
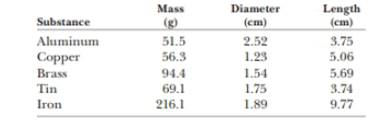
The data in the following table represent measurements of the masses and dimensions of solid cylinders of aluminum, copper, brass, tin, and iron. (a) Use these data to calculate the densities of these substances. (b) State how your results compare with those given in Table 14.1.
(a)
The densities of each substance.
Answer to Problem 1.61AP
The density of aluminum solid cylinders is
Explanation of Solution
Given info: The mass, diameter and length of each substance are given below,
| Substance | Mass
| Diameter
| Length
|
| Aluminum |
|
|
|
| Copper |
|
|
|
| Brass |
|
|
|
| Tin |
|
|
|
| Iron |
|
|
|
Formula to calculate the density of substance is,
Here,
Write the expression for the volume of solid cylinder,
Here,
Substitute
For aluminum:
Substitute
Thus, the density of aluminum solid cylinders is
For copper:
Substitute
Thus, the density of copper solid cylinders is
For brass:
Substitute
Thus, the density of brass solid cylinders is
For tin:
Substitute
Thus, the density of tin solid cylinders is
For iron:
Substitute
Thus, the density of iron solid cylinders is
Conclusion:
Therefore, the density of aluminum solid cylinders is
(b)
The comparison between results of part (a) and table
Answer to Problem 1.61AP
The density of aluminum from table is
Explanation of Solution
Given info:
Formula to calculate the percentage error is,
Here,
For aluminum:
From part (a), the density of the aluminum is
Substitute
Thus, the density of aluminum from table is
For copper:
From part (a), the density of the copper is
Substitute
Thus, the density of copper from table is
For brass:
From part (a), the density of the brass is
Substitute
Thus, the density of brass from table is
For tin:
From part (a), the density of the tin is
Substitute
Thus, the density of tin from table is
For iron:
From part (a), the density of the iron is
Substitute
Thus, the density of iron from table is
Conclusion:
Therefore, the density of aluminum from table is
Want to see more full solutions like this?
Chapter 1 Solutions
PHYSICS F/...V.1 (LL)-W/ACCESS >CUSTOM<
Additional Science Textbook Solutions
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
Physics: Principles with Applications
College Physics
Cosmic Perspective Fundamentals
Essential University Physics (3rd Edition)
- How many cubic meters of helium are required to lift a balloon with a 400-kg payload to a height of 8 000 m? Take He = 0.179 kg/m3. Assume the balloon maintains a constant volume and the density of air decreases with the altitude z according to the expression air = 0ez/8, where z is in meters and 0 = 1.20 kg/m3 is the density of air at sea level.arrow_forwardMercury is poured into a U-tube as shown in Figure P15.17a. The left arm of the tube has cross-sectional area A1 of 10.0 cm2, and the right arm has a cross-sectional area A2 of 5.00 cm2. One hundred grams of water are then poured into the right arm as shown in Figure P15.17b. (a) Determine the length of the water column in the right arm of the U-tube. (b) Given that the density of mercury is 13.6 g/cm3, what distance h does the mercury rise in the left arm?arrow_forwardHow many cubic meters of helium are required to lift a light balloon with a 400-kg payload to a height of 8 000 m? Take Hc = 0.179 kg/m3. Assume the balloon maintains a constant volume and the density of air decreases with the altitude z according to the expression pair = 0e-z/8 000, where z is in meters and 0 = 1.20 kg/m3 is the density of air at sea level.arrow_forward
- Refer to Problem 16 and Figure P14.16. A hydrometer is to be constructed with a cylindrical floating rod. Nine fiduciary marks are to be placed along the rod to indicate densities of 0.98 g/cm3, 1.00 g/cm3, 1.02 g/cm3, 1.01 g/cm3, 1.14 g/cm3. The row of marks is to start 0.200 cm from the top end of the rod and end 1.80 cm from the top end. (a) What is the required length of the rod? (b) What must be its average density? (c) Should the marks be equally spaced? Explain your answer.arrow_forwardA liquid with a coefficient of volume expansion just fills a spherical shell of volume V (Fig. P16.53). The shell and the open capillary of area A projecting from the top of the sphere are made of a material with an average coefficient of linear expansion . The liquid is free to expand into the capillary. Assuming the temperature increases by T, find the distance h the liquid rises in the capillary.arrow_forwardThe spirit-in-glass thermometer, invented in Florence, Italy, around 1054, consists of a tube of liquid (the spirit) containing a number of submerged glass spheres with slightly different masses (Fig. P15.70). At sufficiently low temperatures, all the spheres float, but as the temperature rises, the spheres sink one after another. The device is a crude but interesting tool for measuring temperature. Suppose the tube is filled with ethyl alcohol, whose density is 0.789 45 g/cm3 at 20.0C and decreases to 0.780 97 g/cm3 at 30.0C. (a) Assuming that one of the spheres has a radius of 1.000 cm and is in equilibrium hallway up the tube at 20.0C, determine its mass. (b) When the temperature increases to 30.0C, what mass must a second sphere of the same radius have to be in equilibrium at the halfway point? (c) At 30.0C, the first sphere has fallen to the bottom of the tube. What upward force does the bottom of the tube exert on this sphere?arrow_forward
- Every day, Jill collects one full bucket of water from the well up the hill. The well has a diameter of 0.800 m and a depth of 3.00 m. Currently, only 65.3% of the well is full. If Jill collects one full bucket every day from today, and it takes her 851 days to empty the well, how many pints of water was she collecting per trip? 1 qt = 2 pints, 1 L = 1.06 qt, 1 m3 = 1000 L, V = πr2harrow_forwardWhich statement about isotherms of a cubic EOS is/are FALSE? I. At Tr = 1.0 and any pressure, the first and second derivatives of P with respect to V will both be equal to zero.II. When Tr < 0 the substance is always a VLE mixture inside the pressure dome. a.Neither I nor II b.Both I and II c.I only d.II onlyarrow_forwardYou purchase a rectangular piece of metal that has dimensions 5.0 * 15.0 * 30.0 mm and mass 0.0158 kg. The seller tells you that the metal is gold. To check this, you compute the average density of the piece. What value do you get? Were you cheated?arrow_forward
- 1. a cubical object made of cast aluminum 0.224 m on a side contains a hollow cavity also in the shape of a cube but of unknown dimensions. If the mass of the object is 29.5 kg, what is the length of each side of the interior cubical cavity. density of Al = 2.70 103 kg/m3.arrow_forwardThe U.S. Mint produces a dollar coin called the American Silver Eagle that is made of nearly pure silver. This coin has a diameter of 41 mmmm and a thickness of 2.5 mmmm. The density and approximate market price of silver are 10.5 g/cm3g/cm3 and $0.58 per gram, respectively. Calculate the value of the silver in the coin, assuming its thickness is uniform. Express your answer in dollars to the nearest dollar.arrow_forwardA solid which has a density of 2.7 g/cm3 has a cavity inside. When it is weighed in air, it is found to have a mass of 312 g and when immersed in water, its mass appears to be 160 g. What is the volume of the hidden cavity?arrow_forward
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning




