
Concept explainers
a. After the label fell off a bottle containing a clear liquid believed to be benzene, a chemist measured the density of the liquid to verify its identity. A 25.0-mL portion of the liquid had a mass of 21.95 g. A chemistry handbook lists the density of benzene at15 -C as
0.8787 g/ml. Is the calculated density in agreement with the tabulated value?
b An experiment requires 15 0 g of cyclohexane. whose density at 25 °C is 0.7781 g/ml. What volume of cyclohexane should be used?
c. A spherical ball of lead has a diameter of 5.0 cm. What is the mass of the sphere if lead has a
density of 11.34 g/cm3? (The volume of a sphere is (4 /3) 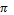
 r3, where r is the radius.)
r3, where r is the radius.)
Trending nowThis is a popular solution!

Chapter 1 Solutions
Student Solutions Manual to Red Exercises for Chemistry: The Central Science
- An antacid tablet weighing 0.853 g contained calcium carbonate as the active ingredient, in addition to an inert binder. When an acid solution weighing 56.519 g was added to the tablet, carbon dioxide gas was released, producing a fizz. The resulting solution weighed 57.152 g. How many grams of carbon dioxide were produced?arrow_forwardWhat is the mass of a 43.8-mL sample of gasoline, which has a density of 0.70 g/cm3?arrow_forwardWhen 12.6 g calcium carbonate (the principal component of chalk) is treated with 63.0 mL hydrochloric acid (muriatic acid sold in hardware stores; density = 1.096 g/mL), the calcium carbonate reacts with the acid, goes into solution, and carbon dioxide gas bubbles out of the solution. After all of the carbon dioxide has escaped, the solution weighs 76.1 g. Calculate the volume (in liters) of carbon dioxide gas that was produced. (The density of the carbon dioxide gas is 1.798 g/L.)arrow_forward
- A 9.55-g sample of a solid was placed in a 15.00-mL flask. The remaining volume was filled with benzene, in which the solid is insoluble. The solid and benzene together weigh 19.33 g. The density of the benzene is 0.876 g/mL. What is the density of the solid?arrow_forwardWhen 10.0g of marble chips (calcium carbonate) is treated with 50.0mL of hydrochloric acid density 1.096g ,the marble dissolves, giving a solution and releasing carbon dioxide gas .The solution weighs 60.4g .How many litres of carbon dioxide gas are released?The density of this gas is 1.798g/Larrow_forwardHow to properly use significant figures in this case?Ccal = (-ΔHrxn x nLR)/ΔTCcal = (-55.85 kJ/mol x 0.00100 mol)/5.3°CCcal = ?????? It would help if you can clarify the rules for multiplication and division. Thanks!arrow_forward
- Ethylene glycol (the main component in antifreeze) has a density of 1.11 g/mL. (a) What is the mass (in g) of 872 mL of ethylene glycol? (b) What is the volume of 2.45 Mg of ethylene glycol?arrow_forwardWire in the US is “labeled” by a number called a gauge. A 16-gauge wire has a diameter of 0.05082 inches. How long, in yards, will a 16-gauge wire be if 5.00 pounds of aluminum formed is used? [V=πr2h] Density of aluminum is 2.70 g/cm3arrow_forwardA 9.55g sample of a solid was placed in a 15.00-ml flask. The remaining volume was filled with benzene, in which the solid was insoluble. The solid and benzene together weigh 19.33g. The density of the benzene is 0.876 g/mL. What is the density of the solid?arrow_forward
- A roll of material weighs 5.32 lbs/ft2 (pounds per square foot). What is the weight in kilograms per square meter(kg/m2 )? Conversion factors: 2.20462 lbs=1kg,10.7639 ft2 =1 m2arrow_forwardAcetone (nail polish remover) has a density of 0.7857 g/cm3.a. What is the mass in g of 28.56 mL of acetone?b. What is the volume in mL of 6.54 g of acetone?arrow_forward1. What is the volume occupied by an aluminum sheet with a mass of 5.28 g? Density of Al = 2.7 g/cm³. 2. What is the area of the filter paper with length of 12mm and width of 1.0cm?arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





