Q: Using the radial relationship, indicate which crystal structure is predicted for TlCl. Yes the Ionic…
A: Given data in question - 1) Ionic radii of (Tl+) = 164 pm. 2) Ionic radii of Cl- = 181 pm. We…
Q: How do call the measurement of optical activity and the change in polarization orientation. O…
A: The measurement of optical activity and the change in polarization orientation is called by what has…
Q: The importance of optical isomerism in organisms .
A: Optical isomerism is very important in organisms because the specific isomer performs a specific…
Q: The equilibrium constant of the reaction in aqueous solution is K = 5.00.…
A: The reaction given is, => CH3Hg(glutathione)+ (aq) + Cysteine (aq) → CH3HG(cysteine)+ (aq) +…
Q: How is ligand bound to a protein? List the physical forces?
A: The forces through which one ion or molecule is bounded to another ion or molecule are termed as…
Q: What is the conduction band?
A: Band theory: One way for distinguishing between conductors (metal), insulators and semiconductors is…
Q: Draw the hybridization for the metal centre of the complex below. You need to determine the geometry…
A: Hybridization is the process of mixing up of atomic orbitals to give new orbitals, hybrid orbitals.…
Q: For any of the following that can exist as isomers, state thetype of isomerism and draw the…
A: a.
Q: Q2: What is the function of catalyst in coordination polymerization?
A:
Q: For any of the following that can exist as isomers, state thetype of isomerism and draw the…
A: (a)
Q: Calculate the isoelectric point of RID. pI =?
A:
Q: What do you mean by coordination complexes?
A: Coordination complexes refers to the addition compounds which do not lose their identity in aqueous…
Q: Define the following terms: Tyndall scattering: Polarization of Radiation:
A: We have to Define the following terms: Tyndall scattering: Polarization of Radiation
Q: is mesosphere solid or liquid?
A: Is mesosphere solid or liquid has to be given.
Q: Which of the following pairs have the same value of magnetic moment a-Nit2,Ti b- Fe,Sc c- Fe,Mn+2 d-…
A:
Q: Describe what happens to the anions in a doublereplacementreaction.
A: What happens, to the anions in a double replacement reaction has to be given.
Q: Explain the concept of Racemic Mixtures ?
A: Racemic mixture is one which containing equal amounts of left right and left handed enantiomers of…
Q: Ca[Mo(CN)2(C2O4)(en)]2 coordination compound name
A:
Q: Predict the magnetic properties and determine the spin-only moment for the following: [Mn(H2O)6]2+…
A:
Q: true or false ? The flowing compound is the amino acid glycine that contains glycinate bidentate…
A: The glycinate ion H2NCH2CO2- is formed by deprotonation of the amino acid glycine can function as a…
Q: Draw diagrams showing the structures of and name all the stereoisomers of [Co(Cl)2(en)2]Cl.
A:
Q: Which of the following will exhibit cis-trans isomerism? Fe°: [Ar]4s?3d6 Cdº: [Kr]5s²4d10 Pt0:…
A: Option D; All of the above
Q: Which of the following will exhibit cis-trans isomerism? Fe°: [Ar]4s²3d6 Cdº: [Kr]5s²4d10 Ptº:…
A: A multiple choice question based on coordination complex, which is to be accomplished.
Q: Two coordination sphere isomers for MnCl3(CN)3
A:
Q: Copperhastwonaturallyoccurringisotopes,Cu-63hasanatomicmassof62.9296amuandan…
A: Isotopes of an atom have same number of protons and different number of neutrons in the nucleus,…
Q: How many total optical isomers would exist for the compound shown below?
A: We have to predict the number of optical isomers possible.
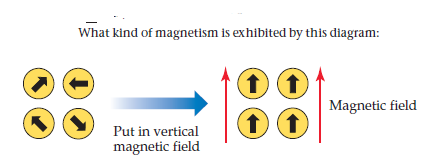
Q: 9. In the graphic below we have two types of materials X and Y: i) a & b refer to material "X" in…
A: Answer: Paramagnetic materials are weakly attracted by the external magnetic field and they…
Q: What can you determine about the structure of a coordination complex by measuring its conductance in…
A: The conductance of the coordination compound is used to predict the no. of ions present in the…
Q: What is the quantum number n of the HOMO orbital for the following dye:
A: HOMO is the highest occupied molecular orbital and LUMO is the lowest unoccupied molecular orbital.…
Q: For any of the following that can exist as isomers, state thetype of isomerism and draw the…
A: Geometrical isomerism or cis-trans isomerism: When two identical ligands are placed adjacent…
Q: How many electronic spectral bands for Ni^2+ and Co^2+ , one could be expected. Assign these…
A: The common oxidation states of cobalt are +2 and +3. The complexes are either octahedral or…
Q: Which of the following are chiral objects? Pick all that apply. A) A beachball B) A fiddler crab C)…
A: Chirality is defined when an object or compound doesn't have a plane of symmetry meaning when it is…
Q: Which type of substance is attracted by a magnetic field, a diamagnetic substance or a paramagnetic…
A: Paramagnetic compounds are compounds with unpaired electrons in their outermost shell. Diamagnetic…
Q: Name the following compound and create ionization, linkage and optical isomers: [Au(SCN)2(en)2]CI
A: We are authorised to solve only one question at a time. Please post rest of the questions…
Q: Which one of the following can form geometric isomers?
