Supply the missing data in the table below. Also, determine the FREUNDLICH isotherm constants for the following adsorption test data. The liquid volume used in the batch adsorption tests was 1 Liter. Equilibrium Concentration of Adsorbate in Solution, mg/L Mass of Adsorbent, Original Concentration of Adsorbate Weight of substance grams in Solution, mg/L adsorbed, mg 3.45 3.45 (A) 0.001 3.45 3.20 (B) 0.010 3.45 2.81 (C) 0.100 3.45 1.76 (D) 0.500 3.45 1.25 (E)
Supply the missing data in the table below. Also, determine the FREUNDLICH isotherm constants for the following adsorption test data. The liquid volume used in the batch adsorption tests was 1 Liter. Equilibrium Concentration of Adsorbate in Solution, mg/L Mass of Adsorbent, Original Concentration of Adsorbate Weight of substance grams in Solution, mg/L adsorbed, mg 3.45 3.45 (A) 0.001 3.45 3.20 (B) 0.010 3.45 2.81 (C) 0.100 3.45 1.76 (D) 0.500 3.45 1.25 (E)
Chapter4: Least-squares And Calibration Methods
Section: Chapter Questions
Problem 6P
Related questions
Question
Refer to the table below. Answer the questions that follow. Express all your answers in four decimal places. Note: Do not round off the values of the slope and the y-intercept.
Please keep your units consistent while calculating for the answers. Enclose in a box/highlight your final answers.
answer letter C
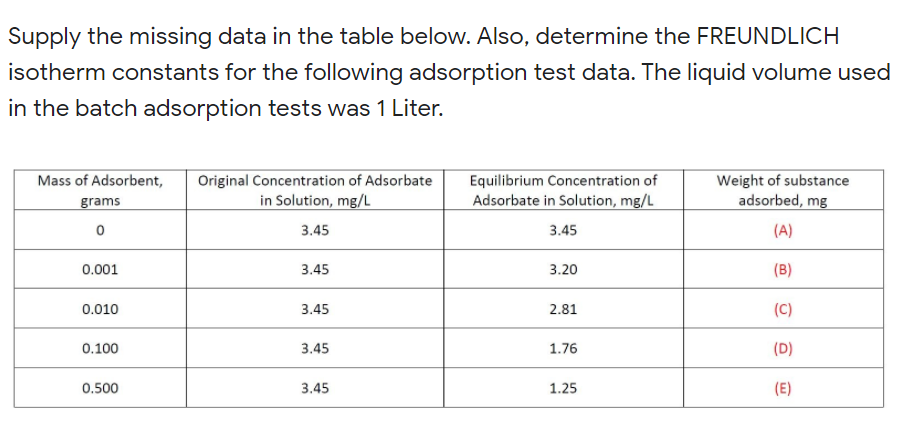
Transcribed Image Text:Supply the missing data in the table below. Also, determine the FREUNDLICH
isotherm constants for the following adsorption test data. The liquid volume used
in the batch adsorption tests was 1 Liter.
Mass of Adsorbent,
Original Concentration of Adsorbate
in Solution, mg/L
Equilibrium Concentration of
Adsorbate in Solution, mg/L
Weight of substance
adsorbed, mg
grams
3.45
3.45
(A)
0.001
3.45
3.20
(B)
0.010
3.45
2.81
(C)
0.100
3.45
1.76
(D)
0.500
3.45
1.25
(E)

Transcribed Image Text:What is the answer to (C)? *
0.64
O 1.69
O 0.25
2.20
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning



Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning