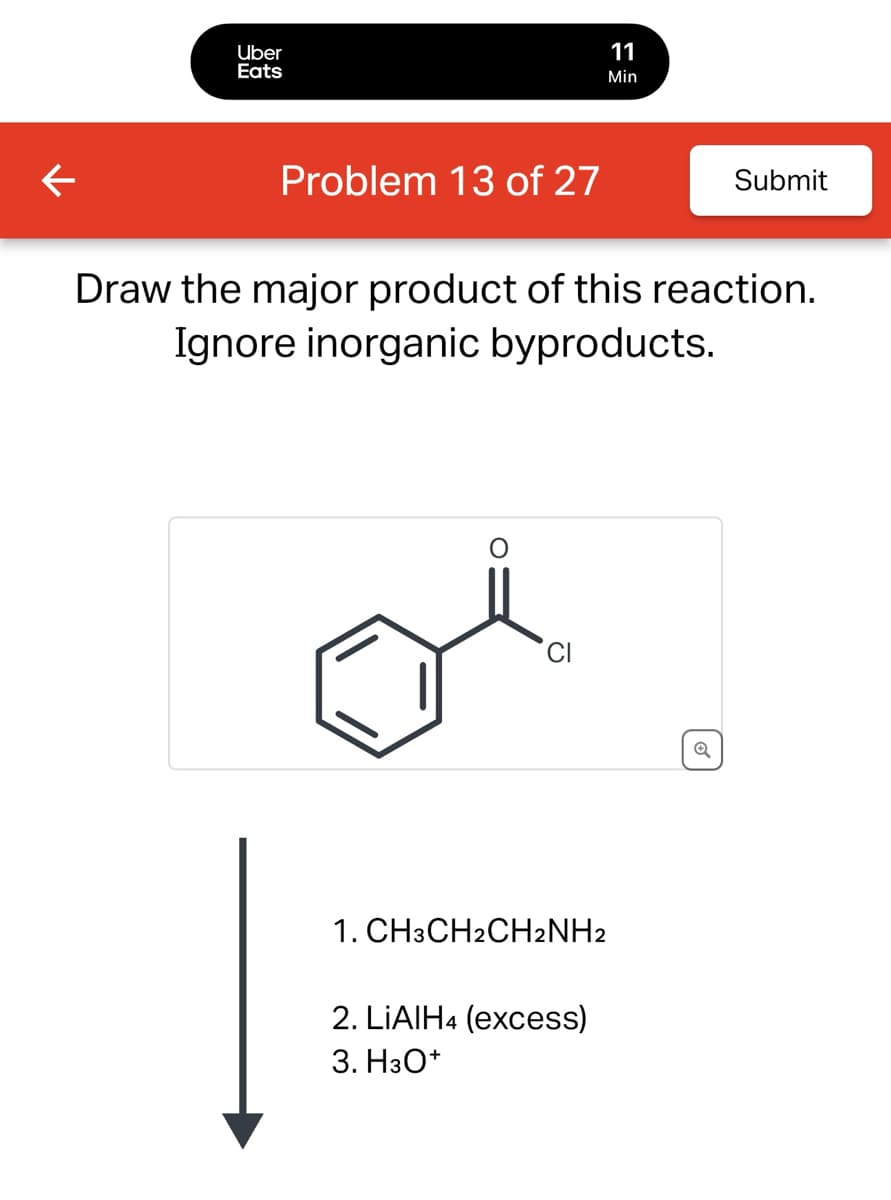
← Uber Eats 11 Min Problem 13 of 27 Submit Draw the major product of this reaction. Ignore inorganic byproducts. 1. CH3CH2CH2NH2 2. LiAlH4 (excess) 3. H3O+ Q
Q: None
A: Let us discuss important terms first related to this problem: Sensible Heat - This refers to the…
Q: ← Problem 17 of 27 Submit Draw the monoalkylation product of an amine reacting with methyl iodide.…
A: Synonyms for N-Methylhexan-1-amineN-Methyl-1-hexanamineN-MethylhexylamineN-Hexylmethylamine
Q: For Co(CO)63+, give both the VBT and LFT diagrams, with electrons included. For the latter, include…
A: The objective of the question is to give and for .Valence Bond Theory () and Ligand Field Theory…
Q: Nitrous acid, HNO2, is a weak acid. What is the reaction of nitrous acid in water? HNO2 (aq) + H2O…
A: The question is asking for the correct reaction of nitrous acid (HNO2) in water. Nitrous acid is a…
Q: Give introduction step and proper explanation please will upvote. Hand written solution is not…
A: Step 1:To find the percent yield of the reaction, we first need to calculate the theoretical yield,…
Q: Chemistry
A: An alkyl halide is a compound containing a halogen atom bonded to a saturated carbon atom. a)…
Q: in addition also is it aromatic non aromatic or anti aromatic
A: Step 1:• For the following electrocyclic ring-opening reaction 1. Choose the correct…
Q: What is the IUPAC name for the compound shown in the figure? Br
A: To name the compound (which is an alkane), follow the steps/rules below. Step 1: Name the parent…
Q: Use the References to access important values if needed for this question. The pOH of an aqueous…
A: Step 1: Step 2: Step 3: Step 4:
Q: Let operator Ĥ. Let be one of the normalized eigenfunctions of a Hamiltonian be an approximation of…
A: The equation given is:Where: is one of the normalized eigenfunctions of the Hamiltonian operator .…
Q: Use retrosynthesis. DO NOT upload images of handwritten or typed solutions.
