The radius of a lead atom is 175 pm. How many lead atoms would have to be laid side by side to span a distance of 1.09 mm? atoms Submit Answer Try Another Version 1 item attempt remaining
The radius of a lead atom is 175 pm. How many lead atoms would have to be laid side by side to span a distance of 1.09 mm? atoms Submit Answer Try Another Version 1 item attempt remaining
Chapter2: Atoms And Molecules
Section: Chapter Questions
Problem 2.66E
Related questions
Question
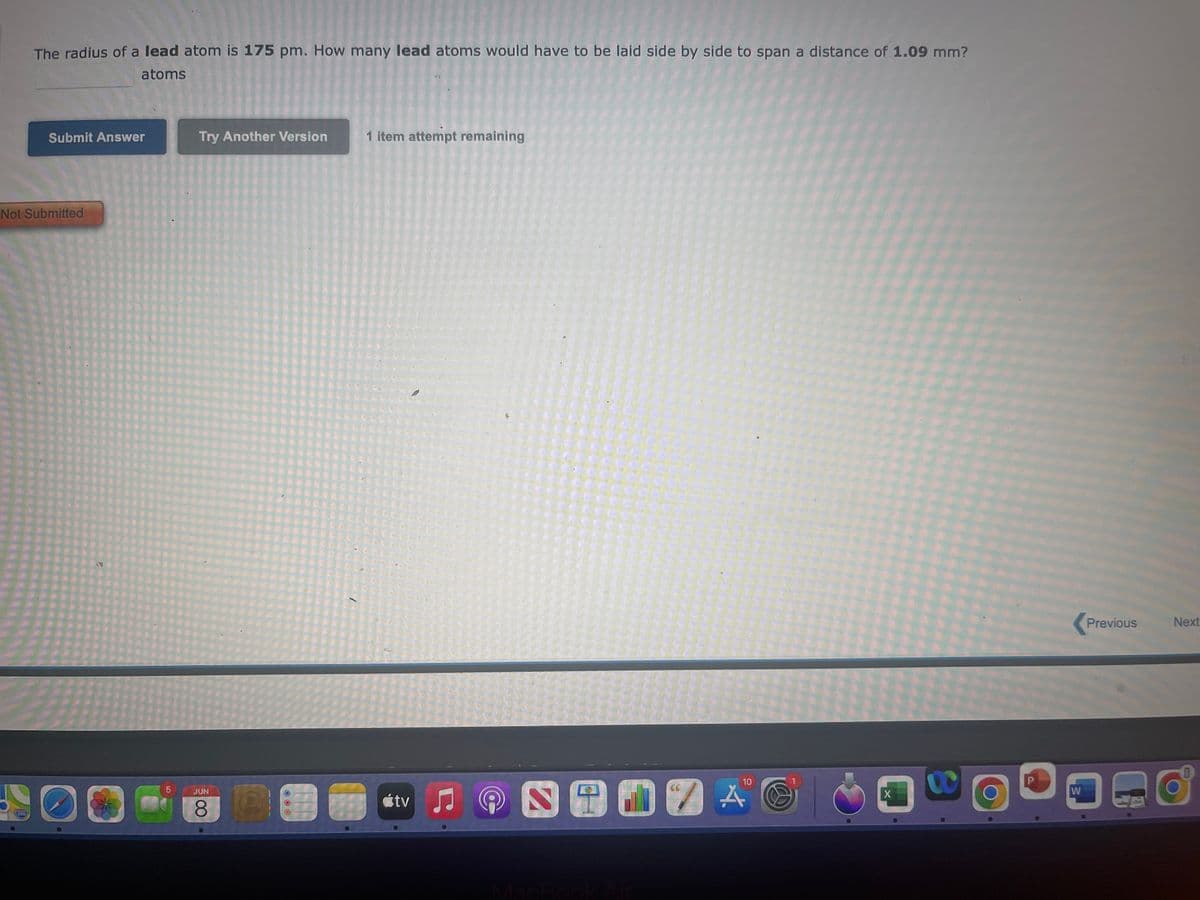
Transcribed Image Text:The radius of a lead atom is 175 pm. How many lead atoms would have to be laid side by side to span a distance of 1.09 mm?
atoms
Submit Answer
Try Another Version
1 item attempt remaining
Not Submitted
280
5
JUN
8
E
tv
5
Ⓒ
10
SENZA
X
P
Previous
W
D
Next
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax