Q: 14. Acetylene gan, CH, can be prodaced by the seachion of cala CaC, () + 2H,0 - How many lites of…
A:
Q: Pentaborane BgHg(s) burns vigorously in 02 to give B203ls) and H2O). Calculate AHoxn for the…
A: Given: ∆Hof[B2O3(s)] = -1273.5 kJ/mol ∆Hof[B5O9(s)] = 73.2 kJ/mol ∆Hof[H2O(l)] = -285.8 kJ/mol moles…
Q: Predict the major produc 1) H2, Lindlar's catalyst 2) HBr Br Br Br Br Br В E D C A
A: Lindlar catalyst: Alkynes can be selectively reduced to an alkene by a Lindlar catalyst. The Lindlar…
Q: The reactions of [PICI4]2- with NH3 (reaction I) and of [PTC14]2– with [NO2]- followed by NH3…
A: Answer: option d: cis -[PtCl2(NH3)2]; II: trans-[PtCl2(NH3)(NO2)]- is a correct option
Q: Please explain why this reaction will NOT WORK. 1. Br2, FeBr3 2. AICI3, Br
A:
Q: d) Calculate the atom economy for Ethylene Oxide (CH;CH2O) preparation CH2=CH2 + Cl2 +H2O CICH,CH2OH…
A: Atom economy - It is a measurement of the amount of starting materials (reactants) that end up as…
Q: Benzene is one of the compounds used as octane enhancers in unleaded gasoline. It is manufactured by…
A:
Q: (b) The rate constant for the solvolysis of X=H, Y=H is 1.14 times greater than that of the…
A: C-D bond energy > C-H bond energy Reaction proceeds through the carbocation intermediate which is…
Q: Where did the last phase of H3O+ come from? Because quenching work is needed to make it OH at the…
A: GIVEN:-
Q: Functional groups such as alkynes react the same in complex molecules as they do in simpler…
A: On hydroboration of the terminal alkyne given the aldehyde, compound that formed in the reaction is…
Q: The following reactions: Pb2++ 2l--->Pbl2 2Ce4++ 2l--> l2 + 2Ce3+ HOAc + NH3--> NH4+ + OAc - are…
A: Formation reaction Formation reaction is the formation of one mole of a substance from its…
Q: below. H2(g) A Pt H2(g) В Lindlar's catalyst H2(g) Na D NH3(1) 1. NaBH4 E 2. H30*
A: When alkyne is treated with Na/liq. NH3 then trans-alkene is formed. The reaction mechanism follows…
Q: What starting materials or reagents are best used to carry out the following reaction? .CI O HNO3,…
A:
Q: J. Carefully evaluate each reaction equation below for any flaws. Briefly explain any errors for TWO…
A: Note- The given question is a multiple question, hence I solved first question according to rules of…
Q: 2. (a) Determine if the following reaction is likely to be SN1, SN2, E1 or E2, discuss your…
A: Given reaction:
Q: What conditions does the Wurtz-Fittig reaction requires for making biphenyl (Temp, duration, N2 or…
A: Formation of alkylated, aromatic hydrocarbons by coupling of alkyl and an aryl halides mediated by…
Q: 2. Write the product(s) for the following chemical reactions. а. + Br2 + Br2 H2O H3C- -CH3 + KMNO4…
