.1: Introduction To Clinical Chemistry
Problem 9RQ
Related questions
Question
Please paraphrase how both experiments (from fig.2 and 3) are asking differing questions
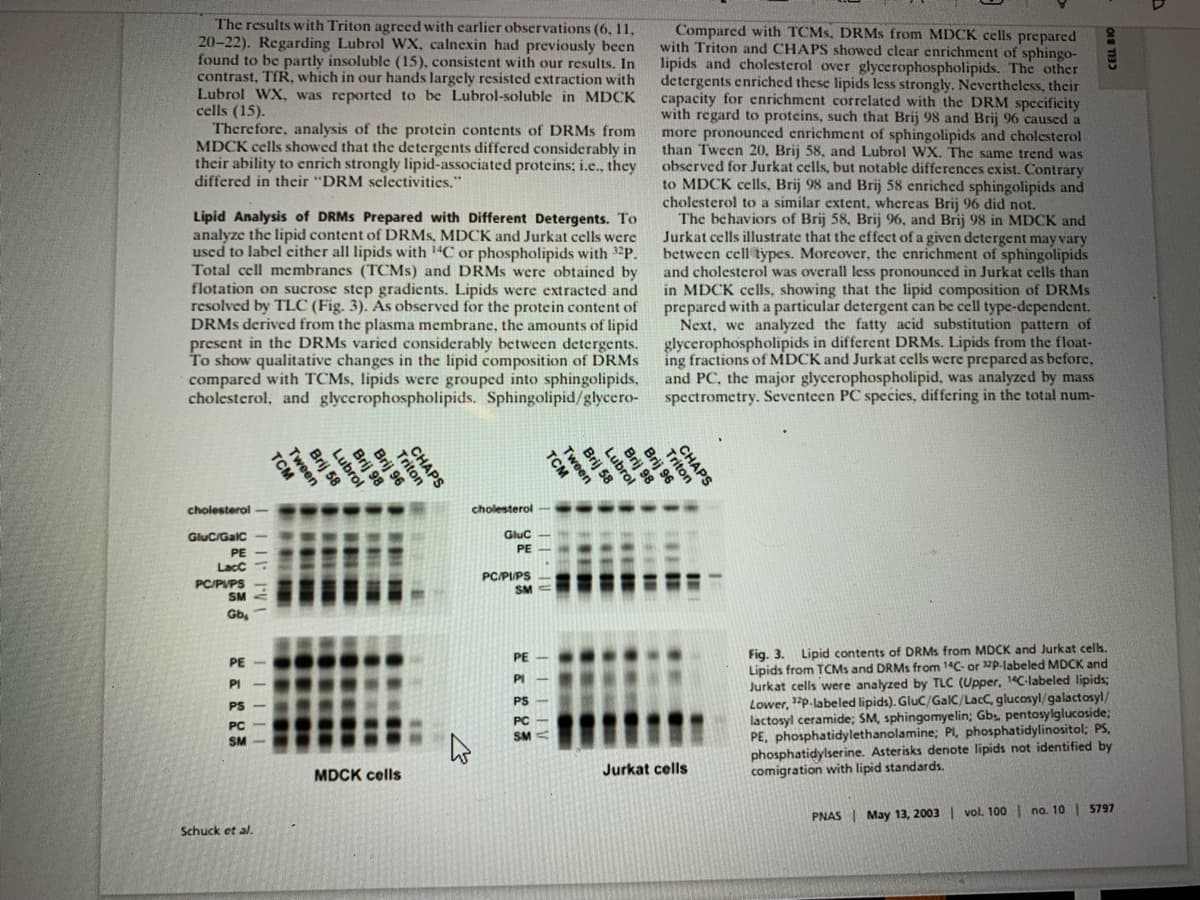
Transcribed Image Text:The results with Triton agreed with earlier observations (6, 11,
20-22). Regarding Lubrol WX, calnexin had previously been
found to be partly insoluble (15), consistent with our results. In
contrast, TfR, which in our hands largely resisted extraction with
Lubrol WX, was reported to be Lubrol-soluble in MDCK
cells (15).
Therefore, analysis of the protein contents of DRMS from
MDCK cells showed that the detergents differed considerably in
their ability to enrich strongly lipid-associated proteins; i.e., they
differed in thcir "DRM selectivities."
Lipid Analysis of DRMS Prepared with Different Detergents. To
analyze the lipid content of DRMS, MDCK and Jurkat cells were
used to label either all lipids with 1"C or phospholipids with 32P.
Total cell membranes (TCMS) and DRMS were obtaincd by
flotation on sucrose step gradients. Lipids were extracted and
resolved by TLC (Fig. 3). As observed for the protein content of
DRMS derived from the plasma membrane, the amounts of lipid
present in the DRMS varied considerably between detergents.
To show qualitative changes in the lipid composition of DRMS
compared with TCMS, lipids were grouped into sphingolipids,
cholesterol, and glycerophospholipids. Sphingolipid/glycero-
Compared with TCMS, DRMS from MDCK cells prepared
with Triton and CHAPS showed clear enrichment of sphingo-
lipids and cholesterol over glycerophospholipids. The other
detergents enriched these lipids less strongly. Nevertheless, their
capacity for enrichment correlated with the DRM specificity
with regard to proteins, such that Brij 98 and Brij 96 caused a
more pronounced enrichment of sphingolipids and cholesterol
than Tween 20, Brij 58, and Lubrol WX. The same trend was
observed for Jurkat cells, but notable differences exist. Contrary
to MDCK cells, Brij 98 and Brij 58 enriched sphingolipids and
cholesterol to a similar extent, whereas Brij 96 did not.
The behaviors of Brij 58, Brij 96, and Brij 98 in MDCK and
Jurkat cells illustrate that the effect of a given detergent may vary
between cell types. Morcover, the enrichment of sphingolipids
and cholesterol was overall less pronounced in Jurkat cells than
in MDCK cells, showing that the lipid composition of DRMS
prepared with a particular detergent can be cell type-dependent.
Next, we analyzed the fatty acid substitution pattern of
glycerophospholipids in different DRMS. Lipids from the float-
ing fractions of MDCK and Jurkat cells were prepared as before,
and PC, the major glycerophospholipid, was analyzed by mass
spectrometry. Seventeen PC species, differing in the total num-
cholesterol
cholesterol
Gluc/Galc
Gluc
PE
PE
LacC
PC/PVPS
SM
PC/PIPS
SM
Gb
Fig. 3. Lipid contents of DRMS from MDCK and Jurkat cells.
Lipids from TCMS and DRMS from 14C- or 12p.labeled MDCK and
Jurkat cells were analyzed by TLC (Upper, 1"C-labeled lipids;
12p-labeled lipids). Gluc/GalC/LacC, glucosyl/galactosyl/
PE
PE-
PI
PIㅡ
PS
Lower,
lactosyl ceramide; SM, sphingomyelin; Gbs pentosylglucoside;
PE, phosphatidylethanolamine; PI, phosphatidylinositol; PS,
phosphatidylserine. Asterisks denote lipids not identified by
comigration with lipid standards.
PS
PC
PC
SM<
SM
MDCK cells
Jurkat cells
PNAS I May 13, 2003 | vol. 100 | no. 10 | 5797
Schuck et al.
Brij 96
Brij 98
Lubrol
Brij 58
CHAPS
riton
Brij 96
Brij 98
Lubrol
Brij 58
Tween
Tween
TCM

Transcribed Image Text:top
bottom
top
bottom
top
bottom
VIP17/MAL
PLAP
caveolin-1
stomatin
Yes
TIR
gp114
calnexin
rab-5
Lubrol WX
Brij 96
Triton
Fig. 2. DRM association of marker proteins from MDCK cells. MDCK-PLAP cell homogenate was extracted with 0.5% Lubrol WX, 0.5% Brij 96, or 1% Triton,
and floated on linear sucrose gradients. The distribution of marker proteins was analyzed by immunoblotting. The light fractions from the top of the gradients
are on the left, the heavy bottom fractions on the right.
96 additionally solubilized gp114, most of TfR, and -50% of Yes
(Fig. 2 Center), whereas Triton solubilized rab-5, calnexin, gp114.
TfR, and most of Yes (Fig. 2 Right). Hence, DRM association of
these marker proteins confirms the graded selectivity of the
detergents obscrved by analysis of the plasma membrane DRM
protein content.
The results with Triton agreed with earlier observations (6, 11,
20-22). Regarding Lubrol WX, calnexin had previously been
found to be partly insoluble (15), consistent with our results. In
contrast, TfR, which in our hands largely resisted extraction with
Lubrol WX, was reported to be Lubrol-soluble in MDCK
cells (15).
Therefore, analysis of the protein contents of DRMS from
MDCK cells showed that the detergents differed considerably in
their ability to enrich strongly lipid-associated proteins; i.e., they
differed in their "DRM selectivities."
phospholipid and cholesterol/glycerophospholipid ratios were
calculated. The fold changes of these ratios relative to the TCMS
are depicted in Fig. 4. No significant changes occurred when
higher detergent concentrations were used or when DRMS
where prepared by pelleting from isolated membranes instead of
flotation from cell homogenate (not shown).
Compared with TCMS, DRMS from MDCK cells prepared
with Triton and CHAPS showed clear enrichment of sphingo-
lipids and cholesterol over glycerophospholipids. The other
detergents enriched these lipids less strongly. Nevertheless, their
capacity for enrichment correlated with the DRM specificity
with regard to proteins, such that Brij 98 and Brij 96 caused a
more pronounced enrichment of sphingolipids and cholesterol
than Tween 20, Brij 58, and Lubrol WX. The same trend was
observed for Jurkat cells, but notable differences exist. Contrary
to MDCK cells, Brij 98 and Brij 58 enriched sphingolipids and
cholesterol to a similar extent, whereas Brij 96 did not.
The behaviors of Brij 58, Brij 96, and Brij 98 in MDCK and
Jurkat cells illustrate that the effect of a given detergent may vary
between cell types. Morcover, the enrichment of sphingolipids
and cholesterol was overall less pronounced in Jurkat cells than
in MDCK cells, showing that the lipid composition of DRMS
prepared with a particular detergent can be cell type-dependent.
Next, we analyzed the fatty acid substitution pattern of
slyserophospholipids in different DRMS, Lipids from the float-
Lipid Analysis of DRMS Prepared with Different Detergents. To
analyze the lipid content of DRMS, MDCK and Jurkat cells were
used to label cither all lipids with 14C or phospholipids with 32P.
Total cell membranes (TCMS) and DRMS were obtained by
flotation on sucrose step gradients. Lipids were extracted and
resolved by TLC (Fig. 3). As observed for the protein content of
DRMS derived from the plasma mrmbrane, the amounts of lipid
presant in the DRMS varicd contaterably between detergents.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

