Which is CORRECT for the flow of electrons from NADH? NADH Complex I→ ubiquinone → Complex III → Cytochrome C Cytochrome B-0₂ NADH Complex I→ Complex III Cytochrome C Cytochrome C1 → Complex IV NADH Complex I→ ubiquinone → Complex III → Cytochrome C → Complex IV NADH C - 0₂ NADH IV Complex I→ Complex II → ubiquinone → Complex III Cytochrome Complex I Complex II ubiquinone → Cytochrome C → Complex
Which is CORRECT for the flow of electrons from NADH? NADH Complex I→ ubiquinone → Complex III → Cytochrome C Cytochrome B-0₂ NADH Complex I→ Complex III Cytochrome C Cytochrome C1 → Complex IV NADH Complex I→ ubiquinone → Complex III → Cytochrome C → Complex IV NADH C - 0₂ NADH IV Complex I→ Complex II → ubiquinone → Complex III Cytochrome Complex I Complex II ubiquinone → Cytochrome C → Complex
Concepts of Biology
1st Edition
ISBN:9781938168116
Author:Samantha Fowler, Rebecca Roush, James Wise
Publisher:Samantha Fowler, Rebecca Roush, James Wise
Chapter4: How Cells Obtain Energy
Section: Chapter Questions
Problem 9RQ: What do the electrons added to NAD do? a. They become part of a fermentation pathway. b. They go to...
Related questions
Question
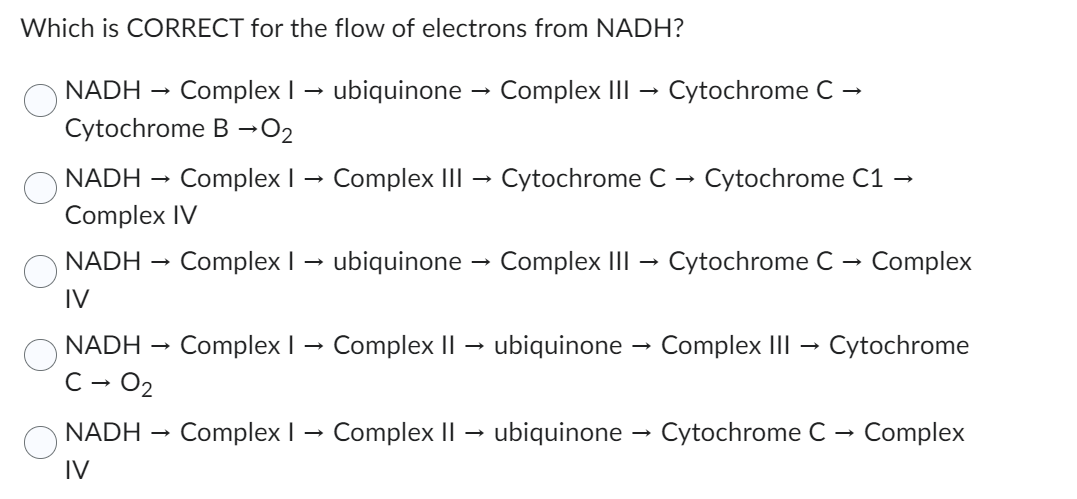
Transcribed Image Text:Which is CORRECT for the flow of electrons from NADH?
NADH Complex I → ubiquinone Complex III → Cytochrome C -
Cytochrome B →O₂
NADH Complex I Complex III → Cytochrome C → Cytochrome C1 →
Complex IV
NADH
Complex I → ubiquinone
IV
NADH
C - 0₂
NADH
IV
Complex I
Complex I
→
Complex III
Cytochrome C → Complex
Complex II → ubiquinone → Complex III Cytochrome
Complex II ubiquinone →→ Cytochrome C Complex
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Concepts of Biology
Biology
ISBN:
9781938168116
Author:
Samantha Fowler, Rebecca Roush, James Wise
Publisher:
OpenStax College


Concepts of Biology
Biology
ISBN:
9781938168116
Author:
Samantha Fowler, Rebecca Roush, James Wise
Publisher:
OpenStax College
