Q: draw the 6 isomers of Ni(NCS)2(PPh3)2?
A: Step 1: Bidentate ligand-NCS is Bidentate ligand (ligands which bind to central metal through two…
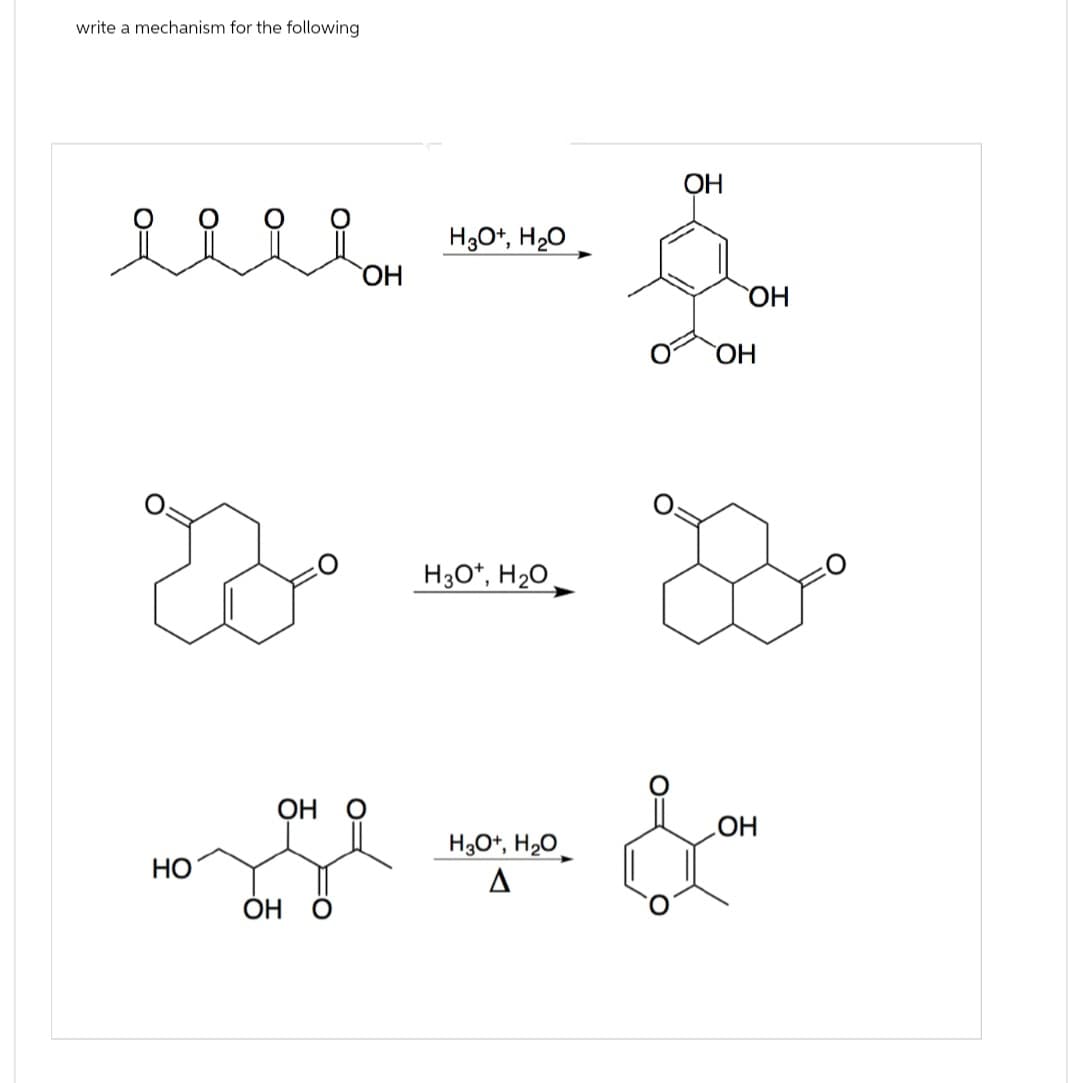
Q: Drow mechanism and full details
A: Step 1: Cl2 + heat does chlorination. Step 2: H2O acts as a nucleophile and substitutes chlorine.…
Q: A high-pressure natural gas tank is at 3600 psi. The tank contains 4.98 mol of methane (CH4) and…
A: The objective of this question is to calculate the partial pressure of methane and ethane in a…
Q: Online teaching and +…
A: Step 1: Step 2: Step 3:
Q: A buffer solution was prepared by adding 4.92 g of sodium acetate, NaCH3CO2, to 2.50 x 10^2 mL of…
A: The objective of this question is to calculate the pH of a buffer solution after adding a certain…
Q: i dot want hand written image
A: Step 1:The reaction involves deprotonation of acetone by LDA to form an enolate ion, which then…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 'N' 1. CH2=CHCH2Br THF, reflux…
A:
Q: Let operator Ĥ. Let be one of the normalized eigenfunctions of a Hamiltonian be an approximation of…
A: The equation given is:Where: is one of the normalized eigenfunctions of the Hamiltonian operator .…
Q: Use the table of indicators below to choose the most appropriate indicator to use when titrating…
A: Benzoic acid is a weak acid and NaOH is a strong base. At the equivalence point where moles of acid…
Q: 5. Sketch a synthetic route using ethanol (CH3CH2OH) as the only source of carbon atoms for the…
A: The objective of the question is to sketch a synthetic route using ethanol as the only source of…
Q: Limiting Reactant Practice If you were to burn 20.5 grams of methanol, CH3OH, in 35.6 liters of…
A: Step 1:The balanced reaction is:2CH3OH(l) + 3O2(g) -----> 4 H2O(l) + 2CO2(g) Steps to…
Q: A small piece of papyrus is to be 14C dated by a 10 minute run of the detector set to record 14C…
A: The objective of the given data is to determine the required values of the papyrus sample that is…
Q: Draw all the products of this reduction reaction: H₂N O= ZIR H3C O CH3
A: Step 1: Reduction of Imine to Amines: The process by which primary and secondary amines are produced…
Q: OWLv2 | Online teaching and lea: X Draw the organic product(s) of t X Dow the mat orga A ✰…
A:
Q: Two enzymes, E1 and E2, respectively catalyze the conversion of A to B, and B to C in two reactions…
A: The objective of this question is to determine the steady-state concentrations in a series of…
Q: Using the spectroscopic data given, determine the structure of the unknown
A:
Q: Name the given compound. Make sure you include the appropriate configuration for each of the chiral…
A: In the structure, the parent chain is the cyclopentane.Next, it has a substituent that has 8 as the…
Q: a) Which of the following chemical species could have their electronic ground state energy and…
A: To identify whether the given system can be analyzed using the Restricted Hatree Fock method, we…
Q: Content C °- OWLv2 | O × Google Do G google slid x Inbox (4,5 x Certificate ☑ > Course E X Cengage L…
A: The objective of the question is to determine whether the BH3 fragment in the reaction between…
Q: 1. Calculate the change in pH when 5.00 mL of 0.0100 M HNO3 is added to 50.0 mL of water. 2. Write…
A: 1) Given, V1 = 5 mL = 5 × 10-3 L M1 = 0.0100 M Molarity, M = number of moles / volume in L,…
Q: Propose an efficient synthesis for the following transformations using ONLY aldol, mciheal and other…
A: Step 1:• Mechanism: • In this reaction. Firstly alcohol is get oxidised to aldehyde to get…
Q: Part A Estimate the optimal number of neutrons for a nucleus of Ho (Z=67). Express the number of…
A:
Q: QUESTION 3 Predict the product(s) for this reaction. Y H-SCH₂CH3 THE SCH₂CH₂ SCH₂ SCH₂CH3 Οι O II ОШ…
A:
Q: Predict the products of the following triglyceride when it undergoes saponification. Draw the arrow…
A:
Q: Identify and provide an explanation of 'Multi-point Calibration'. Provide examples.
A: See answer section.
Q: 3
A: Step 1: Step 2: Step 3: Step 4:
Q: provide a synthetic route for the following
A: Step 1: Step 2: Step 3: Step 4:
Q: For which reaction will ΔS be positive? Group of answer choices N2O4(g) ⟶ 2 NO2(g) 2 H2(g) +…
A: The objective of the question is to determine for which reaction the change in entropy (ΔS) will be…
Q: None
A: I'd be glad to help you predict the products of the following Diels-Alder reactions: (a)Reactants:…
Q: OH H OH H OH H H OHN HH JE OH HI H OH F CH₂OH Consider the structure of the dissaccharide…
A:
Q: Construct a two-step synthesis of (±) leucine by dragging the appropriate formulas into the bins.…
A: The suitable molecules and reagents are used in the given transformation are as follows:Detailed…
Q: 1. Draw the two Watson-Crick Bases Pairs and show how G- C and A-T recognize each other by hydrogen…
A: The concept of the central dogma of biochemistry provides a framework for understanding how genetic…
Q: i dont want hand writting image
A: The reaction in question involves sodium azide (NaN3) and an alkyl halide in DMF…
Q: None
A: Consider the below explanation;In the provided structural formula, the starting compound is a hexane…
Q: 22. Elements that have metallic and non-metallic properties are called a. metalloids…
A: The question is asking to identify the term for elements that exhibit both metallic and non-metallic…
Q: Complete each reaction giving the product or the reactants, whichever applies.
A: Step 1: Step 2: Step 3: Step 4:
Q: For the Diels-Alder reaction, the product's skeletal structure and configuration at the bridgehead…
A: The objective of this question is to complete the structure of the major product of a Diels-Alder…
Q: Synthesis: Give the correct starting material, reagents used or product as indicated. If you are not…
A: 1) Under reductive conditions, ozonolysis produces ketone or aldehyde upon cleavage of alkene or an…
Q: The molar heat of vaporization of substance X at its normal boiling point -185.85 °C is 6.53 kJ…
A: The enthalpy of a process can be described as the amount of heat released or absorbed per mole of…
Q: Complete each reaction giving the product or the reactants, whichever applies.
A: Step 1: Step 2: Step 3: Step 4:
Q: 6. Draw the structures of the major products from the reactions below NBS light NBS light
A:
Q: 3. Draw the correct product for the following Diels-Alder reaction, make sure to include any…
A:
Q: At -5.92 °C the pressure equilibrium constant K = 5.9 for a certain reaction. P Here are some facts…
A: As per the given data, the Kp value at -27 degrees Celsius is calculated below:
Q: 1) Draw the products of the following reactions. Draw stereoisomers and identify them as…
A:
Q: Provide the mechanism for the saponification of the following compound with HCl used in the work up:…
A: Step 1: The reaction involves nucleophilic addition, elimination, deprotonation and acidic workup.…
Q: 12.4 Recorded Macro: Insert the Ideal Gas Constant Record a macro that will insert the ideal gas…
A: Normally we use 8.314 gmolKJ for ideal gas constant in ideal gas law equation pv= nRT
Q: On the molecule below, highlight primary halide carbons in red, secondary halide carbons in blue,…
A: Step 1:to identify primary, secondary, and tertiary halide carbons in a molecule. We need to…
Q: 5) Fill in the missing reagents for the total synthesis below. For the final step, propose a…
A: Step 1: Methanolysis of the ester to give the methyl ester. Step 2: Reaction of the methyl ester…
Q: Using thermodynamic data to calculate K Using any data you can find in the ALEKS Data resource,…
A:
Q: None
A:
Step by step
Solved in 2 steps with 2 images

- Define the mechanism of acid–catalyzed halogenation ?Draw the structures of the intermediate bromonium and cyclic carbocation and propose mechanisms for all three steps.Provide the mechanism for the Friedel-Crafts acylation reaction of compound Q and 2-(1- phenylethyl)-1,3-dioxolane that leads to the formation of intermediate R.
- Explain and detail the reaction mechanism: NaBr + H2O + n-butyl alcohol = ?4 Show how the compound thiodiazine~C21H26N2S2~decomposes anaerobically and how these end products in turn decompose aerobically to stabilized sulfur and nitrogen compounds.[Rh(PPh3)3Cl] is a precatalyst that can be activated by dissociation of a phosphine ligand to form an active catalyst, B, which is used in the hydrogenation of alkenes. The active catalyst can then undergo oxidative addition in the presence of H2 to form complex C. Propene coordinates to C to form complex D, which then undergoes a 1,2- insertion step to form E. Reductive elimination of propane from E regenerates the active catalyst B. Draw complexes B – E and hence provide a full catalytic cycle, including the activation step, for the hydrogenation of propene
- What is the mechanism of the reaction of HBr addition to 1Butene in the presence or absence of peroxideWrite a mechanism for the bromination of trans-cinnamic acid that proceeds through a bromonium ion intermediate that clearly shows the stereochemical outcome of the reaction. Show (R,S) configurations of the product(s).4b)Give the mechanisms for the following:
- 1-Suggest a mechanism by which the ozone, O3 is being destructed by the presence of oxygen molecule, O2, and the sun light. 2-Methyl chloride, CH3Cl, is another molecule that destructs the ozone layer. Suggest a mechanism for the destruction of ozone by methyl chloride and the sun light.Provide mechanisms for this reactionList the following in increasing order of nucleophilic strength. CH3COO-, H2O, CH3O-, OH-, CH3CH20-

