Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter16: Synthesis Workshop 1
Section: Chapter Questions
Problem 28E
Related questions
Question
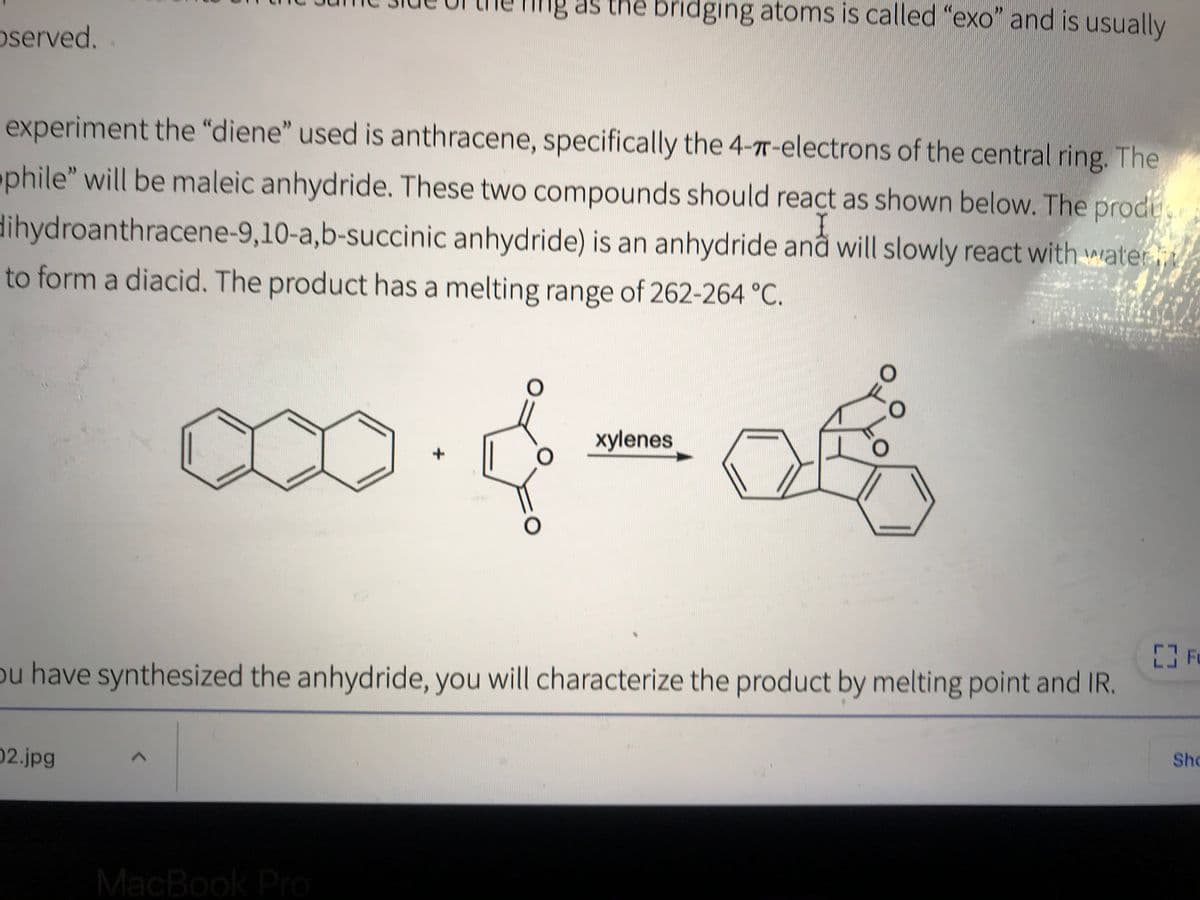
Transcribed Image Text:Ing as the bridging atoms is called "exo" and is usually
oserved.
experiment the "diene" used is anthracene, specifically the 4-7T-electrons of the central ring. The
phile" will be maleic anhydride. These two compounds should react as shown below. The prode
I
dihydroanthracene-9,10-a,b-succinic anhydride) is an anhydride anå will slowly react with water
to form a diacid. The product has a melting range of 262-264 °C.
O.
xylenes
ou have synthesized the anhydride, you will characterize the product by melting point and IR.
