Q: Devise a synthesis of attached product from the given starting material. More than one step is…
A:
Q: 19) (1) Provide the majo Reduction, Clemme (A) Me2N (C) (E) (II) Provide the mechanism
A:
Q: b) Show the major product(s) of the reaction of 4-methylphenylmagnesium bromide (prepared in…
A:
Q: Devise a synthesis of each compound from benzene. но OH CH3 а. b. с. d.
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Devise a synthesis of each compound from benzene. You may also use any organic compounds having four…
A: a.
Q: Sulfur ylides, like the phosphorus ylides of Chapter 21, are useful intermediates in organic…
A: The ion containing -ve charge over C-atom as well as a +ve charge over the heteroatom (S, N and P)…
Q: Hydrogenation of alkene A with D2 in the presence of Pd-C affords asingle product B. Keeping this…
A:
Q: Devise a synthesis of each compound from the indicated starting material.
A: a.
Q: QIV: Starting from Benzene as the only organic reagent you have and use any other inorganic reagents…
A: From the benzene to benzoic acid, schematic representation is shown below.
Q: Devise a synthesis of each compound from benzene. You may also useany organic compounds having four…
A: b.
Q: Select the reagent you would use to form Product B. H2SO4, H2O OLDA OTS O NaCN O H2O elimination 20"…
A:
Q: Draw a stepwise mechanism for the sulfonation of an alkyl benzene such as A to form a substituted…
A:
Q: Devise a synthesis of each compound from the given starting materials. You may also use organic…
A: NOTE: Since, we only answer upto three sub-parts, we'll answer the first 3. Please resubmit the…
Q: Coumarin, a naturally occurring compound isolated from lavender, sweet clover, and tonka bean, is…
A: Acetate Ion will de-protonate the phenol to form phenoxide ion as follows,
Q: Devise a synthesis of attached acetal from 1-bromo-2-methylhexane,alcohols (and diols) containing…
A: We have to show mechanism of conversion of 1-bromo-2-methylhexane to acetal
Q: ducing agent, imines NEt NHẸt -H2O EINH, + HC—C—СH, H3C-C-CH3 H, Pt 30 psi ELOH H,C-CH-CH3 (23.22)…
A: When an amine reacts with ketone, imine is formed as follows:
Q: Devise a synthesis of attached compound from the indicated starting material.
A: The synthesis of given compound from the indicated starting material can be shown as follows,
Q: Devise a stepwise synthesis of attached compound from dicyclopentadieneusing a Diels–Alder reaction…
A: A stepwise synthesis of attached compound from dicyclopentadiene has to be devised.
Q: Devise a synthesis of each compound from an alkyl halide using any other organic or inorganic…
A: Synthesis of given compunds from alkyl halide are as
Q: Draw a stepwise mechanism for the following reaction that forms ether D. D can be converted to the…
A: A stepwise mechanism for the following reaction that forms ether D has to be drawn.
Q: D. Compound KILUA can be synthesized from benzene using four steps. Show the most plausible reaction…
A: A question based on organic reaction sequence, which is to be accomplished.
Q: Which compound will react the slowest upon nitration Br A D
A:
Q: Draw a stepwise mechanism for the following Robinson annulation. This reaction was a key step in a…
A:
Q: vise a synthesis for the following compound using the given starting material (benzyl alcohol) a e…
A: The question is based on the concept of organic reactions. we have to convert the reactant into…
Q: 27(c) Most basic nitrogen A or B? (Hint: The lone pairs on the two play different roles in the…
A: The basicity of the nitrogen depends on the availability of electron density on the nitrogen atom in…
Q: Devise a synthesis of each compound from cyclohex-2-enone and organic halides having one or two…
A: a) The name of the compound to be synthesized is shown below. The method of synthesis is shown…
Q: Propose a mechanism for the following reaction:
A:
Q: pen belongs to industrial method 3) The "ether synthesis" whereas is the William's process. H,SO H…
A:
Q: Provide a stepwise mechanism for the following reaction. F3C. F3C, KOC(CH3)3 HO
A: This reaction is an example of aromatic nucleophilc substitution reaction. In this reaction the…
Q: Draw a stepwise mechanism for the sulfonation of an alkyl benzene such as A to form a substituted…
A: Stepwise mechanism for the sulfonation of alkyl benzene to form a substituted benzene sulfonic acid…
Q: convery Compound C to Compound D and come up with a route for this. Design a route to take C to D by…
A:
Q: Devise a stepwise mechanism for the following reaction, a key step in the synthesis of the…
A: The diester undergoes Dieckmann reaction in the presence of base to form beta-ketoester. The base…
Q: 21.(4) Provide a mechanism for the following transformation. кон heat Br
A: ->KOH is a base which can substrate Hydrogen and give elimination reaction.
Q: Draw a stepwise mechanism for the following intramolecular reaction, which is used in the synthesis…
A: The given compound is the female sex hormone Estrone.
Q: HO.
A: The synthesis of 4-acetylbenzoic acid is given below
Q: Devise a synthesis of each compound using 1-bromobutane (CH,CH,CH,CH,Br) as the only organic…
A: Use a staring material 1 bromobutane and reagents use give in question product formation
Q: Draw a stepwise mechanism for the following reaction that forms ether D. D can be converted to the…
A:
Q: Identify F in the following reaction sequence. F was converted in several steps to the…
A: The entire mechanism of the reaction is as follows:
Q: 15.16 Show how spiro[2.2]pentane can be prepared in one step from organic compounds containing three…
A:
Q: Devise a synthesis of each compound using 1-bromobutane(CH3CH2CH2CH2Br) as the only organic starting…
A: Introduction: In the first reaction, 1-bromobutane reacts sodium cyanide to form 1-cyanobutane.…
Q: Devise a synthesis of attached compound from the indicated startingmaterial. You may also use any…
A: The reactions involving organic compounds are called organic reactions. Electrophilic and…
Q: Draw a stepwise mechanism for the intramolecular Friedel–Crafts acylation of compound A to form B. B…
A: The stepwise mechanism is shown below,
Q: 18.65 Propose a plausible synthesis for the following trans- formation. CH3 CH3 CH₂
A:
Q: Sulfur ylides, like the phosphorus ylides, are usefulintermediates in organic synthesis. Methyl…
A: Introduction: The ion containing -ve charge over C-atom as well as a +ve charge over the heteroatom…
Q: Br from phenol a. b. from benzene CI
A:
Q: By what mechanism is the following reaction likely to occur? (SN1, SN2, E1, E2) Put an "and" between…
A: In 1 reaction alkyl halide is 1°and SH- is strong nucleophile. SN2 reaction will takes place.…
Q: Devise a synthesis of each compound from benzene. You may use any other organic or in organic…
A: (a)
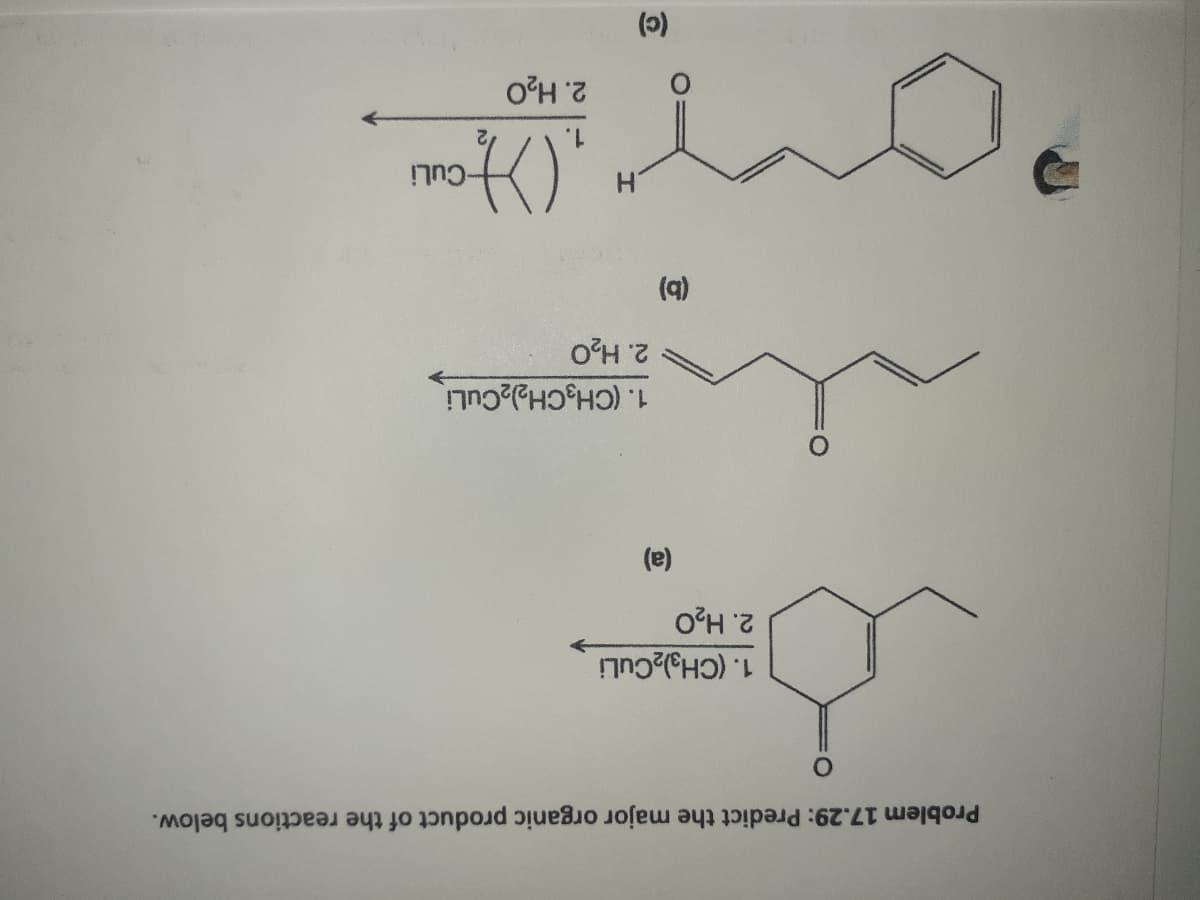
All given 3 reactions are addition of Gilman reagent on conjugate ketone or aldehyde.
This addition is also known as conjugate addition or 1,4 addition
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