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If…
Q: Create sywthesis worng at least your owN 6or the following reagants: ) Towes 2) LIAIRY )…
A:
Q: What potential future problems could be involved in instructing customers of craft breweries to…
A: Craft brewers play a significant role in society and have expanded significantly in the brewing…
Q: In each reaction box, place the best reagent and conditions from the available reagent list. 1.…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
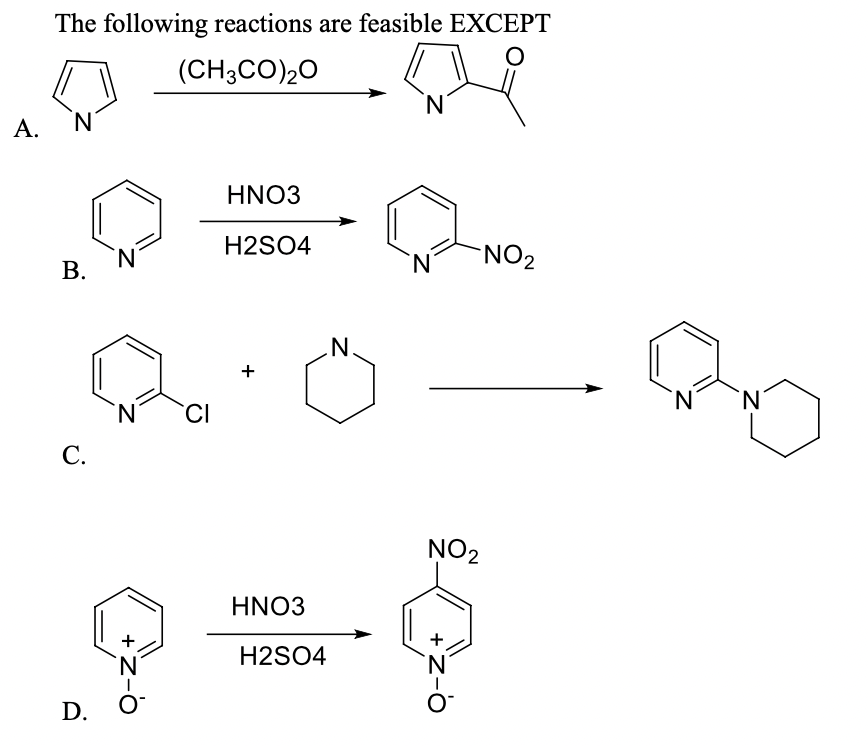
Q: 36. Which of the following reactions is /are feasible: NaNH2 CH3COCI I. NH3 NaCl СНЗСONH2 II. SOCI2…
A: Answer given in step 2.
Q: Please explain why this reaction will NOT WWORK. NO2 1. HNO3, H2SO4, 2. EtCI, AICI3
A:
Q: CH3 CH;CHCH,CH,CH3 „CHCH,CH,CH3 а. Cl AICI3
A:
Q: from H =H + enatiomer Br 3. Hz, Pd, Lindlar's catalyst 4. H,C A 1. NANH2, NH, 2. Br 1. NANH2, NH, 2.…
A: ->NaNH2 is base which can abstract most acidic hydrogen and form nucleophile.which can give…
Q: 1 mole Br /2 moles H,/Pt 2HC1 1 mole H2 (a) Lindlar's cataly st NANH2 -CH3C=CH 1. disiamylborane 2.…
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Give the organic chemical reaction mechanism of the synthesis of 1-bromobutane. link of the video…
A: Given Give the organic chemical reaction mechanism of the synthesis of 1-bromobutane.
Q: NABH4 1) LAH H20 2) H30*
A: The reaction given is,
Q: Why CH3COCI/AICI, mixture is used for Friedel-Crafts acetylation reaction and not CH;COCI/H,SO4…
A: Friedal craft reaction is electrophilic substitution reaction, in which electrophile substitute at…
Q: Which of the following reactions is /are feasible? NaNH2 NaCI H3C CI NH3 H3C `NH2 II SOCI2 OH H3C OH…
A:
Q: In halogenation reactions, why are halogens added on opposite sides of the compound? because the…
A: Ans:- Because of the formation of a halonium ion which sterically blocks one side of the compound…
Q: Which of the following reactions is /are feasible? NANH, NaCI H3C CI NH3 H3C NH2 II SOCI, OH- H3C…
A:
Q: Pls ignore the highlighted part __________ is reduced in thefollowing reaction: Cr2O72- + 6S2O32-+…
A: In this question we have to tell which species is reduced in the given redox reaction.
Q: Do you expect Step 2 to be easier than Step 1? Why or why not?
A: The reaction of bromobenzene with nitric acid and sulfuric acid results in the formation of…
Q: Will the same result be obtained if too much aluminum foil will be added in the reaction? Why or why…
A: Redox reactions are the combination of reduction and oxidation reactions. The type of reaction in…
Q: Draw the product(s) of the following reactions.
A:
Q: >...SH N3 N3 Cl +
A: The conversion of alcohol to thiol can be carried out in a number of steps involving tosylate…
Q: b) Write the products for each of the following reactions (remember to add phase labels for each…
A: Given, Correction: MgNO3(aq) formula is incorrect. The correct formula is Mg(NO3)2(aq). i)…
Q: excess HBr
A: The given reaction of dimethyl tetrahrdrofuran with excess HBr gives the…
Q: Classify each of the following reactions: A. C2H4 (g) + 302 (g)→2CO2(g)+ 2H2O(g) B. 2H9O(s)→2H|(1) +…
A:
Q: (e) Provide alternative to the following conventional reaction schemes and state the major advantage…
A:
Q: (b) Fill in blanks A to D in Scheme Q13b to indicate the missing reaction conditions or products (no…
A:
Q: Grignard reaction: Explain in detail the percent yield and theoretical yield for the following: -…
A: The formation of grignard can be given as :-
Q: nce the given reaction for step 5 (use 1 for single species - even though it is not needed, please)…
A:
Q: PART II. REACTIONS OF HYDROCARBONS Professor Lolita Carbon has assigned the analysis of unknown…
A: A question based on qualitative analysis, which is to be accomplished.
Q: Sample Brz in CCI4 (dark) Brz in CCI4 (light) KMNO4 in NaOH AGNO3 in NH3 Fading of reddish- brown…
A: A question based on qualitative analysis, which is to be accomplished.
Q: In the reactions below, there is a main reaction step followed by one or more workup steps (which…
A: Work up stage is very important For organic synthesis . This is the purification stage or to remove…
Q: In each reaction box, place the best reagent and conditions from the list. 2. H,C H CH;CH, он но…
A: The reaction given is,
Q: Predict deltarH for this reaction: C2H4(g) + H2O(l) -> C2H5OH(l) using the enthalpy changes of the…
A: We are having an aim equation that is equation we have to achieve to get ∆Hr For the reaction. We…
Step by step
Solved in 2 steps with 1 images

- average cycle time with the doubled KBrO3 concentration is 40 and average cycle time with initial KBrO3 concentration is 74.For the reaction 5Ce4+ + Mn 2+ + 4H2 O → 5Ce3+ + MnO4- + 8H+ , given E0 (Ce4+ /Ce3+ ) = 1.70V,E0 (MnO4- /Mn2+ ) = 1.507V, calculate ΔG0 for this reaction ?Which processes shown in this figure involve the phasetransition H2O(l )----->H2O(g)?
- Prof. Neiman and Prof. James were first to discovered in 1973 that chlorofluorocarbons (CFCs) were depleting the Earth’sozone layer when released into the atmosphere. Once they reach the stratosphere, Clis released from the CFCs molecules by interaction with UV light. Free Cl atoms areable to react with ozone in a catalytic cycle that converts O3into the more stable O2.It is estimated that a single Cl atom is able to react with∼100000 O3molecules.Although CFCs production was banned in 1996, there are still a substantial numberof motor vehicle air conditioners (MVACs) that use CFC-12 (CF2Cl2) as refrigerant.The average CFC-12 emission rate from operating MVACs has been estimated tobe 59.5 mg per hour per vehicle (Zhang et al.Environ. Sci. Technol. Lett.2017).How much chlorine, in kg, is added to the atmosphere in a year due to 100 millionMVACs using CFC-12 as refrigerant?Consider the reaction Mn+ + ne- ⇌ M. If ΔG*c = ΔG* + (1-α)nFE and η = E – Eeq, prove that ic = io exp[-(1-α)nFηa)/RT)].This reaction must have a ∆G<0 at SATP: Xe(g)+F2(g)→XeF2(g) 2Na(s)+Cl2(g)→2NaCl(s) 2NaF(s)+I2(s)→2NaI(s)+F2(g) MgSO4(s)→Mg(s)+S(s)+2O2(g)

