Switch KNO, Salt Bridge Cu Sn 1.0 M Cu(NO,), 1.0 M Sn(NO,)2 A student is given a standard galvanic cell, represented above, that has a Cu electrode and a Sn electrode. As current flows through the cell, the student determines that the Cu electrode increases in mass and the Sn electrode decreases in mass.
Switch KNO, Salt Bridge Cu Sn 1.0 M Cu(NO,), 1.0 M Sn(NO,)2 A student is given a standard galvanic cell, represented above, that has a Cu electrode and a Sn electrode. As current flows through the cell, the student determines that the Cu electrode increases in mass and the Sn electrode decreases in mass.
Chapter18: Introduction To Electrochemistry
Section: Chapter Questions
Problem 18.3QAP
Related questions
Question
1. Identify the electrode at which oxidation is occuring. Explain your reasoning based on the student's obervations.
2. As the mass of the Sn electrode decreases, where does the mass go?
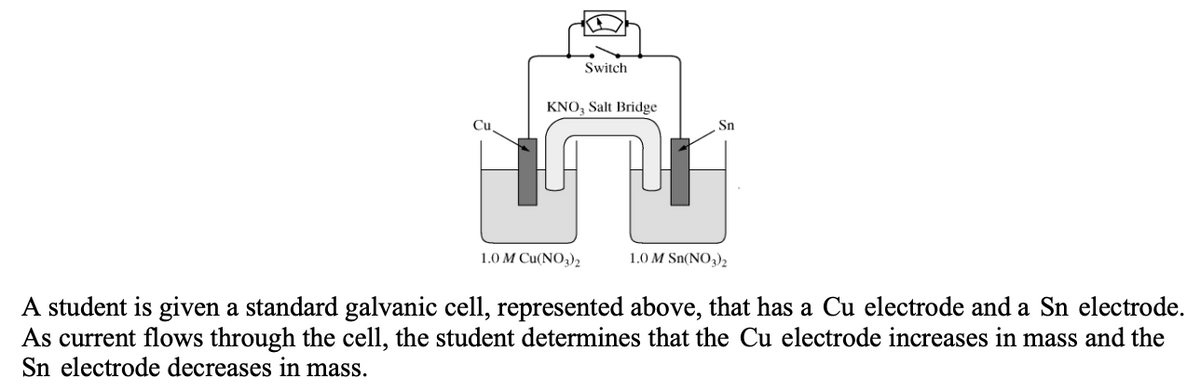
Transcribed Image Text:Switch
KNO; Salt Bridge
Cu
Sn
1.0 M Cu(NO3),
1.0 M Sn(NO3)2
A student is given a standard galvanic cell, represented above, that has a Cu electrode and a Sn electrode.
As current flows through the cell, the student determines that the Cu electrode increases in mass and the
Sn electrode decreases in mass.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning



Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning