
Concept explainers
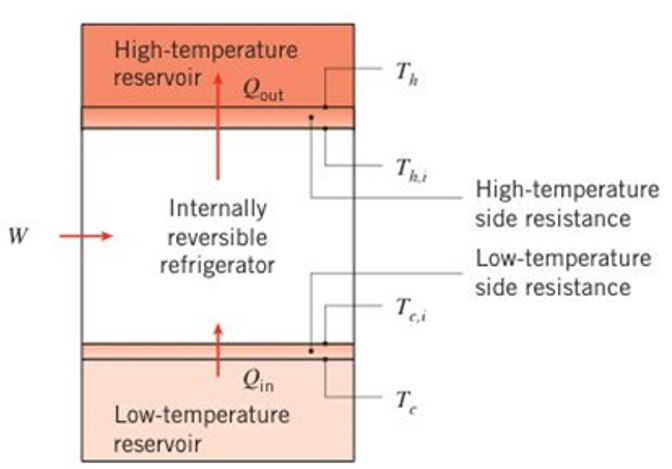
An internally reversible refrigerator has a modified coefficient of performance accounting for realistic heat transfer processes of
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Introduction to Heat Transfer 6th Edition (Wiley Editor's Choice Edition)
- The operating condition for the single compressor in a household refrigerator is the lowest box temperature, which is typically A. 0F B. -20F C. 20F D. 40Farrow_forwardA window air conditioner that consumes 1 kW of electricity when running and has a coefficient of performance of 3 is placed in the middle of a room and is plugged in. The rate of cooling or heating this air conditioner will provide to the air in the room when running is (a) 3 kJ/s, cooling (b) 1 kJ/s, cooling (c) 0.33 kJ/s, heating (d) 1 kJ/s, heating (e) 3 kJ/s, heatingarrow_forwardAn inventor claims to have developed a refrigerator that at steady state requires a net power input of 1.1 horsepower to remove 12,000 Btu/h of energy by heat transfer from the freezer compartment at -30°F and discharge energy by heat transfer to a kitchen at 70°F. Evaluate this claim. This claim is a) impossible b) possiblearrow_forward
- What is the coefficient of performance of a refrigerator that operates with Carnot efficiency between temperatures -3.00°C and +27.0°C?arrow_forwardA Carnot heat engine, shown in Fig. 6–47, receives 500 kJ of heat per cycle from a high-temperature source at 652°C and rejects heat to a low-temperature sink at 30°C. Determine the thermal efficiency of this Carnot engine.arrow_forwardA typical new household refrigerator consumes about 680 kWh of electricity per year and has a coefficient of performance of 1.4. The amount of heat removed by this refrigerator from the refrigerated space per year is (a) 952 MJ/yr (b) 1749 MJ/yr (c) 2448 MJ/yr (d) 3427 MJ/yr (e) 4048 MJ/yrarrow_forward
- A completely reversible heat engine operates with a source at 1500 R and a sink at 500 R. At what rate must heat be supplied to this engine, in Btu/h, for it to produce 5 hp of power?arrow_forwardIt is claimed that a certain cyclical heat engine operates between the temperatures of TH = 448°C and TC = 155°C and performs W = 3.98 MJ of work on a heat input of QH = 4.97 MJ. a. How much heat, in megajoules, would be discharged into the low-temperature reservoir? b. Which choice, by itself, is unreasonable about the claim? 1. The efficiency is greater than the Carnot efficiency for the stated temperatures 2. The work performed is less than the heat input. 3. The temperature difference is greater than 273.15 K. 4. All of these. 5. None of thesearrow_forwardtrue or false A refrigerator (COP = 2.5) is used to maintain a lab temperature at 18° C for that 80 MJ of heat is absorbed. Then its work input would be 8.889 kJ/s.arrow_forward
- 1.Refrigerator 1 simulates an ideal refrigerator and therefore operates on a Carnot cycle using R-134a as the refrigerant at a flow rate of 1.4 kg/sec. The condensing and evaporating temperatures are 30 °C and -10 °C, respectively. To assess the performance of Refrigerator 1, you must submit the report. of the project with the following information: a) Enthalpies corresponding to the states indicated in the cycle (1, 2, 3 and 4); b) The cooling rate (Q L ); c) The work supplied to the fluid by the compressor; d) The work generated by the turbine; e) The condenser heat rejection rate (Q H ); f) The coefficient of performance of the cycle. Important detail: For the energy balance, make the following considerations: ✓ Permanent regimen; ✓ Kinetic and potential energy variations are negligible; ✓ Compressor and turbine operate adiabatically; ✓ Evaporation and condensation steps are labor-free.arrow_forwardProve that a refrigerator’s COP cannot exceed that of a completely reversible refrigerator that shares the same thermal energy reservoirs.arrow_forwardA cycle heat engine does 50 kJ of work per cycle If the efficiency of the heat engine is 75% The heat rejected per cycle is....arrow_forward
 Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
