Table 1. Required components Component Amount Concentration Sodium Citrate 92.118 dihydrate (mw: 0.0003132 M mg 294.10 g/mol) Citric Acid (mw: 131.9 0.0006868 M 192.12 g/mol) mg
Table 1. Required components Component Amount Concentration Sodium Citrate 92.118 dihydrate (mw: 0.0003132 M mg 294.10 g/mol) Citric Acid (mw: 131.9 0.0006868 M 192.12 g/mol) mg
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter15: Solutions Of Acids And Bases
Section: Chapter Questions
Problem 15.130QE: A solution is made by dissolving 15.0 g sodium hydroxide in approximately 450 mL water. The solution...
Related questions
Question
How many grams of citric acid and sodium citrate dihydrate must be added to 1L of water to produce a solution with pH 3.9 and a total solute concentration of 1 mM? (MW Salt = 294.09; MW Acid = 192.13)
I found the answer below, but my calculation does not match it. Can you please show me in detail how the calculation from the image below was done? How the got the amount of citric acid and citrate?
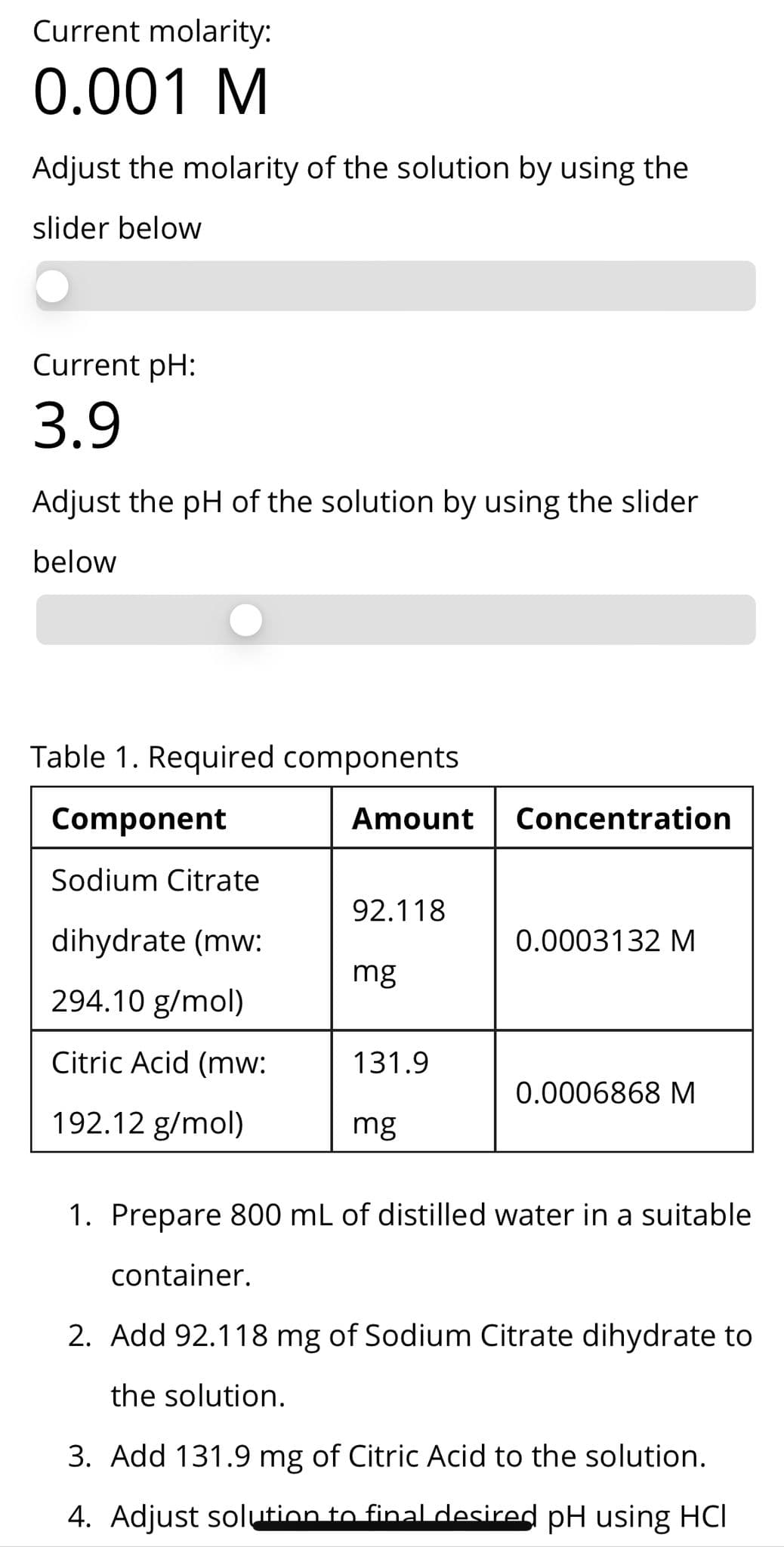
Transcribed Image Text:Current molarity:
0.001 M
Adjust the molarity of the solution by using the
slider below
Current pH:
3.9
Adjust the pH of the solution by using the slider
below
Table 1. Required components
Component
Amount
Concentration
Sodium Citrate
92.118
dihydrate (mw:
0.0003132 M
mg
294.10 g/mol)
Citric Acid (mw:
131.9
0.0006868 M
192.12 g/mol)
mg
1. Prepare 800 mL of distilled water in a suitable
container.
2. Add 92.118 mg of Sodium Citrate dihydrate to
the solution.
3. Add 131.9 mg of Citric Acid to the solution.
4. Adjust solution to final desired pH using HCI
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning