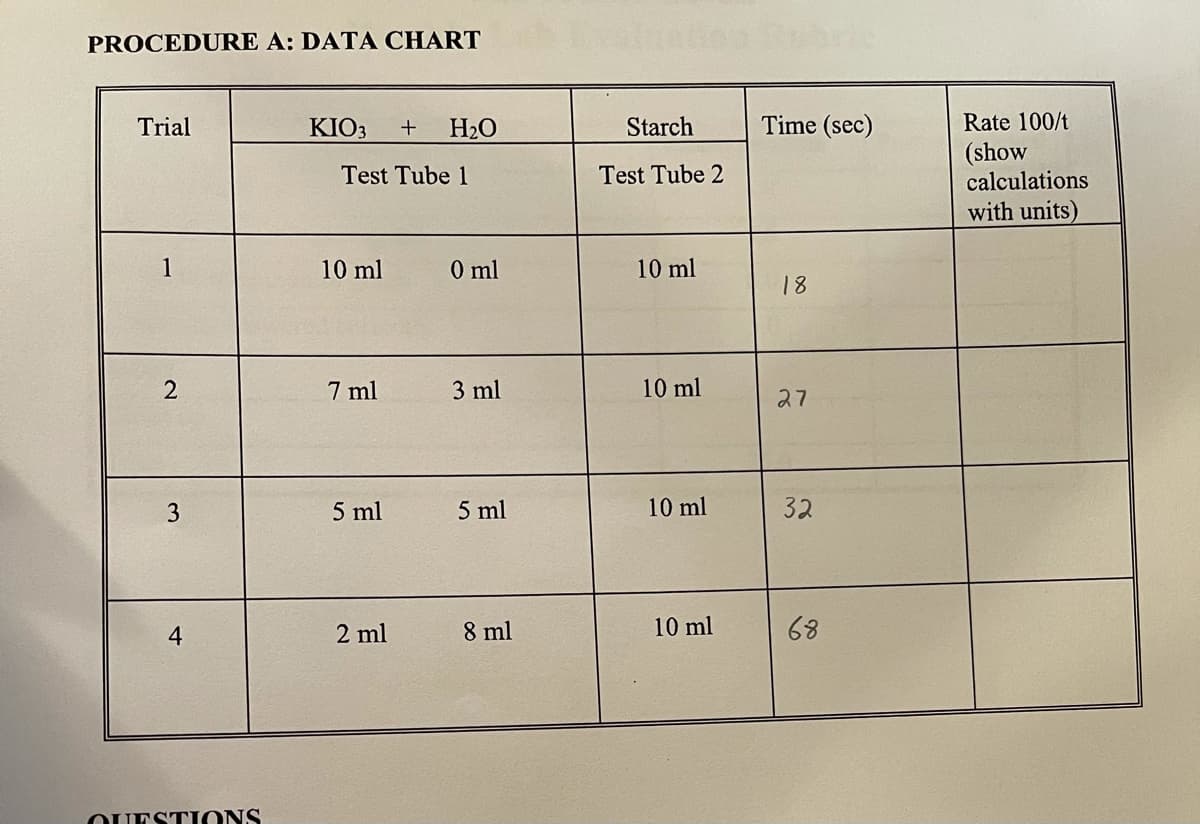
Trial KIO3 H2O Starch Time (sec) Rate 100/t (show calculations Test Tube 1 Test Tube 2 with units) 10 ml 0 ml 10 ml 18 7 ml 3 ml 10 ml 27 3 5 ml 5 ml 10 ml 32 2 ml 8 ml 10 ml 68
Q: Place 5 mL of starch solution in a test tubes. Heat the test tubes to boiling and add 1 mL of…
A:
Q: Weight of egg yolks: 17.8 g Crude cholesterol weight: 232 mg Recrystallized cholesterol weight: 168…
A: Amount of egg yolks = 17.8 gm = 17800 mg Amount of crude cholesterol = 232 mg Amount of…
Q: Agarose gel electrophoresis is relatively simple and straight forward to perform. Agarose is a…
A: two monomer units present in agarose are:
Q: Sequencing. Arrange the following procedures according to the CORRECT manner of preparing products.…
A: Turpuntine emulsion preparation AB AC A B C E
Q: Humulin R U-100 insulin 30Units = ________ml
A: Interpretation- To determine the volume of Humulin R U-100 insulin 30Units = ________ml -…
Q: polystysene (styrofoam cue) PerkinElmer Spectrum IR Version 10 6.0 Tuesday March 22, 2022 6:52 PM…
A: IR Spectroscopy tells us about the functional group present in the molecules.
Q: You are given a 1:1 (by weight) solid mixture of vitamin C and vitamin A, and you are going to use…
A: A 1:1 solid mixture of vitamin C and Vitamin A is to be separated from one another by liquid-liquid…
Q: Select which type of GC column is best suited to analyze a mixture containing o-xylene, m-xylene,…
A: The method of separating a compound which can be vaporized without the decomposition process and…
Q: Methylated samples hydrolysate samples can be analyzed using a gas chromatography. True False
A: Chromatography is a technique which is used for seperate the reaction mixture
Q: 4000 3000 2000 1S00 NAVENUMBERI -l which CA-F) corresponds with d IR ? H. A H2N. но но E F
A: IR spectroscopy is also known as vibrational spectroscopy. The molecules must have a dipole moment.…
Q: Materials and detailed methods: paragraph or two explain how the experiment was done Raw Data:…
A: Diels -Alder reaction takes place in reactions between conjugated diene and an alkene. In this…
Q: LEARNING TASK 4!
A: Given,
Q: 1.5%(mass:volume) carrageenan gel is comprised of 0.75g carrageenan dissolved in 50ml 1% KCl.You…
A: Given - Mass/volume percentage of carrageenan gel = 1.5% Correct mass of carrageenan = 0.75 g…
Q: Experiment Isolation and Saponification:HNMR of isolated Myristic Acid B00 200 100 10 ppm 5 ppm O…
A:
Q: skip
A: Question 36: Residual titration are performed because the reaction proceeds slowly, solubility of…
Q: 11. Paper Production (cont.) Another problem is that water-based inks are absorbed too heavily into…
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you.…
Q: which fare does the hyolide attlack CR)- CB scataly THE 25°,3omin
A: BH3 along with THF works as a reducing agent and reduces the compound to alcohol when further…
Q: ka = 5.70 x 10^-10 d0.0120 M in NaOH and 0.630 M in NH3. pH = | 12.25 An error has been detected in…
A: Concentration of NaOH = 0.0120 M Concentration of [OH- ] = 0.0120 M Concentration of NH3 = 0.630…
Q: Choice 141 - 0-12 snucoon 564 + 10-1 jinucieon 137 - 10-2 sinucleon 548 - 10- Jinucieon
A: According to the question, we need to determine the nuclear binding energy of potassium-40. Given,…
Q: Tyrosine gave a positive reaction to these tests except l. Hopkins-Cole |I. Biuret I.Sakaguchi IV.…
A: Hopkins Cole test - to detect tryptophan Biuret test - for present of peptide bond and…
Q: A sterile ophthalmic suspension contains 0.5% timolol maleate (m.w. 433). How many milligrams of the…
A: We have 0.5% w/v opthalmic solution. Solute here is timolol maleate. Mol mass of timolol maleate =…
Q: b Success Confirmation of Questio X C Circle every active position and i = с ONDERRIGTOETS 1…
A:
Q: In a mixture of the five proteins listed below, which should elute second in size- exclusion (gel-…
A: Size Exclusion Chromatography : It is a chromatographic method in which the components of the…
Q: A + OCH3 Heat ОСНЗ inder Imeeliate heart proderet & Gras Y Z
A: Given reaction is a example of (4n+2)π cyclo addition reaction. It is also called as diels - alder…
Q: он H H To -
A: The given reaction is ethyne to 2-methylpentan-2-ol.
Q: Show the multistep synthesis
A:
Q: What is meant by misorientation?
A: Misorientation has to be defined.
Q: what are possible sources of error in a soap making experiment? (non-human error) I want clear…
A: Soap making experiment involves the process of saponification in which the reaction of triglycerides…
Q: 1. Place 5 mL of starch solution in each of 2 test tubes. 2. Heat the test tubes to boiling and add…
A:
Q: How many milligrams of piroxicam is required to make 5 solid ounces of 1:270 (w/w) piroxicam gel?
A:
Q: Hey, could you do q24 aswell?
A: The two reactant molecules are - carboxylic acid and an alcohol. H2SO4 acts as the catalyst whereas…
Q: HO and ?
A: Alcohol can be defined as an organic compound in which carbon atom is attached to hydroxyl group.
Q: Please answer only 4 and 5 Which us polyurethanes and epoxyresins
A: (4) Polyurethanes - polyurethanes are prepared by Reaction between polyol (more than one hydroxyl…
Q: CI t-BUOK major product: Edit minor product: Edit
A:
Q: Match the following terms to the following situations. * Gross Random Systematic Defect Аccuracy…
A: In determiningthe angle ofrepose, Justinforgot to mixthe rock saltand talcproperly. gross error…
Q: MW (mg/mmol) Density (mg/uL) Amount used vanillin 152.15 XXXXX 400.0 mg acetic anhydride 102.09 1.08…
A: Number of millimoles=Amount/MW Amount of acetic anhydride=500 mg (since, 1ul=1mg) MW of acetic…
Q: ml Diluted Undiluted Diluted to mL Water Absorbance Unknown Conc. (M) Conc. (M) 0.5 4.5 0.4IS 1.25…
A: Given are the volumes of unknown sample which are diluted with given varying volume of water. The…
Q: What are annulenes ?
A: The compounds containing carbon and hydrogen only are termed as hydrocarbons. Hydrocarbons are…
Q: 1. You have determined the mp of pure Urea and pure Cinnamic acid. Here is your actual data: Urea:…
A: For drawing the plot we have to calculate average melting point for a particular % composition.
Q: Trial vol. of starch sol'n vol. of vol. of NazS203 diluted vol. of diluted vol. of K2S208 diluted…
A: It is required to calculate the concentration of the given compounds. The final volume of the…
Q: Determine the dilution factor for your diluted unknown U1 based on how you prepared the solutions in…
A: Using a concentrated solution of a given compound, different concentration of the same solution can…
Q: HO- 1) DMSO, (COCI) 2) Et,N Edit
A: The given reaction is oxidation of primary alcohol to an aldehyde by using swern reaction. The…
Q: #1. Part A. in a drug discovery program, a hit compound________. A. Shows activity in an animal…
A: In drug discovery program, a hit compound shows the required type of activity in the screening…
Q: answers
A: a. 0n1 + 5B12 -----> 5B13 addition of neutral neutron on boron there will be no change on atomic…
Q: You were assigned to determine the concentration of an unknown starch solution. [This unknown is…
A: Calculation of concentration (C = A/(L x Ɛ))The Lambert-Beer law, which forms the physical basis…
Q: Relative Intensity 100 60 40 8 / Find out the functional group 10 20 50 40 50 60 70 1860 my 2000 Wer
A:
Q: Identify the compound that gives the mass spectrum and infrared spectrum shown here.
A: EXPLANATION: To calculate the molecular formula of the compound, the following steps to be followed:…
Q: Show the step-by-step process. Do not use shortcut methods. Make it as detailed as it can be.…
A: Chiral centre is that carbon which is attached to the four different groups. If any one group is…
Q: Create a Schematic Diagram Delivering an aliquot and dilution 1. Using a serological pipette,…
A: We need to draw a schematic diagram for the mentioned procedure.
Step by step
Solved in 2 steps

- 1. Find HCl Mol weight 2. Find NH3 Mol. weight 3. HCI Diff. Distance: 10 4. NH3Diff. Distance: 60 5. Find rate ration (exp.) : Rate ration =Distance A/Distance B 6. Find rate ration (theor.) = MWb/MWaPLS HELP VERY URGENT Q5Trial Conc. A (M) Conc. B (M) Rate 1 0.706 0.771 6829.54 2 0.706 3.855 34147.702 3 2.118 0.771 6829.54 4 0.647 0.407 ? Given the information in the table above provide the rate for Trial 4Write all answers in standard form rounded to 3 decimal places " Order for A Order for B Overall Order The rate for Trial 4 Round to nearest whole number
- Initial temp: 19.06 C trial 1: 0.5 M concentration trial 2 : 1.0 M concentration 200 mL of HCl and 200 mL of NaOH are combined in an insulated container. HCl = 0.5 mol/ml x 200 x 10^-3 ml = 0.1 mol NaOH = 0.5 mol/ml x 200 x 10^-3 ml= 0.1 mol H+(aq) + OH-(aq) - - -> H2O (l) ∆H H++ = 0, ∆H OH−− = -230 kJ/mol, ∆H H2O = -286 kJ/mol ∆H reactants = ∆H H++ + ∆H OH−− = -230 + 0 = -230 kJ/mol ∆H reactants = -286 kJ/mol - ( -230 kJ/mol) = -56 kJ/mol For each trial, calculate how much energy is released during this reaction.urgent pls I will rate right away no need of explanationy= mx+ b Slope () = -4E-09 y-intercept () = 0.4021 and OD= 0.540 , Calculate the concentration (x)
- Which of the following would result in an increase in the calculated ngas 10, assuming the rest of the parameters are held constant? I. Inserting 23.0 mg instead of the prescribed 15.0mg of folded Mg ribbon into the eudiometer. II. Performing the experiment in a different laboratory with a cooler temperature. III. A shorter equilibration time prior to volume measurement. IV. Acid used was 12.0 M HNO3 instead of 6.0 M HCI.You are trying to come up with a drug to inhibit the activity of an enzyme thought to have a role in liver disease. In the laboratory the enzyme was shown to have a Km of 1.0 x 10-6 M and Vmax of 0.1 micromoles/min.mg measured at room temperature. You developed an uncompetitive inhibitor. In the presence of 5.0 x 10-5 M inhibitor, the apparent Vmax was determined to be 0.02 micromoles/min.mg. What is the Ki of the inhibitor?Full accurate details Mechanisms need steps by steps. Dont answer recent u people answer not good. Need new computational method etc mechanism steps by steps
- Faat pls i will give u like for sure solve this question correctly in 5 min plsExtraction of Caffeine Post-lab 5. On the report sheet, you calculated your percent recovery of caffeine. a. If your percent was less than 90%, give at least one reason why it was less. b. A group of students have their percent yield more than 100%, give at least one reasonwhy it was more than 100%. Do NOT simply say “human error.” Be specific about the typesof errors (human or otherwise) that may have occurred, leading to a recovery more than eorequal to 100% (Is this possible Why?) 6. Based on what you observed during this experiment, is dichloromethane denser OR lessdense than water? Explain your reasoning. 7. Based on what happened in this experiment, is caffeine more soluble in water OR moresoluble in dichloromethane? Explain your reasoning.HELP !!!!!!!!! PLEASE HELP ME ON THE DARK BULLET POINT TABLE CALCULATION. BE SURE UPLOAD THE GRAPH. make sure Answer all of them Experiment 1 Exact concentration of H2O2 stock solution (M) 1.014 Temperature of water bath (°C) 24 Experiment 1: Volume of O2 vs time for decomposition of H2O2 Time (min) Volume of O2 (mL)2.0 0.00 4 1.22 6 2.41 8 3.64 10 4.93 12 6.34 14 7.78 16 9.49 18 10.99 20 12.35 22 14.64 24 16.31 26 19.16 28 20.97 30 24.52 32 26.03 34 28.99 36 32.30 38 35.17 40 38.83 42 44.83 44 50.50 46 58.26 48 63.46 Experiment 2: Volume of O2 vs time for decomposition of H2O2 Time (min) Volume of O2 (mL)2.0 0.00 4 0.63 6 1.31 8 1.96 10 2.42 12 2.93 14 3.56 . Experiment 3: Volume of O2 vs time for decomposition of H2O2 Time (min) Volume of O2 (mL)2.0 0.00 4 0.45 6 0.88 8 1.44 10 2.29 12 2.92 14 3.51 Calculations For all three experiments plot the volume of O2 evolved in…