A:
Q: Calculate the potential at the equivalence point for titration of 0.100MFe^2+ with 0.100M…
A: The objective of the question is to calculate the potential at the equivalence point.
Q: Incorrect Your answer is wrong. In addition to checking your math, check that you used the right…
A: Step 1:Step 2:
Q: Question 1 In carbon NMR, the carbon atom of the carbonyl group in aldehydes and ketones has a…
A: In carbon NMR spectroscopy, the chemical shift refers to the position of a carbon atom's resonance…
Q: Super hero expert Hand written solution is not allowed
A:
Q: Ethylene oxide, C2H4O, may be made by passing ethylene, C2H4, and air over a catalyst at 2500C. When…
A: The given reaction is the reaction between ethylene, water and CO to form ethylene glycol. The…
Q: i dontt want hand writting image
A: Step 1: Predict arrow movement Explanation: An arrow always depicts a region of high electron…
Q: Home - Profession... A mixture of nitric and sulfuric acids generates the nitroniium ion, *NO2…
A:
Q: Determine the density of CO2 at 745 mmHg and 65 degrees C?
A: The objective of this question is to calculate the density of carbon dioxide (CO2) under specific…
Q: What spectral features would you expect to see in MS and IR to determine whether the synthesis was…
A: The objective of the question is to find the spectral features expected to be seen in MS and IR to…
Q: What is the outcome of the reaction shown in the box? - Assume addition- elimination mechanism. A) F…
A: Step 1:Grignard reagent (RMgX) give nucleophilic aromatic substitution reaction.A nucleophile…
Q: What is the pH of the solution that results when the following solutions are combined? 0.020 L of…
A: Thank you,Please rate my response.
Q: and 18 [References] 1 pt 2 pts 1 pt 2 pts 1 pt 2 pts Strontium hydroxide, Sr(OH)2, dissolves in…
A: Step 1:The dissociation equation for strontium hydroxide (Sr(OH)₂) dissolving in water is: Sr(OH)₂…
Q: == Electrochemistry Designing a galvanic cell from two half-reactions A chemist designs a galvanic…
A: The objective of the question is to understand the reactions happening at the cathode and anode in a…
Q: 22. Elements that have metallic and non-metallic properties are called a. metalloids…
A: The question is asking to identify the term for elements that exhibit both metallic and non-metallic…
Q: A solution has a pOH of 7.40. What is the hydroxide ion concentration?
A: The objective of this question is to find the hydroxide ion concentration of a solution given its…
Q: give me a synthetic route for the following
A: First, react benzene with tert-butylalaninoyl chloride to achieve a substituted benzene with a meta…
Q: Below is the resolved absorption spectrum of HBr for the vibrational transition v = 0 → V = 1;…
A: The objective of the question is to determine the effective spring constant and bond length for HBr,…
Q: 10. Provide a Fischer projection for the sugar shown below. At which position(s) is the…
A:
Q: Construct a two-step synthesis of (±) leucine by dragging the appropriate formulas into the bins.…
A: The suitable molecules and reagents are used in the given transformation are as follows:Detailed…
Q: Describe how you could achieve a higher degree of separation for the bands of food dyes on a TLC…
A: The objective of the question is to understand how to achieve a higher degree of separation for the…
Q: NaOH Ph 'Ph Δ Br
A: The objective of the question is to determine the chemical reaction that occurs when sodium…
Q: What is Ksp for calcium hydroxide if the concentration of OH in a saturated calcium hydroxide…
A: The objective of this question is to calculate the solubility product constant (Ksp) for calcium…
Q: 1. Provide a mechanism for the following reaction. (You should show all intermediates and curved…
A: Step 1: Step 2: Step 3: Step 4:
Q: What is the Kyoto Protocol?
A: The Kyoto Protocol is an international treaty that was adopted in Kyoto, Japan, in December 1997. It…
Q: A mixture of nitric and sulfuric acids generates the nitronium ion, + NO2 (mechanism provided, which…
A:
Q: Uncatalyzed Reactio Time (s) Energy (kJ/mol) 0 40 1 40 2 40 3 40.1 4 40.2 5 40.3 60 6 40.4 7 40.5 68…
A:
Q: Please select the appropriate reagent to complete the following transformation. Ph3P-CH2 MgBrCH3…
A: Ketones react with phosphorus ylides in a reaction called the Wittig reaction. The Wittig reaction…
Q: × Incorrect. Identify the major product(s) for the following reaction. Select all that apply. If the…
A: In bromination of alkyl in presence of hv, bromine atom will atach to secondary carbon as at…
Q: 4. Predict the products for the following enolate reactions. a) LDA b) MeBr a) 0.95 equiv LDA RT b)…
A:
Q: 5. How many grams of CuSO4 * 5 H₂O are needed to prepare 100 ml of a 0.10M solution? (This is like…
A: The target volume to prepare is 100 mL and the concentration is 0.10M CuSO4. So we have…
Q: Which of the following is a correct net ionic equation for the reaction of a metal and an acid? ○ Mg…
A: The objective of the question is to identify the correct net ionic equation for the reaction of a…
Q: Draw the MO diagram for NiMe2(PMe3)3. Be sure to properly label everything!
A: has a trigonal bipyramidal shape with hybridasation
Q: O Chemical Reactions Using molarity to find solute moles and solution volume 3/5 A chemist adds…
A: Step 1: The millimole of NaCl in 365 ml of 2.07 M solution is calculated as
Q: Is the following compound saturated or unsaturated: CH3CH2CH2CH2CH3 This compound is…
A: The objective of the question is to determine whether the given compound CH3CH2CH2CH2CH3 is…
Q: The half-life of gallium-67 is 78.2 hours. If you begin with 44.1 mg of this isotope, what mass…
A: Step 1: Step 2: Step 3: Step 4:
Q: 5) What series of synthetic steps could be used to carry out the transformation shown below? Draw…
A: Question 5(a)Detailed explanation: Key references:
Q: 91 D. Calculate the moles of NaOH used in each reaction. In Reactions 1 and 2, this can be found…
A: The objective of the question is to calculate the moles of NaOH used in each reaction and the moles…
Q: To preview image Click Here + Assuming the pi-systems in the following structures are flat, indicate…
A: thanks
Q: Predict the major product(s) for the following reactions. :0: A. Cl₂/AICI 3 B. NO2 1. H₂/Pd/C 2.…
A: Step 1: Step 2: Step 3: Step 4:
Step by step
Solved in 2 steps with 3 images

- Predict the product of the following reaction and classify the reaction. Pb(NO3)2+FeSO4--->PbSO4+________Fill in necessary products reactants or reagants of these reactions. Please note the existence of enantionmers in some cases.Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acid
- Draw themajor product of this reaction. Ignore inorganic byproducts. PPC, CH2Cl2Which of the following reactions result in a positive ∆ Ssys? A. Pb(NO3)2 (aq) +KCl (aq) ---> PbCl2 (s)+ KNO3 (aq) B. HCl (g) + H2O (l) ---> HCl (aq) C. H2 (g) + I2 (g) ---> 2HI (g) D. 2H2O (g) ---> 2H2 (g) + O2 (g) E. H2O (g) ---> H2O (l)14. For the reaction shown, select the expected major organic product. (see attached screenshot) A. I B. II C. III D. IV E. V
- Prove that the Keq for the reaction between Pb2+ and HF is 12.4. (Ksp, PbF2 = 6.76 x 10-4 and dissociation constant of HF is 3.7 x 10-8)The methoxy group is reported as having σpara and σmeta values that differ in sign (σmeta =+0.12; σpara = –0.27). What specific experiments are performed to determine these s values?Explain why the σpara and σmeta values are of different sign and what those signs indicate.Which of these is the rate-determining step in the nitration of benzene? A. Protonation of HNO3 B. Loss of H2O by HNO3 C. Formation of sigma complex D. Loss of H+ by sigma complex E. Protonation of H2SO4
- The following reaction has a ΔSsystem < 0 O2(g) → 2O(g) T or F can you explain why this is false?9. Product of the reaction of nitrobenzene with sulfuric acid in the presence of oleum at 60 °C:a) o-nitro benzenesulfonic acidb) m-nitro benzenesulfonic acidc) p-nitro benzenesulfonic acidd) None of the above 10. Due to their ability to move through ducts or pipes, they are generally called fluids:a) Solidsb) Solid mixturesc) Gasesd) Liquids and gases6. Identify the best reagent(s) for this reaction. (see attached screenshot). a. H2SO4, HgSO4, H2O b. 1. Disiamylborane, 2. HO–, H2O2 c. K2Cr2O7, H+ d. NaOCl e. H2, Pd

