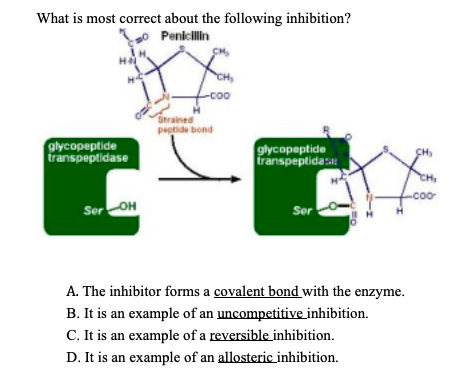
What is most correct about the following inhibition? Penicillin
Q: ATP stock (50 µM): Make pre-dilutions with 20, 15, 10, 5, 2,5 and 1,25 µM ATP (1000 μL of each)…
A: Molarity is a way of representing the concentration of a solution. Molarity is the number of moles…
Q: a) Draw a non-covalent interaction between a water molecule and the charge on a phosphorylated…
A: Unlike covalent interactions where there is sharing of electrons between two atoms, in non-covalent…
Q: 7. H 1 H-C -OH H-C-OH 1 H -C 8. H 9. НО I 1 H J=D 0=)) с HIV-H OH HH -C-C-H I OH OH F-DIE-DI - CH…
A: 7. Chemical formula for structure number 7. = C4H8O4 or C4(H2O)4. Yes, this structure is a…
Q: Q4.3- For each of the five major protein types in eukaryotic cells 1) provide a specific example of…
A: Proteins can be classified into different types based on their function, chemical nature,…
Q: 1. What is the sequence of the heptapeptide produced upon treatment with trypsin? Fill each blank…
A: Specific enzymes have specific cleavage point in amino acids. Chymotrypsin is serine…
Q: 1. What two enzymes in glycolysis are regulated by high ATP concentrations?
A: INTRODUCTION : Glycolysis : It is a metabolic pathway in which glucose is breaken down into pyruvate…
Q: 2. The attached figure is a titration curve for the amino acid glycine. At which pH is glycine…
A: Amino acids are biomolecules that have an amino group, a carboxyl group and a side group that is…
Q: Please help with 2a) 2a) There are two different DNA polymerase enzymes, DNA Polymerase I and DNA…
A: Replication is the process of duplication of two strands of a double stranded DNA. In bacteria, the…
Q: Which of the following would you expect to happen if amino acids 579 to 582 (YIYV) in the PDGF-Beta…
A: PGDF beta-receptor contain multiple domains that activate the various pathways inside the cell. The…
Q: H K Br Br H trans-2,3-dibromo-2-butene cis-1,2-dibromoethene Otrans-1,2-dibromoethene…
A: The structure given the question represents- Answer- trans - 1,2- dibromoethene
Q: b) It's said that secondary structures form because of intra- and intermolecular hydrogen bonding…
A: A protein's function depends on its structure. There are four levels of protein structure: primary,…
Q: Describe how a protein’s structure determines its function. What determines a protein’s structure?
A: Proteins have four levels of conformations known as primary, secondary, tertiary, and quaternary. In…
Q: ________ is a regulatory mechanism in which the end product of a metabolic pathway inhibits an…
A: Enzymes are biological catalysts. The enzymes, whose ability to catalyze a reaction depends upon…
Q: 1. Under what circumstances in the cell would the entire pentose phosphate pathway be carried out…
A: The pentose phosphate pathway is also called HMP shunt pathway. It branches from glucose 6-phosphate…
Q: Describe the signal transduction pathway for Two or Three of the following: a) ß-adrenergic…
A: Beta adrenergic receptor is a G-protein-coupled receptor communicating through the Gs alpha…
Q: Formation of a peptide bond is a. dehydration reaction b. reducing reaction c.…
A: Proteins are composed of twenty naturally occurring amino acid. The amino acids in a protein are…
Q: Please answer fast What types of interactions likely promote the dimerization of a protein?
A: Proteins are polymers of amino acids that are joined together through peptide bonds. A peptide is a…
Q: 6. What term is used to describe reactions that generate metabolic intermediates of a pathway? A.…
A: The reactions that generate metabolic intermediates of a pathway is termed as - Answer- B.…
Q: For the amino acid tryptophan: Draw its complete protonic equilibria. Indicate the net charge of…
A: The proteins are composed of twenty naturally occurring amino acids. The net charge on an amino acid…
Q: Experiment 1.2 - Reaction over time 2. Using the standard curve in 1.1, calculate the amount of…
A: The given graph is representing the relation between the absorbance of Product and the concentration…
Q: b) It's said that secondary structures form because of intra- and intermolecular hydrogen bonding…
A: Examples of secondary structures are alpha-helices and beta-sheets. The primary structure of a…
Q: Amino acid sequence in a protein that can form N-glycosylation is called a glycosylation sequon.…
A: N-linked glycosylation is a post-translational modification process that occurs in the endoplasmic…
Q: A. Will decreasing the amount of energy needed to make an exergonic reaction occur cause the…
A: The thermodynamics of a chemical reaction focuses on the energetics of the products and the…
Q: Although phosphoesters are generally not energetic enough to donate the phosphate to make a high…
A: For an energetically favorable reaction, the change in free energy will be negative, and for an…
Q: Question 9 Which of the following fatty acid does have greater solubility in wat O 16:0 O 18:0 O…
A: Fatty acids are a class of lipids that are nonpolar and are insoluble in water. The naturally…
Q: Which metabolic event is not in the mitochondria? Oxidation of fatty acids ETC…
A: Glycolysis is a the pathway in which glucose is broken down into two three-carbon compounds and…
Q: what does this picture indicate? what is difference bewteen A, B, and C ?
A: INTRODUCTION : Myoglobin - It is an iron and oxygen binding protein found in the skeletal &…
Q: 1a Briefly describe or explain what the term "supercoiling" means in the context of DNA structure.…
A: Supercoiling means the coiling of the coil. Cellular DNA is extremely compacted and implies a high…
Q: In the peptide, SWEETLY, bond via its side chain. can still form a standard peptide bond while can…
A: A standard peptide bond is formed as a result of a dehydration ( also called condensation ) reaction…
Q: Which of the following experiments can be used to determine the localization of a protein in living…
A: Confocal microscopy is an optical imaging technique used for increasing optical resolution and…
Q: What does it mean if a compound is said to have “high phosphoryl transfer potential”?
A: ATP is known as the energy currency of the cell. ATPs are required by the cell for various reactions…
Q: Given the following reaction and equation for the initial velocity of the reaction: k₁ k3 E+SES E +…
A: We are given two equations for initial velocity (V). V=kcat [ES] -------(eq1) andV=k3 [ES]…
Q: ATP is deemed the Universal currency for Cell, why do you think now it is the Fact
A: Energy-rich compounds are chemicals with specific structural characteristics that cause an energy…
Q: 8. Which of the following members of the ETC does not pump any protons into the intermembrane space?…
A: Electron Transport Chain (ETC). This component of aerobic respiration and the only component of…
Q: What is the product of the oxidation of this fatty acid?
A: The aerobic fatty acid oxidation mechanism in the mitochondria converts a fatty acid into acetyl-CoA…
Q: Triacylglycerols Store Energy Q4.3 - Describe the three sources of triacylglycerols in humans and…
A: Introduction: Triacylglycerols are also known as neutral fats or triacylglycerides, which are esters…
Q: Select the correct response:
A: Tyrosine gives rise to catecholamines which includes dopamine, norepinephrine, and epinephrine.
Q: What is the product of the beta oxidation of this fatty acid?
A: The given fatty acid is caproic acid. Beta-oxidation is the oxidation of fatty acid on the…
Q: a) What is the Kd you measure for this interaction?
A: INTRODUCTION : Actin - Actin is a family of globular multi-functional proteins which form…
Q: What differences would you expect in the structure of a membrane bound protein versus a cytosolic…
A: A protein molecule must fold into its appropriate three dimensional structure in order to become…
Q: 29. What is the action of the Na/K pump? A. It is bidirectional for both ions B. It actively…
A: Introduction Plasma membrane or cell membrane is a outermost membrane in animal cell. It protect the…
Q: if you have a dialysis tube filled with 10 mL of 20% sucrose solution in a beaker with 100 mL of…
A: Osmosis is the movement of water across the semipermeable membrane driven by the difference in…
Q: During anaerobic conditions, lactate travels from the muscle to the liver via the bloodstream. What…
A: The Tricarboxylic acid(TCA) cycle is inactive under anaerobic conditions, thereby the process of…
Q: The side chain of cysteine contains: OA) a hydroxyl group OB) an amine group C) a carboxyl group OD)…
A: Proteins are composed of amino acids, which are bound together by peptide linkage. Amino acids…
Q: DNA: Explain nick translation and strand displacement
A:
Q: a. Provide names for both metabolites. b. What kind of reaction occurs here? c. What enzyme…
A: Biochemical reactions are catalyzed by enzymes in which bio-metabolites are transformed into another…
Q: Problem 1: Calculate the hydrolysis of ATP at pH 7 at 37°C under steady state conditions in which…
A: In a general reaction such as: aA + bB ⇌ cC + dD At equilibrium (steady state), the concentration of…
Q: What is the product of the oxidation of this fatty acid?
A: Fatty acids are carboxylic acids with a hydrocarbon chain ranging from 4 carbon to 36 carbons.…
Q: Question 15 of 25 Which of the following is true for the acid-base properties of amino acids? Select…
A: The proteins are made of 20 naturally occurring amino acids. The net charge on the side chain of the…
Q: The degradation of nutrients for energy is also known as oxidative metabolism. True False
A: Metabolism is the total of all chemical transformation that takes place in a living cell. One…
Give correct answer
Step by step
Solved in 2 steps with 1 images

- Using the ActiveModel for aldose reductase, describe the structure of the TIM barrel motif and the structure and location of the active site.A biochemist wants to determine the effect of inhibitor A to enzyme B which catlyzes the conversion of C to D. The effect of A to the rate of formation of D is shown below: 1. The Km (report to the nearest whole number) for the enzyme-catalyzed reaction in the absence of inhibitor A is _____ mM. 2. The Km for the enzyme catalyzed reaction in the presence of inhibitor A is ____mM. 3. The Vmax for the enzyme catalyzed reaction in the absence of inhibitor A is ____ mM/min 4. The Vmax for the enzyme catalyzed reaction in the presence of inhibitor A is ____mM/min 5. Inhibitor A is a/an ________ inhibitor of enzyme BAn enzyme catalyzes the reaction M↽−−⇀N M ↽ − − ⇀ N . The enzyme is present at a concentration of 2.0 nM 2.0 nM , and the ?max V max is 2.1 μM s−1 2.1 μM s − 1 . The ?m K m for substrate M M is 6.3 μM 6.3 μM . Calculate ?cat k cat .What values of ?max and ?m would be observed in the presence of sufficient amounts of an uncompetitive inhibitor to generate an α′ of 1.9?
- An enzyme catalyzes the reaction M ßàN. The enzyme is present at a concentration of 1 nM, and the Vmax is 2 M s-1. The Km for substrate M is 4 μM. a)Calculate kcat. b)What values of Vmax and Km would be observed in the presence of sufficient amounts of an uncompetitive inhibitor to generate an α’ of 2.0?A competitive inhibitor interacts with the free enzyme to form an enzyme•inhibitor complex(E•I). This equilibrium reaction can be described as follows:E + I ⇌ EIModify the simplified kinetic scheme for the reaction E + S ⇌ E + P to include this equilibriumexpressionA mixed inhibitor of an enzyme (sometimes called a mixed non-competitive inhibitor) can decrease the rate of a reaction by any of the following EXCEPT by: a) binding to a site other than the active site of the enzyme. b) binding to the active site of the enzyme, preventing substrate binding. c) decreasing kcat. d) Increasing KM.
- Studies at diff erent pH’s show that an enzyme has two catalytically important residues whose pKs are ∼4 and ∼10. Chemical modifi cation experiments indicate that a Glu and a Lys residue are essential for activity. Match the residues to their pKs and explain whether they are likely to act as acid or base catalysts.The Lineweaver-Burk plot and other linear transformation of the Michaelis-Menten curve of kinetics are valuable for _____. A. determination of Vmax. B. determination of Km. C. determination of kcat. D. determination of types of enzyme inhibition. E. All of the above An inhibitor binds to a site other than the active site of the enzyme. Which statement below correlates with this observation? A. The inhibition must be irreversible. B. It must be a competitive inhibitor. C. It could be noncompetitive or uncompetitive inhibition. D. It could be irreversible, competitive, noncompetitive or uncompetitive. The data do not relate to the type of inhibition.You are studying the kinetics of a novel competitive inhibitor of ATP-citrate lyase that can be used to prevent obesity. What is likely true about this inhibitor? (A) This inhibitor lowers the Vmax of ATP-citrate lyase. (B) The inhibitor likely looks structurally similar to acetyl CoA. (C) The inhibitor binds to an allosteric region on ATP-citrate lyase and prevents catalysis. (D) You can distinguish between an uninhibited and inhibited enzyme by comparing the x-intercepts on a Lineweaver-Burk plot. (E) This inhibitor lowers the KM of ATP-citrate lyase. Related to this question, you should also be able to: • explain the function of ATP-citrate lyase and why inhibition would prevent obesity • interpret Michaelis-Menten & Lineweaver-Burk plots related to this question • explain the differences between a competitive and non-competitive inhibitor
- An uncompetitive inhibitor interacts with the enzyme•substrate complex to form a ternarycomplex (ES•I). This equilibrium reaction can be described as follows:ES + I ⇌ ESIModify the simplified kinetic scheme you drew for E + S ⇌ E + P to include this equilibriumexpressionIt's a three part question based on the chart provided asking: a) Which of these enzymes has the weakest binding of substrate? (I chose fumarase since it has lowest Km but that was wrong so I don't understand) b) Which enzyme has the fastest conversion of ES? (I would assume this means highest Kcat value, so should be catalase) c) Which enzyme is most closely catalytically perfect? (I'm assuming this means highest Kcat/Km ratio, so I was thinking Crotonase, although I don't know what classifies an enzymes as 'catalytically perfect', please explain)The text discusses three forms of enzyme inhibition: uncompetitive inhibition, competitive inhibition, and irreversible inhibition.(a) Describe how an enzyme inhibitor of each type works.(b) What kinds of bonds are formed between an enzymeand each of these three kinds of inhibitors?



