
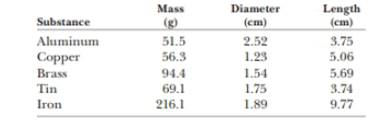
The data in the following table represent measurements of the masses and dimensions of solid cylinders of aluminum, copper, brass, tin, and iron. (a) Use these data to calculate the densities of these substances. (b) State how your results compare with those given in Table 14.1.
(a)
The densities of each substance.
Answer to Problem 1.61AP
The density of aluminum solid cylinders is
Explanation of Solution
Given info: The mass, diameter and length of each substance are given below,
| Substance | Mass
| Diameter
| Length
|
| Aluminum |
|
|
|
| Copper |
|
|
|
| Brass |
|
|
|
| Tin |
|
|
|
| Iron |
|
|
|
Formula to calculate the density of substance is,
Here,
Write the expression for the volume of solid cylinder,
Here,
Substitute
For aluminum:
Substitute
Thus, the density of aluminum solid cylinders is
For copper:
Substitute
Thus, the density of copper solid cylinders is
For brass:
Substitute
Thus, the density of brass solid cylinders is
For tin:
Substitute
Thus, the density of tin solid cylinders is
For iron:
Substitute
Thus, the density of iron solid cylinders is
Conclusion:
Therefore, the density of aluminum solid cylinders is
(b)
The comparison between results of part (a) and table
Answer to Problem 1.61AP
The density of aluminum from table is
Explanation of Solution
Given info:
Formula to calculate the percentage error is,
Here,
For aluminum:
From part (a), the density of the aluminum is
Substitute
Thus, the density of aluminum from table is
For copper:
From part (a), the density of the copper is
Substitute
Thus, the density of copper from table is
For brass:
From part (a), the density of the brass is
Substitute
Thus, the density of brass from table is
For tin:
From part (a), the density of the tin is
Substitute
Thus, the density of tin from table is
For iron:
From part (a), the density of the iron is
Substitute
Thus, the density of iron from table is
Conclusion:
Therefore, the density of aluminum from table is
Want to see more full solutions like this?
Chapter 1 Solutions
Physics for Scientists and Engineers, Volume 1
Additional Science Textbook Solutions
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
Physics: Principles with Applications
College Physics
Cosmic Perspective Fundamentals
Essential University Physics (3rd Edition)
- A hollow spherical container has an outer diameter of 10.50 cm. The thickness of the walls is 0.50 cm. The container is filled with water. Water molecules are approximated to be spheres with a diameter of 275 pm. How many water molecules are present inside of the container? Your answer needs to have the correct number of significant figures.arrow_forwardQuestion: Lets think about the pressure difference for bubbles in soda. If the bubble has a radius of 1 um and a surface tension of 0.072 N/m what is the pressure difference between the bubble and the soda? Give your answer in 3 significant figures. Solution: The pressure difference is Paarrow_forwardThe unit cell of platinum has a length of 392.0 pm along each side. Use this length (and the fact that Pt has a face-centered unit cell) to calculate the density of platinum metal in kg/m3 (Hint: you will need the atomic mass of platinum and Avogadro's number).arrow_forward
- Question 1. The table below shows a collection of data for a thermocouple. Temperature is in degrees Celsius, and the Voltage is in millivolts. (a) Plot the graph using rectangular graph paper with Temperature as the independent variable and determine the basic functional relationship between Temperature and Voltage. Alternatively, you can use Microsoft Excel to plot the data, but attach the graph in your answer page. (b) Determine specific the equation, using the method of selected points that best fits the data. Show how you determined the equation. If you need help, review Lecture-5 (c) What is the thermocouple voltage when the temperature is 1225 degree Celsius? Temperature (degree Celsius) Voltage (millivolts) 50 100 150 200 300 400 500 600 700 800 900 1000 2.5 6.7 8.8 11.2 17 22.5 26 32.5 37.7 41 48 55.2arrow_forwardRead the volume of the liquid in the Erlenmeyer flask. Estimate the volume to the nearest 5 mL increment and enter it with the proper number of significant figures. volume: volume: ml Ű Read the volume of the liquid in the beaker. Estimate the volume to the nearest 5 mL increment and enter it with the proper number of significant figures. Show Transcribed Text mL 100 Ċ M 200 ml 150 30 100 50 100 50arrow_forwardIn the figure below, what is the pressure of the gas in the flask (in atm) if the barometer reads 693.4 torr? (Ah = 8.37 cm) oo Round your answer to 4 significant figures. Open end Gas Ah Note: Reference the SI prefixes and Conversion factors for non-SI units tables for additional information. Pressure = 0.9123 atm 0 x10 X olo Ar Barrow_forward
- What is the pressure of the gas in the cylinder, in kPa (kiloPascal)? h = 0.620 m, Pmercury = 13,600 kg/m3, 1.0 atm = 1.00 x 105 Pa = 100 kPa, and g = 10.0 m/s?. Your answer needs to have 3 significant figures, including the negative sign in your answer if needed. Do not include the positive sign if the answer is positive. No unit is needed in your answer, it is already given in the question statement. Mercury. p = 13,600 kg/m² Penarrow_forwardWhat is the pressure of the gas in the cylinder, in kPa (kiloPascal)? h = 0.700 m, ρmercury = 13,600 kg/m3, 1.0 atm = 1.00 × 105 Pa = 100 kPa, and g = 10.0 m/s2. Your answer needs to have 3 significant figures, including the negative sign in your answer if needed. Do not include the positive sign if the answer is positive. No unit is needed in your answer, it is already given in the question statement.arrow_forward5.10arrow_forward
- What is the pressure of the gas in the cylinder, in kPa (kiloPascal)? Pmercury = 13,600 kg/m3, 1.0 atm = 1.00 x 105 Pa = 100 kPa, and g = 10.0 m/s?. Your answer needs to have 3 significant figures, including the negative sign in your answer if needed. Do not include the positive sign if the answer is positive. No unit is needed in your answer, it is already given in the question statement. Pgas -Mercury 16 cm 6 cmarrow_forwardA balloon contains 6 L of He. The pressure is reduced to 1 atm and the balloon expands to occupy a volume of 12.25 L, T constant. What was the initial pressure exerted on the balloon? (Give your answer correct to two decimal places) The pressure is atmarrow_forwardI am a little confused on this question , please help. Thank you for your time! I'm supposed to solve using Scientific Notation and stating the correct units in answer. I have attached the problem .Please view attachment before answering. Thank you again!arrow_forward
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning





