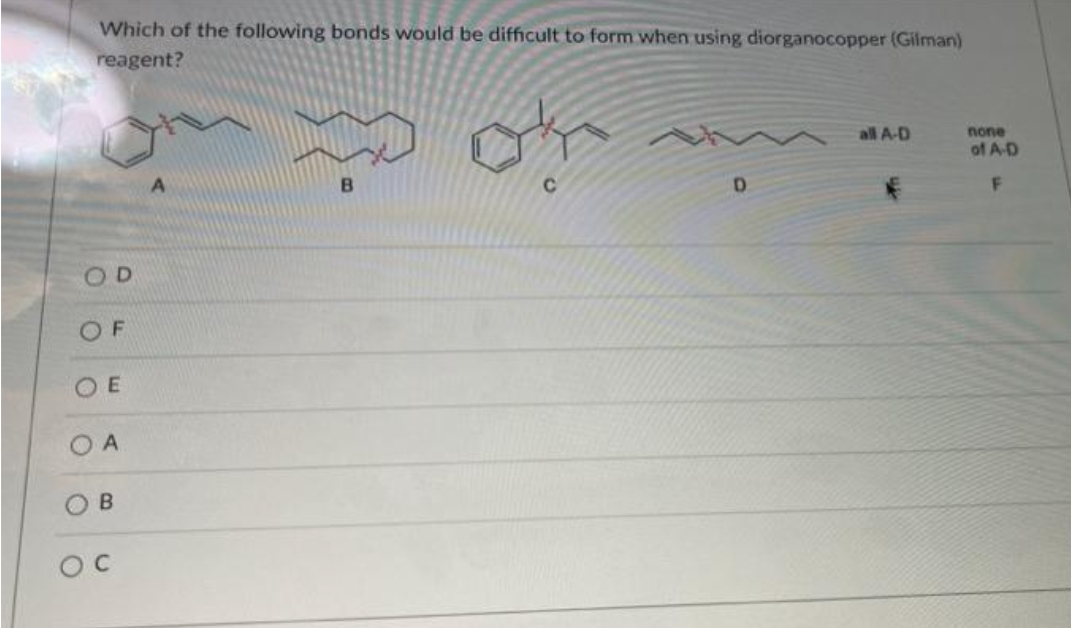
Which of the following bonds would be difficult to form when using diorganocopper (Gilman) reagent? OD OF OE OA O B OC B C D all A-D none of A-D F
Q: How many atoms are there in 2.5 moles of C4H10O2?
A: To find the number of atoms in 2.5 moles of C4H10O2, we need to use Avogadro's number, which is the…
Q: 3. What is the hybridization of each atom (excluding hydrogen atoms) in the following structure?
A: when C atom have one pi bond then hybridization is sp2 and when attached with two pi bonds then…
Q: Fill in the orbital energy diagram for nickel. 4s E 3s- 2s 1s 3p- 2p- 3d AV AV The lowest E levels…
A: This question is related to atomic structure. In the given question, it is asked to draw orbital…
Q: Balance the equation 35U+n→7+Zr+2n. O Te O Te O 137 50 O 135 15 Sn O None of the above
A:
Q: 12. Consider the reaction 4PH3(g) →→→ P4(g) + 6H₂(g) - If, in a certain experiment, over a specific…
A: Given -> 4PH3(g) ---> P4(g) + 6H2(g) Moles of PH3 consumed = 0.0048 mole Volume= 2.0 L
Q: Four liquids are described in the table below. Use the second column of the table to explain the…
A: Given 2.1 g potassium nitrate in 150. mL water 2.1 g of potassium chloride in 150 mL water 2.1 g HI…
Q: The equilibrium constant, K, for the following reaction is 5.00×102 at 642 K. CoCl₂(g) CO(g) +…
A: The given equilibrium equation for the chemical reaction is COCl2(g) ⇔CO(g)+Cl2(g) K…
Q: How many moles of potassium in 6.91 grams of potassium? 0.177 moles O 270. moles O 214 moles 0.223…
A:
Q: 11. Below is the reaction rate of a particular enzyme in the body. Explain what how the enzyme…
A: For an enzyme based reaction, the quantity of product generated per minute is described by the rate…
Q: Calculate the rate of formation for H2, if the rate of change for H2O is a - 0.0559 M/s for the same…
A:
Q: Predict the major product(s) for each of the following reactions. Show both enantiomers if a racemic…
A: -> In presence of peracid there is formation of epoxide . -> Alkene give markovnikov addition…
Q: 5. Draw the structure of the major product for each of the following reactions. CHO NO2 A A
A: The given two reactions are the examples of Diels-Alder reaction. Note: The dienophile with…
Q: The standard enthalpy change for the following reaction is 641 KJ at 298 K. Mg(s) + Cl₂(g) AH° = 641…
A: Given -> MgCl2(s) ------> Mg(s) + Cl2(g) ∆H°= 641 kJ
Q: Given [Ni(CN)4]2 complex ion. i. Identify the ligand. ii. Determine the oxidation state of Nickel.
A: A ligand is an ion or molecule, which donates a pair of electrons to the central metal atom or ion…
Q: A 0.477 mol sample of O₂ gas has a volume of 10.0 L at a certain temperature and pressure. If all…
A:
Q: Give the complete IUPAC name for each of the following compounds.
A: Given : structure of molecules
Q: 3. Write the name for each of the following covalent compounds: (a) CBR4 (b) N₂O3 (c) NF3 (e) P₂Ss…
A: Note: As per the guidelines, solution of 3rd questions has been made. For the expert solution of 4th…
Q: Write a detailed mechanism for the following reaction. Draw the structures of all the expected…
A: In this question, we will give reaction mechanism and also find out the products A, B and C In…
Q: Regarding the chemical compound Ethanol and how Ethanol combustion is exothermic and releases heat,…
A: Ethanol is a commonly used biofuel that is known for its exothermic combustion reaction, releasing…
Q: 2) For the reaction 2A + B →3D, which statement(s) below is/are true? Circle all that apply. a. The…
A: This question is related to chemical kinetics. Chemical kinetics is a branch of chemistry that…
Q: How many of the following compounds contain dispersion forces? a) b) 요 H3C CH3 CH3 NH₂ 4 2 3 1 9 H₂C…
A: In this question, we will see that how many molecules have dispersion forces. You can see the…
Q: Identify the correct stereoisomer and the most stable conformation of the following compound.…
A:
Q: If the starting concentration of CO2 is 0 M, and the reaction rate for the first 10 seconds is 0.060…
A: The rate of appearance of the product is equal to the change in concentration of the product divided…
Q: A chemical engineer is studying the two reactions shown in the table below. In each case, he fills a…
A: Recall the given reactions, 2 CH3OH g + 3 O2 g → 2 CO2 g + 4…
Q: What are some general and specific implications of using titrations to identify unknown acids?
A: Titration is a laboratory technique used to determine the concentration of a solution of known…
Q: lewis struture for C4H9O2
A:
Q: 1. Provide the proper IUPAC name for the following structure: CH3 H3C. CH3 CH3
A:
Q: It takes 945. kJ/mol to break a nitrogen-nitrogen triple bond. Calculate the maximum wavelength of…
A:
Q: A 2 3 3 LOH OH+SOCI, ridine SOCI₂ MgCl OH OH + PBry H.GO H₂O, acetone
A: -> In presence of SOCl2 and pyridine there occur SN2 mechanism and alkyl chloride formed. ->…
Q: 4. Select all that is considered spontaneous a. Cleaning your room b. A piece of bread molding after…
A: A spontaneous process is a process that occurs without the need for an external influence and…
Q: Thalidomide exists in two enantiomeric isomers as shown below. ((R) N۰۰۰ ((S) If a solution is…
A: Given that thalidomide exists in two enantiomeric isomers as shown below A solution is prepared by…
Q: If a reaction is exothermic, and K=100 and Q=1,000, would reactants or products be favored and in…
A: Given: K = 100 Q = 1000 To determine: What direction would equilibrium shift?
Q: Calculate the percent yield and theoretical yield for the following reaction: oxidation of…
A: Given data 0.2093 grams of Cyclododecanol 0.5 mL of acetone 0.20 mL of Glacial acetic acid 2.0 mL…
Q: For each solute, click the button under the better solvent. solute :0: || NH,−C. NH Br NH₂ HO · CH₂…
A: Solubility of a substance in a solvent follows "Like dissolves like " which means if any solvent is…
Q: can you help me what is the polar bond, nonpolar bond, covalent bond, polar molecule, and non-polar…
A: we gave to explain polar bond non polar bond covalent bond polar molecule non polar molecule…
Q: Question: How can this IR graph of the crude product be an evadience that proof it is pure and the…
A: Given that, a synthesis from 9-anthracenemethanol by reacting with N-methylmaleimide to form…
Q: What is the pH of a 2.6 M solution of HCIO? pH =
A: The given information is as follows: The concentration of HClO4 = 2.6 M The pH of solution = ?
Q: What is the activation energy of a reaction which takes place at 300 K, where the rate constant of…
A:
Q: Select the one of the following statements that is NOT correct O If the pka of a weak acid is 4.0,…
A: pKa is a measure of the acidity of a molecule or functional group. It is defined as the pH at which…
Q: C5H₂ClO (one with an alcohol and one with a ketone)
A: Given A molecular formula C5H9ClO
Q: What kind of intermolecular forces act between a hydrogen iodide molecule and a chloroacetylene…
A: Introduction Intermolecular forces are the forces of attraction or repulsion that act between…
Q: Calculate the oxidation number for nitrogen in the follow- compounds. ing (a) NH3 (c) LigN (b) N₂O4…
A:
Q: 1c) What are the expected NMR and IR for the following compound? O₂N.
A: Given that, a structure of a compound is We have to predict its NMR and IR peaks.
Q: hybridization states for each C and O atom in ketene -o atom -c atom 1 -c atom 2
A:
Q: 5) Ca¹2(aq) + Ba(s) = Ca(s) + Ba*²(aq) T (K) Ke 198 6.5 248 12.3 298 18.7 Use the experimental data…
A: Explanation Experimental determination of ∆Ho and ∆So Since we know from the relation between ∆Go…
Q: Write the complete electron configuration for the sodium atom. Using NOBLE GAS notation write the…
A: The electronic configuration can be written by using three basic rule 1. Aufbau principle. 2. The…
Q: The reaction A→ B has a standard state free energy of -15 kJ/mol and a cellular free energy of 0…
A: Introduction Spontaneous reaction in chemistry is a process in which a chemical reaction proceeds…
Q: AE - - بهتر n = 2,726 -> 3 | 1,
A: Given that, ∆E = 2.18×10-181nf2-1ni2. Also, given that, nf = 2, λ = 650 nm, ∆E = 3.1×10-19 J. We…
Q: A certain reaction was found to have a rate constants 7.04x10-3 min-1 at 152°C and 5.54×10-2 min-1…
A:
Q: ОН POCIĄ pyridine
A: In presence of POCl3 and pyridine there is removel of Water and double bond formed.
Bb.25.
Step by step
Solved in 2 steps

- 1. Write balanced redox rxns for the rxn of KMnO4 with compound A to give compound B 2. Draw the products obtained from the following rxnWhat is the purpose of using NaHCO3 solution in the isolation step of the Fischer esterification reaction? Draw a reaction scheme to support your answer.A wittig reaction experiment: Week 1 -》 preparation of phosphonium salt Materials : triphenylphosphine (5.3g),methyl Bromoacetate 3.36g (2.1ml) , ethanol 30ml . The yield obtained was 8.93g . Calculate the percentage yield Week 2 -》 Formation of the tlide and wittig reaction Materials :Phosphonium salt (5g), napthyl-2- carboxwaldehyde( 2.65g), 20 ml of water and 5 nl of Nahco3. Yield obtained was 0.51g .Calculate the percentage yield Week 3: Solvent free wittig reaction Materials: Benzyltriphenylphosphonium chloride (0.5g ), 4 - Bromoabenzaldehyde (0.24g), Potassium phosphate (tribasi c) 0.275g .The yield obtained was 1.21g. Calculate the percentage yield
- 2-Chlorobutane reacts much more slowly than 2-chloro-2-methylpropane in the silver nitrate test. Explain this difference in reactivity.When oxalic acid is added to water the oxalic acid will dissolve. If D2O were used in place of H2O would the deuterium become incorporated into every site where there was a hydrogen atom in the original structure? Explain2. Anhydride means “without water”. Suppose 1 M H2SO4 were substituted for the concentrated H2SO4. Would the yield of acetylsalicylic acid be increased, decreased, or unaffected by the substitution? Explain.
- For the following pairs, determine what could be the best solvent to differentiate them based on their predicted solubility. 1. beta-naphthol and butyric acid 2. anthracene and benzylamine 3. p-xylene and benzaldehydeResults and Discussion: Nitroprusside Reaction: Samples: cysteine, cystine, glycine Reagents: Saturated Sodium Carbonate (Na2CO3) solution, Saturated Sodium Nitroprusside solution, 1.0% sodium cyanide (NaCN) -To 0.5 ml of the sample, add 0. 25 ml of saturated sodium carbonate solution and a drop of freshly prepared saturated solution of sodium nitroprusside (HANDLE WITH CARE). A purple color will develop if free sulfhydryl groups are present. If negative, add 1 drop of 1.0% NaCN. The development of a red color indicated the presence of disulphide bonds.Kiko, a Chem 31.1 student, accidentally shuffled two test tubes with different compounds on his way to the laboratory. To confirm the possible identities of the compounds, Kiko added neutral KMnO4 and Lucas reagents to both test tubes labeled A and B. Test tube A gave a brown precipitate in neutral KMnO4 while test tube B produced a purple solution in the same test. Upon subjecting to Lucas Test, both test tubes produced layers. Which of the following are the possible identities of the compounds A and B? a. Sec-butanol and 3-methyl-3-pentanol b. n-propanol and tert-butanol c. isopropyl alcohol and 4-nitrophenol d. hexanol and cyclopentanol

