This graph shows how the vapor pressure of three liquids varies with temperature: 900- B00 700- 600 50 400 300 acetylacetone piperidine - pyridine 200 100. 100 110 120 130 140 temperature, °C Use the graph to answer the following questions: most volatile: choose one Which liquid is the most volatile? Which is the least volatile? least volatile: choose one acetylacetone: What is the normal boiling point of each liquid? Note: your answer must be within 1°C of the exact answer to be graded correct. piperidine: °C pyridine: O more Suppose a beaker of piperidine is put inside a sealed tank containing piperidine gas at 103. degree C and 848. torr. After ten minutes, will there be more liquid in the beaker, less liquid, or the same amount? O less O the same vapor pressure, tor
This graph shows how the vapor pressure of three liquids varies with temperature: 900- B00 700- 600 50 400 300 acetylacetone piperidine - pyridine 200 100. 100 110 120 130 140 temperature, °C Use the graph to answer the following questions: most volatile: choose one Which liquid is the most volatile? Which is the least volatile? least volatile: choose one acetylacetone: What is the normal boiling point of each liquid? Note: your answer must be within 1°C of the exact answer to be graded correct. piperidine: °C pyridine: O more Suppose a beaker of piperidine is put inside a sealed tank containing piperidine gas at 103. degree C and 848. torr. After ten minutes, will there be more liquid in the beaker, less liquid, or the same amount? O less O the same vapor pressure, tor
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter8: Molecules And Materials
Section: Chapter Questions
Problem 8.87PAE: 8.87 Use the vapor pressure curves illustrated here to answer the questions that follow. (a) What is...
Related questions
Question
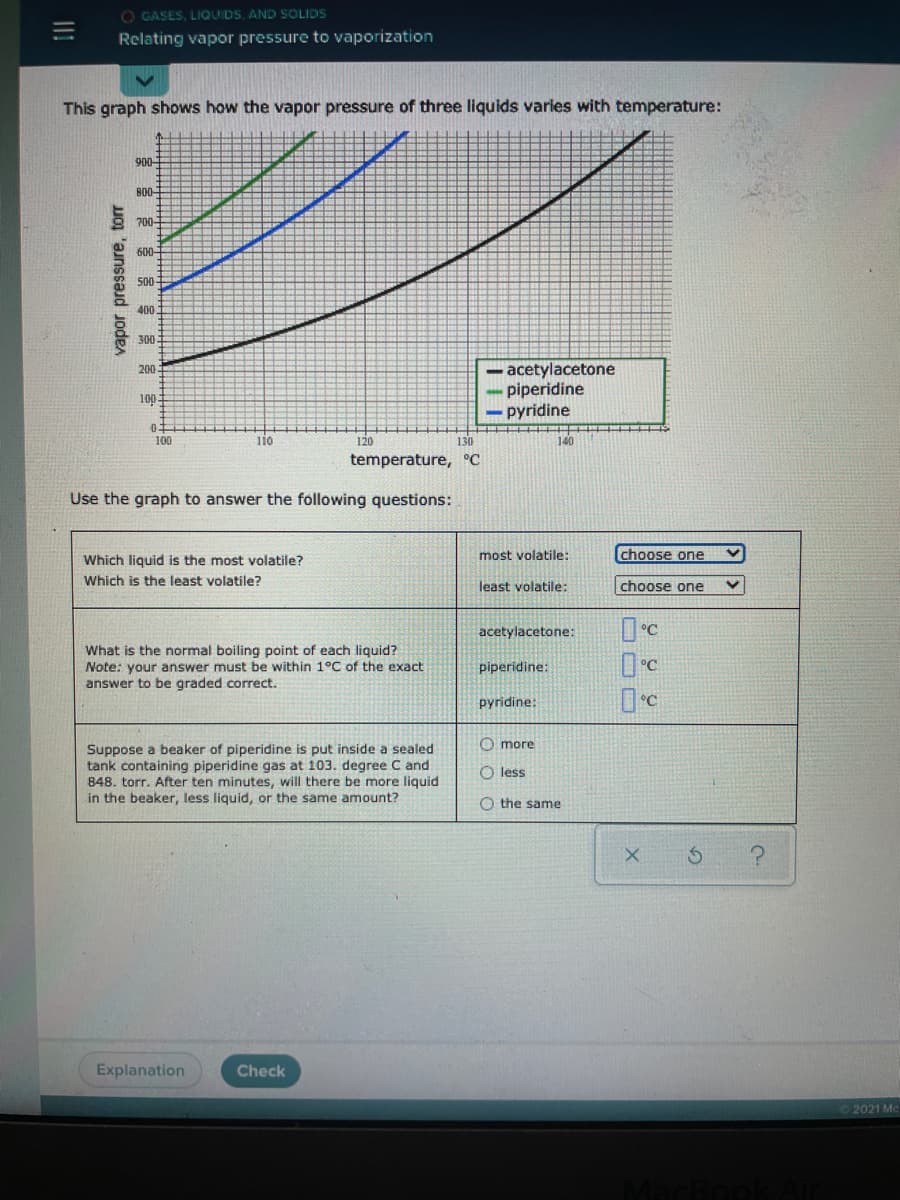
Transcribed Image Text:O GASES, LIQUIDS, AND SOLIDS
Relating vapor pressure to vaporization
This graph shows how the vapor pressure of three liquids varies with temperature:
900-
B0-
700
600
500
400
300
- acetylacetone
piperidine
- pyridine
200
10.
03
100
iio
120
130
140
temperature, °C
Use the graph to answer the following questions:
Which liquid is the most volatile?
most volatile:
choose one
Which is the least volatile?
least volatile:
choose one
acetylacetone:
What is the normal boiling point of each liquid?
Note: your answer must be within 1°C of the exact
answer to be graded correct.
piperidine:
pyridine:
O more
Suppose a beaker of piperidine is put inside a sealed
tank containing piperidine gas at 103. degree C and
848. torr. After ten minutes, will there be more liquid
in the beaker, less liquid, or the same amount?
O less
O the same
Explanation
Check
2021 Mc
vapor pressure, tor
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning