Q: 1. Which of the following is the best primary standard for the standardization of NaOH? a. KHP b.…
A: (1) A main standard is a reagent that can be easily weighed and is sufficiently pure that it…
Q: For the following reaction identify the arrow-pushing pattern that is being utilized: Pattern…
A: In this question we have to tell the name of the step of the given reaction .
Q: Draw the major product of the each of the reactions in the presence of acid and water and the…
A: Potassium dichromate is the stroger oxidising agent which oxidises primary alcohol into direct…
Q: PV=nRT R=0.0821 atm L/mol•K STP: 0 °C, 1.0 atm °C+ 273.15 =K NA= 6.022 x1023 An open flask sitting…
A: Given data :
Q: Draw the structure of isopropylbenzene in the window below. • You do not have to consider…
A:
Q: Br Pd°Ln Me Me MeCN, EtzN Me Me
A:
Q: What's the equilibrium expression for the electrochemical cell w/ the shorthand :Mo│MoO2(aq),…
A: Given the shorthand notation of the electrochemical cell is: Mo(s)│MoO2(aq), H+(aq)││RuO4 (aq),…
Q: During the chemical reaction in an electrochemical cell, the anode is where oxidation happens.…
A:
Q: What will be the expression for Koverall silver carbonate in acidic solution using a combination of…
A:
Q: Nitric acid and nitrogen monoxide react to form nitrogen dioxide and water, like this: 2…
A:
Q: A flavor chemist attempted to isolate and identify the major flavor component of a recently…
A: Given information: Compound 1 has a smaller Rf value (0.2) than compound 2 with Rf value 0.6.
Q: Cve the mechamis m for given bleous reaction n HO.
A: The reaction occurs in a 2- step synthesis reaction. It's mechanism is shown in the following step.
Q: grams of CaCl2
A:
Q: NaH, THF 1. Ph3P. Br e 2. NaOH, H2O2, НаОТHF 3. Mezs I H. NaH, THF
A: In wittig reaction carbonyl group is converted into double bond. NaOH and H2O2 is used for…
Q: equilibrium
A:
Q: Draw the product(s) of the following reactions. H2 CH;CH2-CEC-CH3 Lindlar catalyst
A:
Q: CH3 1) MCPBA 2) CH3NH2
A:
Q: t mass of oxygen is
A:
Q: The fluorocarbon C2Cl3F3 (molar mass: 187.38 g mol-1) has a normal boiling point of 47.6°C. The…
A:
Q: A flavor chemist attempted to isolate and identify the major flavor component of a recently…
A: IR : 3100 cm-1 sp2 C-H stretch, 2950 cm-1 sp3 C-H stretch, 1735 cm-1 carbonyl stretch of ester, 800…
Q: Question 5 How many degrees of unsaturation are present in the following molecules: ii C;H,CI i ii…
A:
Q: Is the molecule below polar or non-polar? Why? Hint: the electronegativity of Ec is 3.4 H. H. Н—Ес —…
A:
Q: Question 5 Choose the best answer! Some questions may have more than one correct answer. Choose all…
A:
Q: CH, c=c CH3 H (1) major organic product ROOR hv (3) process H20, Hgso, (2) enol product (4) keto…
A: The first substrate undergoes polymerization reaction in the presence of peroxide, ROOR and light.…
Q: choose the substance expected to have highest surface tension in liquid phase: a) CH3CH2F b)…
A: Choose the substance expected to have highest surface tension in liquid phase: a) CH3CH2F b)…
Q: HN KON HC Her. H0
A: The mechanism of the following is:
Q: What islare) the predicted productis) of the reaction shown? I. KOH 2. CH,CH,CH,CH,Br 3. H,0 но. он…
A:
Q: 27. The freezing point of camphor is 178.4 C and its molal freezing point, k is 40.0 °C/m. Find the…
A: When the solute particals are added to any solvent, the freezing point of the pure solvent decreases…
Q: value for ΔrS°, in units of J K-1‑mol-1 at 25 °C?
A:
Q: Fill in the table
A:
Q: A reaction proceeds with a 13:3 ratio of enantiomers. Convert this ratio to the percent enantiomeric…
A: % enantiomeric excess is given by the difference of percent major enantiomer and percent minor…
Q: A 36.5 liter balloon holding 2.9 moles of carbon dioxide leaks. If we are able to determine that 1.2…
A: Given : Initial moles of gas = 2.9 mol Initial volume = 36.5 L No.of moles of gas escaped = 1.2…
Q: 182 −mL−mL sample of a 1.3 MM sucrose solution is diluted to 600 mLmL . Part A What is the…
A:
Q: How could you prepare phenol from benzene? 1. HONO, 0°C 2. CuCI 3. NaOH 1. HONO, 0°C 2. CuCl 3. Cu,…
A: Reaction 1 : Nitration of benzene gives nitro benzene. Nitro benzene is converted into aniline suing…
Q: What's the pOH of a 0.0173 mol L-1 KOH solution at 25°C
A: In this question we have to tell the POH of the given solution.
Q: Hypothetical metal M has a molecular weight 159 g/mol and crystallizes in a body-centered cubic…
A:
Q: Consider the following reaction: Calculate the amount of heat transferred when 3.53 g of Mg(s)…
A: Given reaction is 2Mg(s) + O2(g) ------> 2MgO(s) ; ∆H = -1204kJ This reaction is exothermic…
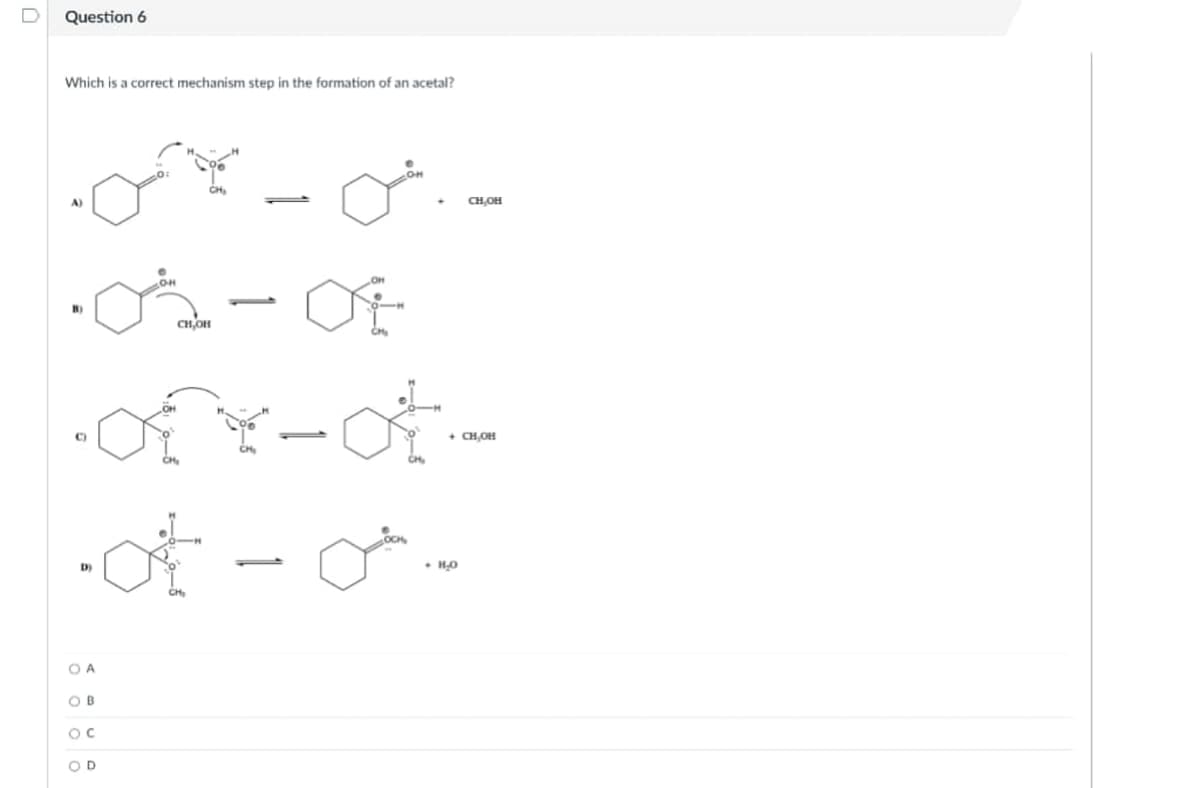
Q: Question 18 Consider the following reaction mechanism: CH3OH(ag) + H*(ag) = CH3OH2*(ag) CH3OH2 (ag)…
A: from the given reaction mechanism, Catalyst is the species which increases the rate of reaction…
Q: bz Portal [Review Topica] [References] Unshared, or lone, electron pairs play an important role in…
A: Unshared pairs can be counted from the structure. Positive charge indicate that the electron is…
Q: The initial rate of the reaction 2 A + B → C + D was studied in a series of kinetics experiments.…
A: To find rate law, first write rate law with given values and doing appropriate calculations, find…
Q: When 1.06 g of a certain molecular compound X are dissolved in 85.0 g of dibenzyl ether…
A:
Q: According to the following thermochemical equation, what mass of HF (in g) must react in order…
A: ΔH°rxn = —184 kJ
Q: 11. From the table of data below: Trial [A] [B] [C] Rate 0.36 M 0.19 M 0.35 M 6.40 X 10-3 0.36 M…
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: Calculate the solubility at 25 °C of BaCrO, in pure water and in a 0.0100M BaCl, solution. You'll…
A:
Q: I have a balloon that can hold 100 liters of air. If I blow up this balloon with 3 moles of oxygen…
A:
Q: What is the pH at the equivalence point in the titration of a 20.7 mL sample of a 0.452 M aqueous…
A: “Since you have asked multiple question, we will solve the first question for you. If youwant any…
Q: Draw the Lewis structure of PF3 and PI3. Determine which statement best describes the Lewis acid or…
A:
Q: 7. Meo HN MeN Br Me
A: Suitable reagents are used for the formation of desired product. Substitution, elimination and…
Q: The KC for the following equilibrium is 2.00 at 1300 K. COF2(g) = CO2(g) + CF4(g) Will a mixture…
A: The balanced reaction is 2COF2 -----> CO2 + CF4 Also there is a rule that if Qc<Kc, reaction…
Q: CH3OH(aq) + H+(aq) ⇋ CH3OH2+(aq) CH3OH2+(aq) + Br-(aq) → CH3Br(aq) + H2O(l) which species…
A: Given :- Step 1 CH3OH(aq) + H+(aq) ⇋ CH3OH2+(aq) Step 2 CH3OH2+(aq) + Br-(aq) → CH3Br(aq)…
Step by step
Solved in 2 steps with 2 images

- 1. Propose the complete reaction mechanism and reagents used for the production of hexan-2-olfrom hex-1-ene. Name the reagents, intermediate and final product/s formed.4.35 Assuming that the rate-determining step in the reaction of 1-hexanol with hydrogen bromideto give 1-bromohexane is an attack by a nucleophile on an alkyloxonium ion, write an equationfor this step. Use curved arrows to show the flow of electrons.QUESTION 7 Identify the best nucleophile in a substitution reaction at a primary carbon. a. H2O b. (CH3)3CO- c. CH3CH2O- d. HO-
- 2-butanol + NOx + hv (assume the carbon-hydrogen bond on the carbon adjacent to the carbonwith the alcohol group is the weakest) For each mechanism, provide the photolytic cycle, a source for hydroxyl radical (if needed) AND atermination step. You may stop when you have accounted for all radicals AND all VOCs are stable (i.e., ifformaldehyde is a product of your mechanism, you do not have to oxidize that molecule to).find a synthesis for each one given below. reaction mechanism is not necessary a) acetylene ---> 2-bromopentane c) ethyl bromide --->trans-3-hexane e) gasoline alcohol ---> benzaldehyde7. Complete the following reaction and propose a mechanism.

