The tonicity of the solution in container C is
Biology (MindTap Course List)
11th Edition
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Chapter2: Atoms And Molecules: The Chemical Basis Of Life
Section2.6: Acids, Bases, And Salts
Problem 1C
Related questions
Question
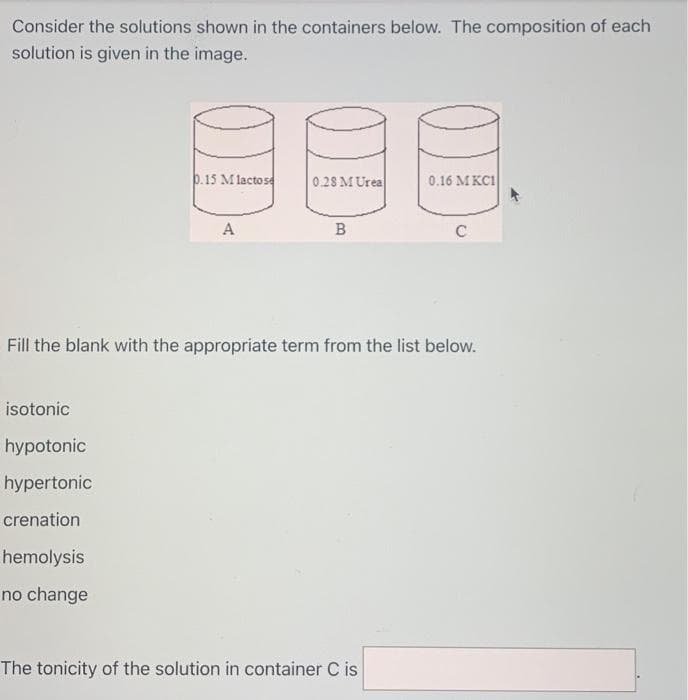
Transcribed Image Text:Consider the solutions shown in the containers below. The composition of each
solution is given in the image.
0.15 Mlactose
isotonic
hypotonic
hypertonic
crenation
hemolysis
no change
A
0.28 M Urea
B
Fill the blank with the appropriate term from the list below.
0.16 MKCI
The tonicity of the solution in container C is
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning



Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning



Human Biology (MindTap Course List)
Biology
ISBN:
9781305112100
Author:
Cecie Starr, Beverly McMillan
Publisher:
Cengage Learning