A: Isomerism : molecules with same molecular formula but differ in their properties is called…
Q: Magnets come in different shapes and sizes. O True O False
A: Magnets are defined as those materials in which the atoms of the material are aligned in such a way…
Q: junctional group
A:
Q: Which of the following can be used to differentiate between optical isomers? O heat O temperature O…
A: Optical isomers :- - Optical isomers are also known as enantiomers . - Two enantiomers are related…
Q: Please Indicate the coordination number for the iron surfur protein in the picture. A) 5 B) 6 C)…
A:
Q: Q5. Fill in the following table the correct values for F(CO) in the following complexes ((2055, 2090…
A:
Q: 5. Which coordination ion could be called chelate? (A) [Cr(NH)]- (B) [Fe(C;O4);]° (C) [AIF3- (D)…
A:
Q: Uses crystal field theory (CCT) to predict the number of unpaired electrons in the following…
A:
Q: Explain the relationship between chain symmetry and density of polymers.
A: Chain symmetry affects the polymer density as follows.
Q: In X-ray crystallography, the wavelength of an X-ray is ~ 1.5 Å. This is useful in determining a…
A: A question based on the X-ray crystallography, which is to be accomplished.
Q: Describe the bonding in PCl, without using d orbitals. As a hint, the geometry of PCI, is as shown.…
A: PCl5 is a geometrical molecule. It has a central atom Phosphorus (P) which is bonded to five…
Q: Which hybridization(s) is/are suitable to form a metal complex? (You can select more than one option…
A: Hybridization which are suitable for metal complex formation are sp, sp2, sp3 , dsp3 . Let's see by…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- is the field pattern for a solenoid the same as for a magnet except for the normalization factor?What would be the magnetic susceptibility of [Mn(CN)6]4- assuming the spin-only formula applies? (μ = (n(n + 2))0.5) a) 5.92 b) 0.0 c) 1.73 d) 4.90Mass spectrometry and X‑ray diffraction are common biochemical techniques for characterizing proteins. Classify each statement based on whether it applies to mass spectrometry, X‑ray diffraction, or both techniques.
- The absorbance values observed during the determination of the complex stoichiometry with the Job method using the spectroscopic method were plotted and the following correct equations were obtained. According to this, what is the n value of the compound formed by Fe + 3 and SCN- ions?1. Line equation: y = 2.6188x + 0.1608 2. Line equation: y = -2.5963x + 2.7203In optical analysis, what are the methods used to calculate the concentration ofa solution from the absorbance measurements of colorimeters?The absorbance values observed during the determination of the complex stoichiometry with the Job method using the spectroscopic method were plotted and the following correct equations were obtained. What is the n value of the compound formed by Fe + 3 and SCN- ions? 1. Line equation: y = 2.6188x + 0.16082. Line equation: y = -2.5963x + 2.7203
- Contrast non-complex splitting and complex splittingPlot the following Transmittance and absorbable curves for FCF brilliant blue dissolved in waterThe absorbance values observed during the determination of the complex stoichiometry with the Job method using the spectroscopic method were plotted and the following correct equations were obtained. What is the n value of the compound formed by Fe + 3 and SCN- ions? 1. Line equation: y = 2.6188x + 0.1608 2. Line equation: y = -2.5963x + 2.7203 a) 1 b) 3 c) 0.5 d) 2 e) 5
- Given caesium has a body-centred cube with density of 1.93 g/cm3. i. Determine the lattice constant of caesium if the molar mass of caesium is 132.9 g mol-1. (NA = 6.022 x 1023 atom mol-1) ii. Assuming the body-centred caesium is replaced by an anion, X, suggest the type of structure of this derivative. iii. Determine coordination environment for both ions.What procedures are utilized in optical analysis to determine a solution's concentration based on colorimeter absorbance measurements?Describe how to create metal atomic chains and to measure the G quantization.

