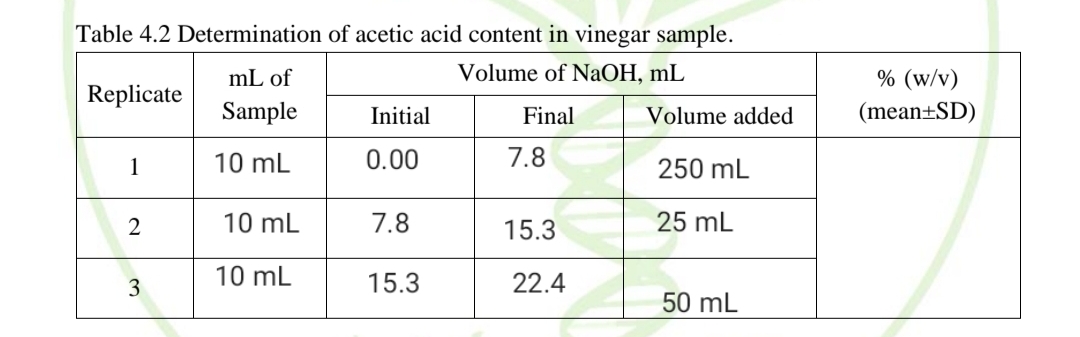
Table 4.2 Determination of acetic acid content in vinegar sample. mL of Volume of NaOH, mL % (w/v) Replicate Sample Initial Final Volume added (mean±SD) 1 10 mL 0.00 7.8 250 mL 10 mL 7.8 15.3 25 mL 10 mL 15.3 22.4 3 50 mL
Q: Results: Part 1: Sensor reading Solution Step Initial Vol (ml) Final Vol (ml) Molarity Moles of…
A: We are given the volume(V) and molarity(C) of each solution. We can find the moles of the solute…
Q: Study the tabulated data during an gravimetric analysis of BaSO4 experiment Trial T1 T2 T3 Mass…
A:
Q: B. DAY 2. Percent Acetylsalicylic Acid in the Aspirin Sample Calculation for the mass of aspirin for…
A: Given: The molarity of the NaOH solution is 0.1 mol/L. The mass of the aspirin sample taken in trial…
Q: Calculate the concentration of Pd<t ion in an unknown sample based on the following data: Vol. of…
A: According to the mole concept, in terms of mass, the amount of substance in moles is equal to the…
Q: Assume the data below was collected for two different sample solutions using the same experimental…
A:
Q: Study the table below and identify the best solvent for recrystallization for compounds A, B, and C.…
A: Solution- Data given- Solubility data in Hot and cold solvent.
Q: A. Standardizatlon of thlosulfate solution Trial 1 Trial 2 Trial 3 Initial volume of thiosulfate…
A: Molarity = number of moles of solute / volume of solution We are given data for initial and final…
Q: Preparing and Diluting Solutions Data Table Test Tube 1* 2 4 10ML/3.8mL 2.4mL SmL 1.53ML…
A: We are given the data for the concentration of different diluted stock solutions and their…
Q: Prepare six different concentrations of Bovine Serum Albumin (BSA) standard from a 2 mg/mL stock.…
A: As we are given the reactant as BSA which is referred as Bovine serum albumin and we are given that…
Q: The thiourea in a 1.455 g sample of organic material was extracted into a dilute sulfuric acid…
A: Generally, qualitative analysis refers to the presence or absence of different chemical (number of)…
Q: (Concentration) You are tasked to prepare 62.5 ppm of a caffeine standard for an analysis. You need…
A: Concentration in ppm=(mass of solute/Mass of solvent) × 10^6 Previously Concentration 0.025gm…
Q: Part A Are the following solutions isotonic, hypotonic, or hypertonic compared with physiokogical…
A:
Q: lews SEC # Evaluation of Commercial Bleaching Solutions 22 DATA SHEET Brand name of commercial…
A: Concentration of NaOCl = M1 = 0.175M VOlume of NaOCl = V1 Concentration of KI = M2 = 0.225M Volume…
Q: . Does the value of the equilibrium constant change when the concentration of either the reactants…
A: You have asked about part (B) only, I have to answer part (B),so, let's start. Given, Balanced…
Q: Calculate the solubility of Mg(OH)2 in 0.10 M Mg(NO3)2. Calculate the solubility in grams/liter…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 7.7 Separation of Two Solutes Having Linear Equilibrium Two solutes have a linear equilibrium with…
A: The effective velocity of solute: u=vε+1-εK The superficial velocity is given by: v=ratearea=rAπr2
Q: Volume of Volume of 0.1 M HCI solution Volume of Concentration 0.2 M Na,s,0, Time (s) distilled of…
A: Titration is the quantitative method of estimating the strength of the any component. Molarity is…
Q: Chemistry 1) A new well has been drilled for a municipality. Based on the analysis, please recommend…
A: The quality of drinking water is very much related to the health of individuals. World Health…
Q: PROCEDURES: A) Preparation of three Standard ASA solutions. 1) These solutions will be prepared…
A:
Q: Table 1: The result for test tube I to 8 using of BSA stock solution. Tube BSA Cone H;O (ml) BSA…
A: Lambert Beer's law states that absorbance of a solution is proportional to concentration of…
Q: A 50.00 (±0.03) mL portion of an HCl solution required 29.71(±0.03) mL of 0.01963(±0.0030) M Ba(OH)2…
A: Given, MHCl=29.71 ml×0.01963 mmol Ba(OH)2ml×2mmol HClmmol Ba(OH)250.00 ml
Q: Table 1: Volumes in of iodine in mL used to titrate mixtures of 10 mL of reaction mix and 10 mL of…
A: Given : CF = 1.06 TVS (mg/hour) = x (ml) X CF ( mg vit.C/ mL iodine ) X TV (mL/…
Q: V. Data and Results Given: 0.1 N standard NaOH solution Sample 1 1.3296 g Sample 2 0.78 g Sample 3…
A: Answer:- This question is answered by using the simple concept of stoichiometry which involves the…
Q: What is the solubility of CdS (Ksp = 1.0 x 10-28) in pure water? Group of answer choices 1.0 x…
A: The solubility reaction is CdS (s) -----> Cd2+ (aq) + S2- (aq) Hence Ksp = [Cd2+][S2-] where…
Q: Is this calculation correct? See image below....
A: The calculation is correct.
Q: Show how to take 3.6E-7 mol/L AgCl and convert it to g/100 mL
A: Given: 3.6 x 10-7 mol/L AgCl To convert: 3.6 x 10-7 mol/L AgCl to g/100 mL AgCl conversion…
Q: 2. 25.00 mL of Saturated solution of KHTar that contained 0.10 M NaCl: NaOH Volumes: Final 46.30 mL…
A:
Q: For the CO2 impact experiment station: 1) Obtain two plastic cups, red cabbage indicator, a…
A:
Q: Explain why it is incorrect to collect data for temperatures above 100 C in terms of the solute to…
A: A question based on calibration method that is to be accomplished.
Q: Methodology: Make schematic diagram for the procedure below B. % SO3 determination Dry the soluble…
A: We need to make a schematic diagram for the given procedure.
Q: Objectives: To determine the percent mass of acetic acid in vinegar Data: Vinegar Sample : Trial 1…
A: We are given the experimental data and we have to calculate the mass % of acetic acid in vinegar…
Q: A 10 ml of concentrated acetic acid was added in a sufficient quantity of water to make 350 ml of…
A:
Q: In order to form Ba(IO3)2, 500 mL of 0.5000 M Ba(NO3)2 was mixed with 500 mL of 0.0500 M NaIO3.…
A:
Q: -Volume of unknown acid___ -Average molarity of NaOH from above__ -mL of NaOH at equivalence point__…
A: Solutions- Given data- Sheet of pH readings, Molarity of NaOH etc.
Q: Please provide a detailed solution for the following question You have constructed a standard curve…
A:
Q: 0.02 M NaOH reagent was standardized using 0.1000 g Potassium hydrogen phthalate (204.22 g/mol) and…
A: Given:- Mass of Potassium hydrogen phthalate = 0.1000g Molar mass of Potassium hydrogen phthalate…
Q: Objective: To determine the percent NaCl (w/v) of a water sample 5.00 mL sample diluted to 50 mL…
A: The question is based on the concept of quantitative estimation. we have to calculate percentage…
Q: Jake carried out three titrations to determine the concentration of unknown nitric acid (HNO3) using…
A: [NaOH] = 0.1050 M Vol of HNO3 used = 100.0 ml Vol of Na OH used (Va)= 20.56 ml Vol of Na OH used…
Q: Chemistry Rx Cocaine HCl 1.5% (E = 0.16) Eucatropine HCl…
A:
Q: Part 2 Calculations Trial 1 Trial 2 Trial 3 24.20 24.60 24.70 4. Volume NaOH Used (mL) Part 3 of 10…
A: In this question, given the three different values of the volume of NaOH And given molarity is…
Q: In this experiment it takes about 10 microliters of solution to produce a spot 1 cm in diameter. If…
A: Different units are used to express the volume and mass of substances such as grams, micrograms,…
Q: B. %SO3 determination 1. Dry the soluble sulfate sample at 100°C for 1-2 hours, and cool in…
A: We need to make a schematic diagram for the given procedure.
Q: Q2: Calculate the solubility of Mg(OH)2 in 0.10 M NaOH.Calculate the solubility in grams/liter (use…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Stock Solution : 5.5% m/v Dilute To : 4.8 % New Volume : 80mL FIND THE : Assigned concentration for…
A:
Q: Isotonic saline solution is 0.154 M NaCl(aq). What is the solubility of AgCl (Ksp = 1.8 × 10−10) in…
A: Given data : Concentration of NaCl = 0.154 M Ksp of AgCl = 1.8 × 10-10
Q: A 200 mL water sample was treated with SPE column for pe- concentration of a certain pesticide. The…
A: Introduction: High-performance liquid chromatography (HPLC) is an analytical technique to separate,…
Q: Post-Lab Question(s) 1. Write an experimental procedure describing how to use recrystallization…
A: According to the given question, the compound contains two impurities. Both the impurities are…
Q: What is the concentration of HI being serial diluted 6 times with a total dilution factor of 106?…
A: Given: Concentration of stock HI solution = 120 mM = 0.120 M (Since 1 M…
Q: Initial burette reading (mL) 2.29 1.41 1.95 Molarity of NaOH (M) 0.100 0.100 0.100…
A:
Q: qualitative and quantitative analysis?
A: Ans. Sodium chloride can be analysed by various analysis techniques: Flame photometry is an atomic…

Step by step
Solved in 2 steps with 2 images

- Which of the following methods of preparing 0.0010M HCl solution from 1.0M stock HCl Solution willyield the smaller overlall uncertainty? a. A one step Dilution that uses 1mL(±0.006) pipet and 1000mL(±0.30) Volumetric Flask, or b. A two-step dilution that uses 20mL (±0.03) pipet and a 1000mL(±0.30) volumetric flask and a25 mL (±0.03) pipet and a 500mL(±0.20) volumetric flask for second dilution.A 18 g of unknown organic sample was dissolve in 756 mL of benzene. The boiling point of benzene was increased by 3.36oC. As the first step of analysis, determine the moecular weight of the unknow sample? Kb of benzene= 2.64oC/m Bb of benzene = 80.09 oC density of benzene = 0.874 g/mL at 25 °C Answer in whole number, no units required.A chemist receive different mixtures for analysis with the statement that it contain NaOH, NaHCO3 , Na2CO3 or compatible mixtures of these substances together with the inert material. From the data given, identify the respective materials and calculate the percentage of each component. 1.000g samples and 0.2500 N HCl were used in all cases. Sample 1 With phenolphthalein as the indicator, 24.32 ml of HCl was used. A duplicate sample required 48.64 ml HCl using methyl orange as the indicator. Sample 2. With phenolphthalein as the indicator it uses 28.2 ml of HCl to make it colorless and added with methyl orange indicator and uses 11.3 ml of HCl to reach the end point.
- The gravimetric factor used to express CoCBr6·H20 in a sample that is finally weighed as PbClBr is choose below: FW PbClBr / 6 x FW CoCBr6·H20 FW CoCBr6·H20 / FW PbClBr FW CoCBr6·H20 / 6 x FW PbClBr 6 x FW PbClBr / FW CoCBr6·H20Study the tabulated data during an gravimetric analysis of BaSO4 experiment Trial T1 T2 T3 Mass of sample (g) 1.0040 1.0100 1.0050 Constant weight of empty crucible (g) 10.2453 10.2454 10.2454 Constant weight after ignition (g) 10.3253 10.3252 10.3253 1. Calculate the mean mass of precipitate as BaSO4 a. 0.0799 g b. 0.0800 c. 0.08 g 2. Calculate the mean weight of sulphate (MM BaSO4 = 233.39 g/mol, So42- = 176 g/mol)? a. 0.0603 g b. 0.06 g c. 0.06025 g 3. Calculate the mean % SO42- in the sample a. 6.00% b. 5.991% c. 5.9908%Volume of an unknown used was 30 mL, Initial Buret volume was 0 and the Final Buret volume was 8.5 mL. What is the molarity of the unknown solution if the Net volume of NaOH being used was 8.5 mL and Millimoles (mmoles) NaOH reacted was 0.791? Then, what is the Mass (g) of Acetic Acid in unknown sample and thr average percent (%) Acetic Acid? (assume density = 1g/mL)
- Q3 / The solubility of sodium chloride NaCl in water at 290 Kis 35.8 kg / 100 kg of water. Express the solubility as the following: 1. Mass fraction and the mass percent of Naci 2. Mole fraction and mole percent of NaCl 3. kmol of NaCl per 1000 kg of water Note: molecular weight of NaCl = 135.8, and water = 18.016Gypsum (CaSO4) is a common percipitant in water desalination. CaSO4 <=> Ca2+ + SO42- where Ksp=10-4.6. Assuming that: [Ca2+]=2x10-2 M; [SO42-]=2x10-3M: a) Find Qsp or IAP for the given water b) Find the solubility index (SI) and determine whether CaSO4 is under, super, or at saturation in water. Feel free to make any assumptions you wish, as long as they're stated.Bristol Community College Fall River, Massachusetts Experiment 6: Molar Mass of a Molecular Solid from Freezing Point-Depression Measurement Name: __________________________________ Date: _______________ Approved: ___________ DATA SHEET Mass of lauric acid (in Part II) Mass of benzoic acid (in Part II) Freezing temperature of pure lauric acid (from Part I) data from Video 2 of Part I Freezing temperature of solution (from Part II) data from Video referenced in Part II Freezing point depression, Tf ( = Tf, lauric acid – Tf, solution) Molality (m) of solution ( Eq. 1) Moles of benzoic acid ( Eq. 2) Experimental molar mass of benzoic acid (Eq. 3) Calculate the molar mass of benzoic acid, C6H5COOH. Percent error Summary Questions A student determines…
- A 50.00 (±0.03) mL portion of an HCl solution required 29.71(±0.03) mL of 0.01963(±0.0030) M Ba(OH)2 to reach an end point with bromocresol green indicator. The molar concentration of the HCl is calculated using the equation below (attached image): a.) Calculate the uncertainty of the result (absolute error). M=0.02333(±?????) M b.) Calculate the coefficient of variation for the result. CV= (Sy/y) x 100%Given the following data and the λmax determined in the previous questions, determine:1) the molar absorptivity of copper at λmax of cobalt 2) the molar absorptivity of copper at λmax of copper 3) the molar absorptivity of cobalt at λmax of cobalt 4) the molar absorptivity of copper at λmax of cobalt 5) the concentration of cobalt in mM 6) the concentration of copper in mM Copper concentration, mM λmax cobalt λmax copper 20.00 0.177 0.238 40.00 0.382 0.487 60.00 0.518 0.731 80.00 0.765 1.134 100.00 0.902 1.391 Cobalt concentration, mM λmax cobalt λmax copper 20.00 0.186 0.163 40.00 0.412 0.354 60.00 0.637 0.477 80.00 0.754 0.694 100.00 0.961 0.831 unknown, Abs at λmax cobalt 0.893 unknown, Abs at λmax copper 0.997EX 2.Given-> Volume of Na+ = 500 ml Molarity of Na+= 0.0100M Molar mass of Na2CO3 = 105.99 gm/mole Millimole of Na+ = molarity × volume Number of millimole = 0.0100 × 500 = 5 millimole Na2CO3 ---> 2Na+ + CO32- Millimole of Na2CO3 = millimole of Na+/2 Millimole of Na2CO3 =5/2 = 2.5 millimole Mole of Na2CO3 = 2.5 × 10-3mole (1 mole = 10^3 millimole) Weight of Na2CO3 required = mole × molar mass = 2.5 × 10-3 × 105.99 =0.26 gm Hence, 0.26 gm Na2CO3 must dissolve in 500 ml of water.





