Chapter16: Chemistry Of Benzene: Electrophilic Aromatic Substitution
Section16.SE: Something Extra
Problem 27VC
Related questions
Question
Which of the following alcohol/s would react to Lucas reagent after:
-
<6 min
-
1-5 min
-
<1 min
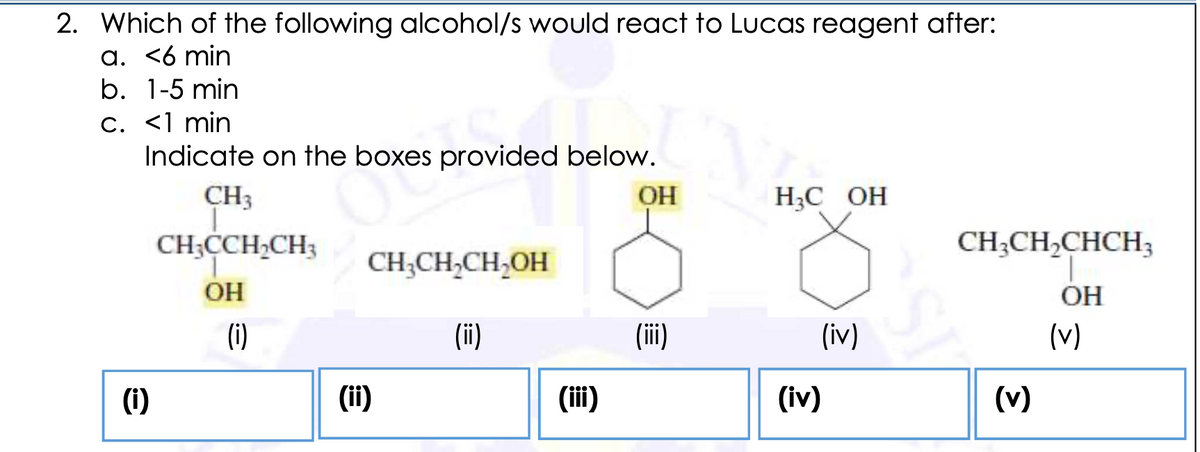
Transcribed Image Text:2. Which of the following alcohol/s would react to Lucas reagent after:
a. <6 min
b. 1-5 min
C. <1 min
Indicate on the boxes provided below.
CH3
OH
НС ОН
CH;CCH,CH3
CH;CH,CHCH3
CH;CH,CH,OH
OH
OH
(i)
(ii)
(ii)
(iv)
(v)
(i)
(ii)
(ii)
(iv)
(v)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Recommended textbooks for you


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning



Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning