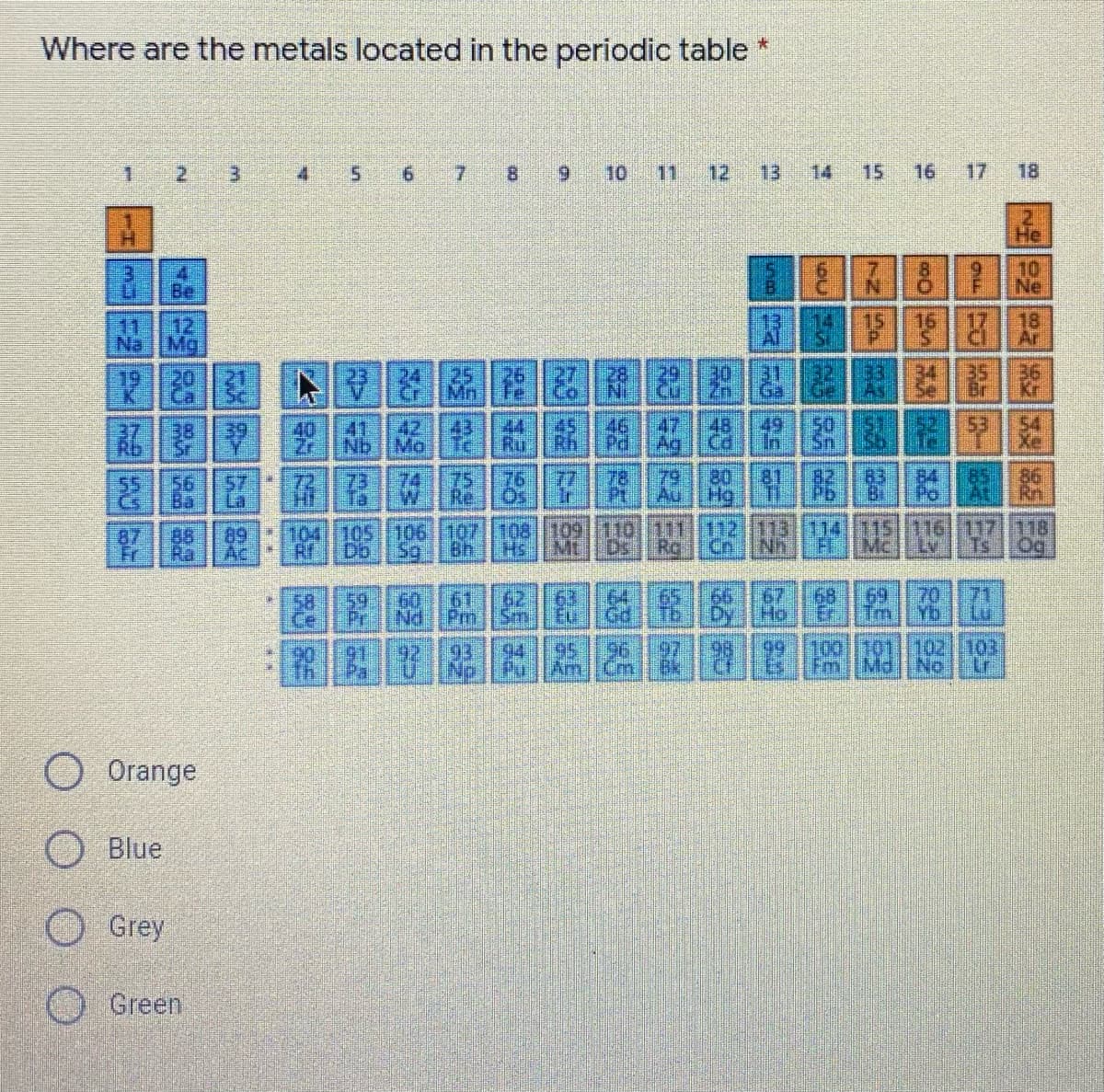
Where are the metals located in the periodic table 1. 4. 6. 6. 10 11 12 13 14 15 16 17 18 He 10 Ne Be 18 Ar 11 12 15 16 Na Mg Al 20 24 Mn Fe Ge Br 47 48 Ag Cd In 53 Xe 40 41 42 43 44 45 46 Zr. Nb Mo Ru Rh Pd 78 77 76 Pt Os Ir 79 80 81 Au Hg 83 84 85 Bi Po 86 Rn 82 75 74 W Re. 55 72 89 87 88 Fr Ra Ac 104 105 106 107 108| 109 110 111 112 113 114 Mt Ds. Hs 115 116 117 118 Rr Db Sg Bh Ro Cn Nh Mc Lv Ts Og 64 65 66 T6 Gd Dy 67 Ho 68 69 Tm Yb Er 70 0 59 58 Pr Nd 60 61 62 Sm Pm. 63 Eu 71 97 Bk 100101|102 103 91 Th 92 93 94 95 96 99 Np Pu' Am Cm Fm Md No Lr O Orange Blue Grey Green
States of Matter
The substance that constitutes everything in the universe is known as matter. Matter comprises atoms which in turn are composed of electrons, protons, and neutrons. Different atoms combine together to give rise to molecules that act as a foundation for all kinds of substances. There are five states of matter based on their energies of attraction, namely solid, liquid, gases, plasma, and BEC (Bose-Einstein condensates).
Chemical Reactions and Equations
When a chemical species is transformed into another chemical species it is said to have undergone a chemical reaction. It consists of breaking existing bonds and forming new bonds by changing the position of electrons. These reactions are best explained using a chemical equation.
Step by step
Solved in 2 steps