Sho
02.jpg
MacBook Pro
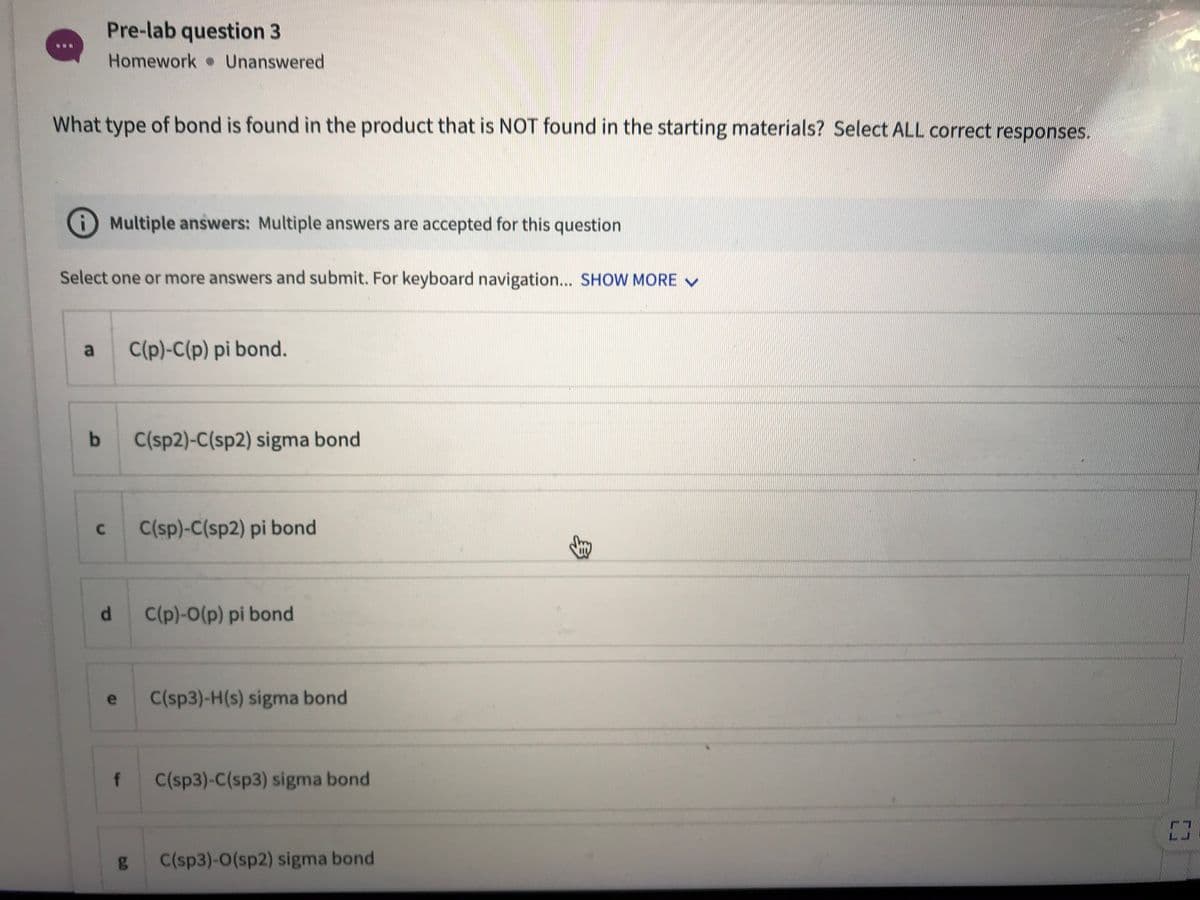
Transcribed Image Text:Pre-lab question 3
Homework Unanswered
What type of bond is found in the product that is NOT found in the starting materials? Select ALL correct responses.
Multiple answers: Multiple answers are accepted for this question
Select one or more answers and submit. For keyboard navigation... SHOW MORE V
a
C(p)-C(p) pi bond.
C(sp2)-C(sp2) sigma bond
C(sp)-C(sp2) pi bond
d.
C(p)-0(p) pi bond
C(sp3)-H(s) sigma bond
e
f
C(sp3)-C(sp3) sigma bond
g C(sp3)-0(sp2) sigma bond
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole
