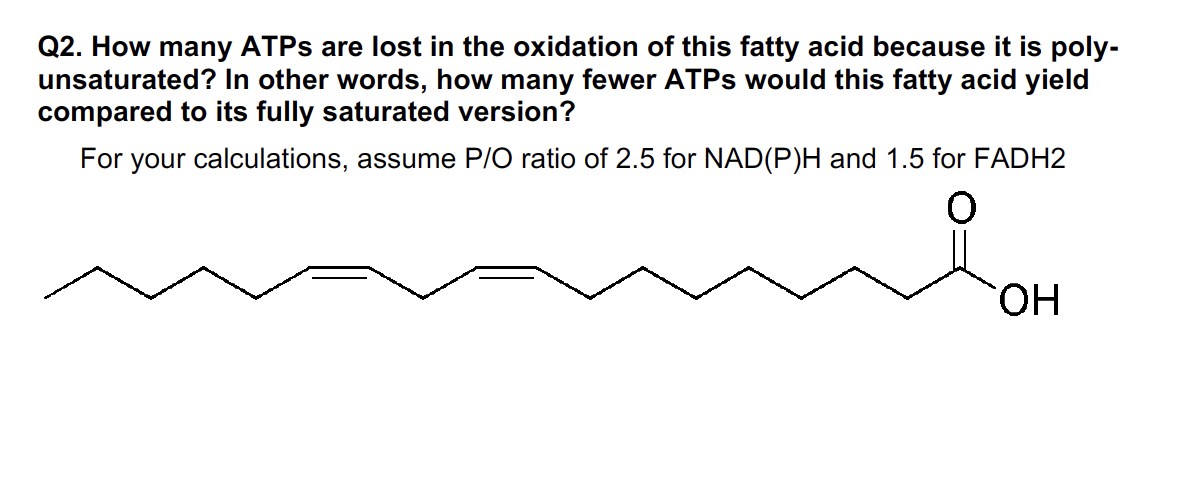
Q2. How many ATPs are lost in the oxidation of this fatty acid because it is poly- unsaturated? In other words, how many fewer ATPs would this fatty acid yield compared to its fully saturated version? For your calculations, assume P/O ratio of 2.5 for NAD(P)H and 1.5 for FADH2 OH
Q: Which of the following components are found in lipoproteins? Choose all that apply a. DNA b.…
A: Lipoproteins are essential components of the body that transport nutrients around. Lipoproteins are…
Q: In the ESTIMATION OF TOTAL SERUM CHOLESTEROL using ZAK AND HENLY’S METHOD, explain why should the…
A: In this estimation method, proteins in serum are precipitated with ferric chloride-acetic acid…
Q: Match the following descriptions with the correct lipid-based compounds:…
A: Introduction: Lipids are a heterogeneous group of biomolecules that include fats, oils, waxes, and…
Q: Which of the following is not a net product (i.e. of both phases) of the pentose phosphate shunt?…
A: Pentose phosphate pathway involves two phases , oxidative and non oxidative . It is a primary…
Q: In Zak-Henley's method in Determination of Total Serum Cholesterol, determine role of the sulfuric…
A: Cholesterol is a type of steroid. The normal serum cholesterol is 125-200mg/dL. Elevated serum…
Q: Table Q1(a) shows typical values for the intracellular and extracellular concentrations of the major…
A: All cells have an electrical potential difference or membrane potential across their plasma…
Q: What is Diabetes Mellitus?How and why does it occur?
A: Diabetes takes place while the body frame isn't always capable of absorb sugar (glucose) into its…
Q: TRUE OR FALSE: a) Okazaki fragments are short DNA pieces that explain how the DNA polymerase can…
A: DNA replication process is semi conservative. The semi conservative replication of DNA refers to the…
Q: Folate deficiency may lead to: a.Megaloblastic anemia b.Neural tube defects c.Microcytic anemia…
A: Folic acid is B family Vitamin. It aids in the formation of healthy new cells in the body. Folic…
Q: Identify the polar and nonpolar parts of the assembly. Identify the pocket and the two bilayers that…
A: Polar components are those that are hydrophilic and soluble in water. Non polar components are those…
Q: Question 11 Match the different carbohydrates' nomenclature/ glycan representation with their…
A: IUPAC-IUBMB, symbol nomenclature, LINUCS, and linear code are different nomenclatures used to name…
Q: Explain the meaning behind the term "reducing sugar ".
A: Any sugar that acts as a reducing agent due to the presence of its free aldehyde or ketone…
Q: We use 3.16 fold dilution why this dilution schedule is important to determine unknown DNA…
A: The dilutions are the geometric series i.e Serial dilutions are prepared using same dilution step as…
Q: A. Write the structure of the following peptide at pH 5.0 and calculate its net charge at this pH.…
A: Hi. Thank you for the question. As per the honor code, We'll answer the first question since the…
Q: Table Q1(a) shows typical values for the intracellular and extracellular concentrations of the major…
A: All cells have an electrical potential difference or membrane potential across their plasma…
Q: During glycolysis, glucose is converted to pyruvate. Pyruvate then is converted to Acetyl coA and…
A: Glycolysis and TCA cycle are metabolic pathways in the cell. In glycolysis one molecule of glucose…
Q: describe the structural features of protein transaminase/aminotransferases (primary, secondary,…
A: Hi! Since you have posted multiple questions and have not mentioned which to answer , we are…
Q: The reaction is reversible. Check all that apply. acetyl CoA lactate ethanol
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Write the saponification reaction of the following triglyceride with NaOH Н-С-О CH3 H-C-O- `CH3…
A: Triglycerides, also known as fat molecules, are composed of long chains of fat. In the presence of…
Q: Blood type O is also known as universal donor because it does not contain plasma antibodies. True…
A: Depending on the presence or absence of antigen or antibodies, the blood has been categorised into…
Q: As the strands are synthesized in replication, which of the following is true? the leading strand…
A: Replication:- process of formation of replica's of DNA in a semiconservative manner. Bidirectional…
Q: translation to the plasma membrane
A: Translation is a process in which mRNA is decoded into short peptide sequences made of amino acids.…
Q: 97) In order for a retrovirus to be infectious a. The p25 protein is cleaved by the protease enzyme…
A: Introduction: Retroviruses are a family of viruses that are grouped together based on how it is…
Q: .outline the processes used in the preparation of agarose gel of 1.5 concentration 5.outline the…
A: Electrophoresis is the migration of charged molecule in a medium under the influence of an electric…
Q: Mechanism for Acid-Base Balance What happens with this mechanism to regulate the body to become more…
A: The bicarbonate/carbonic acid buffer system is found in blood and many other tissues. It balances…
Q: (a) In what important ways do the α helix and β sheet structures differ? (b) How are they similar?
A: α helix and β sheet structures are the forms of the secondary shape of the protein. They each are…
Q: Match the blood glucose source with its fate/outcome.…
A: Blood sugar which is also known as glucose is described as one of the crucial as well as major sugar…
Q: When the energetic needs of hepatocytes are being met, which of the following occurs? Acetyl-CoA…
A: When carbohydrates are plentiful, the liver not only uses glucose as the primary metabolic fuel, but…
Q: List the components of the enzyme complex involved in the transition step. Simply describe what does…
A: A enzyme is made up of one or more polypeptide chains of amino acids. The amino acid causes the…
Q: Okazaki fragments are short DNA pieces that explain how the DNA polymerase can continue the…
A: Replication is the process of synthesis of new strands of DNA from the parental DNA molecule and it…
Q: Problem 2. Competitive inhibitors are commonly used to make pharmaceuticals. Some cancer drugs act…
A: We need to plot the Lineweaver Burk Plot (LB Plot) to solve this problem. So first we need to find…
Q: What is phenylketonuria? Discuss its occurrence, symptoms if any, treatments if there are, and any…
A: Phenylketonuria, also known as PKU, is an autosomal recessive disorder. Since phenylketonuria is an…
Q: On average, 180 liters of plasma are filtered each day. A If humans had to expend one molecule of…
A: The cell uses and stores ATP (Adenosine Triphosphate) as an energy source. Adenosine, ribose sugar,…
Q: What is the name of the molecule when glucose is bound to hemoglobin? a. Glycohemoglobin b.…
A: Hemoglobin is an iron-containing oxygen-transport metalloprotein located in almost all vertebrate…
Q: Glucocorticoid use results in in the amount of glucose that is taken up into muscle cells and…
A: One of the class of corticosteroid is glucocorticoids, which ate class of steroid hormones.…
Q: explain why is the importance of pharmacogenetics in the world of forensic sceicn
A: Introduction: Pharmacogenetics is the study of how genetic differences in a single gene develop…
Q: Zidovudine and Stavudine are nucleoside reverse transcriptase inhibitors which consists of a…
A: Nucleoside reverse transcriptase is RNA-dependent DNA polymerase enzyme which synthesizes the DNA…
Q: Which of the following reactions is the most exergonic? a Conversion of PEP to Pyruvate b…
A: Exergonic reaction is the reaction in which energy is released when reactants are converted to…
Q: How does the degree of unsaturation and structure of fats affects its functionality, for example in…
A: Lipids are not polymers. The simplest form of lipid is fatty acids which are a long chain of…
Q: The origin of the word "enzyme" comes from Greek and means: Group of answer choices in bacterium in…
A: An enzyme is a biological catalyst that increases the rate of biochemical reactions.
Q: To establish a standard curve for a BSA standard curve using Bradford, the spectrophotometer…
A: Proteins are composed of twenty standard amino acids attached together via peptide bonds. These…
Q: The lipase substrate emulsion contains 0.500 mg of olive oil per 3 mL Also, the molar mass of the…
A: The number of moles of a substance is calculated by using the equation, n=mM, where, "n" is the…
Q: α-D-galactose from β-D-glucose can be differentiated using which method of analysis? A.…
A: During methylation, a methyl group gets added to every free hydroxyl (-OH) group in the sugar. This…
Q: Incubating 10µl of an LDH sample for 5 min in the presence of 60mM of lactate and 100mM NAD+…
A: Lactate Dehydrogenase (LDH) activity can be figured using a spectrophotometric assay. The reaction…
Q: Categorize the electron carrier by the number of hydrogens that it can transfer in a single…
A: In the process of electron transport, one or more molecules accept electrons from another and donate…
Q: Below is a structure of a? monosaccharide oligosaccharide disaccharide polysaccharide
A: Carbohydrates are the most prevalent biomolecules on the planet. Carbohydrates are largely composed…
Q: In a different instance of (d) two intants suspected to have a defi- ciency of the PDH complex…
A: During cellular respiration, respiratory substrates like glucose may undergo complete or…
Q: Match each Sl unit to the quantity it measures. degree Fahrenheit second gram kelvin nanosecond…
A: A unit of measurement is a conventionally defined magnitude of a quantity, that is used as a…
Q: Regarding the regulation of the citric acid cycle (TCA), what are the true alternatives: a.the TCA…
A: Allosteric regulation governs the TCA cycle. The metabolites control the cycle through feedback…
Q: Give the functions of both water-soluble and fat-soluble vitamins
A: The B vitamins (folate, thiamine, riboflavin, niacin, pantothenic acid, biotin, vitamin B6, and…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Q No 2. During Respiration, various oxidation–reduction reactions occur. Could you expand upon this point further?9Calculate the total amount of ATP that is produced by the complete oxidation of a 20 carbon even-chain saturated fatty acid. Include the number of each cycle required, describe each stage involved, and list the number of ATP that correspond with each stage.Q How much more ATP is possible from aerobicallyrespiring glucose instead of fermenting it to lactate?Why is this so? Does the citric acid cycle only have acatabolic function?
- Consider decosanoic acid C12H43CO2H SUB PART TO BE SOLVED How many cycles of beta-oxidation are needed for complete oxidation? How many molecules of ATP are formed from the complete catabolism of this fatty acid? Show the complete computation. How many moles of ATP per gram of fatty acid is formed from the complete catabolism of the given fatty acid? What is the molar mass of the given fatty acid? Solution: Show here the complete computations, [from 1 to 4]12) Considering the phases of glucose utilization of glycolysis, pyruvate oxidation, and the citric acid cycle, summarize the overall net reactants and products if we start with one glucose molecule.10How many moles of ATP are generated by the complete oxidation of the fatty acid 22:3 in the presence of a functional electron transport chain and oxidative phosphorylation? Give your answer in moles, but don't include the units in your answer
- A1 Calculate the ATP yield for the complete oxidation of oleate.Write balanced biochemical equations for all the reactions in the catabolism of glucose to two molecules of pyruvate (the preparatory & payoff phase of glycolysis), including the standard free-energy change ΔG for each reaction. Then write the overall or net equation of glycolysis, with the net standard free-energy change when one molecule of glucose is converted into 2 molecules of pyruvate.Pls help ASAP, thank you! "Match the gluconeogenic precursor to the type of reaction(s) that bring it to gluconeogenesis as pyruvate" (answer choices for all drop down menus are: "alanine", "lactate", "glycerol", and "glutamine")
- a- During intense exercise the transformation glucose to lactate causes very less ATP production compared to aerobic glycolysis. Explain, does anaerobic glycolysis lead to waste of energy in muscle? b-Glycogen phosphorylase enzyme catalyzes the removal of glucose from glycogen. Describe, glycogen metabolism regulation through glycogen phosphorylase.1. For a saturated fatty acid with 28 carbon atoms: a. Write the products obtained from one cycle of beta oxidation. b. How many cycles of beta oxidation will be required for its complete breakdown? How many Acetyl CoA will be produced after its complete breakdown? (show the calculation) c. Calculate the total number of ATP molecules generated (total energy yield) after complete oxidation of this fatty acid. (show the complete Stepwise calculation)Calculate the net ATP yield from the complete processing of a saturated fatty acid containing 17 carbons. Consider the b-oxidation steps, processing of acetyl-CoA through the citric acid cycle, and electron transport