- Bromoetherification, the addition of the elements of Br and OR to adouble bond, is a common method for constructing rings containingoxygen atoms. This reaction has been used in the synthesis of thepolyether antibiotic monensin (Problem 18.34). Draw a stepwisemechanism for the following intramolecular bromoetherification reaction.Salsolinol is a naturally occurring compound found in bananas,chocolate, and several foods derived from plant sources. Salsolinol isalso formed in the body when acetaldehyde, an oxidation product of theethanol ingested in an alcoholic beverage, reacts with dopamine, aneurotransmitter. Draw a stepwise mechanism for the formation ofsalsolinol in the following reaction.Devise a synthesis of each compound from benzene. You may also useany organic compounds having four or fewer carbons and any requiredinorganic reagents.
- Devise a stepwise synthesis of attached compound from dicyclopentadiene using a Diels–Alder reaction as one step. You may also use organic compounds having ≤ 4 C's, and any required organic or inorganicreagents.Identify F in the following reaction sequence. F was converted in several steps to the antidepressant paroxetine (trade name Paxil; see also Problem 9.9).Devise 2 synthesis for the given compound starting from BENZENE and other pertinent inorganic or organic reagents.
- susceptible to nucleophilic attack a to b c only a only a and cDraw a stepwise mechanism for the following Robinson annulation. This reaction was a key step in a synthesis of the steroid cortisone by R. B. Woodward and co-workers at Harvard University in 1951.can you solve 12.66. Devise a synthesis of A from the three strating materials given. You may use any other needed organic or inorganic reagents
- Slow addition of organolithium reagent A to B afforded C, anintermediate in the synthesis of the chapter-opening molecule,resiniferatoxin. Draw a stepwise mechanism for this process.Reaction of a sulfonitric mixture witha) a terminal alkyneb) a non-terminal alkyneInterestingly, reduction of the complex shown above mostly gives thealcohol enantiomer shown below after workup. a. Draw the other minor diastereomeric alcohol product. b. Briefly explain the diastereoselectivity of the reduction – why is oneenantiomer heavily favored? Draw a structure or structures to support youranswer.

