
Consider a N2 molecule in its first excited electronic state, that is, when an electron in the highest occupied molecular orbital is promoted to the lowest empty molecular orbital. (a) Identify the molecular orbitals involved and sketch a diagram to show the transition. (b) Compare the bond order and bond length of N2* with N2, where the asterisk denotes the excited molecule. (c) Is N2* diamagnetic or paramagnetic? (d) When N2* loses its excess energy and converts to the ground state N2, it emits a photon of wavelength 470 nm, which makes up part of the auroras lights. Calculate the energy difference between these levels.
(a)
Interpretation:
The molecular orbital involved in transition should be identified and to sketch the transition. Bond order of 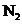 and
and 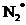 should be found and the bond length should be compared. The magnetic properties of
should be found and the bond length should be compared. The magnetic properties of 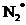 should be found out. The energy difference of the given transition should be determined
should be found out. The energy difference of the given transition should be determined
Concept Introduction:
- In molecular orbital theory, when the bonding takes place the atomic orbitals that take part combine to get a new orbital that has the properties of the whole molecule. The newly formed orbitals are known as molecular orbitals
- The bond order gives an idea about the stability of a molecule. It can be calculated using the molecular orbital theory. The stability of a molecule increase as the bond order increases.
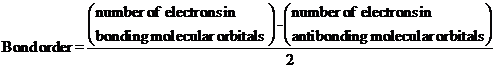
- Paramagnetic species contains at least one unpaired electrons and can be attracted towards magnetic fields. Diamagnetic species does have any unpaired electrons. That is spins of all the electrons are paired. It slightly repelled towards the magnetic fields
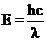

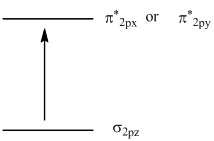
To identify: molecular orbital involved in transition and to sketch the transition.
Answer to Problem 10.113QP
The transition sketch is,
Explanation of Solution
In molecular orbital theory, when the bonding takes place the atomic orbitals that take part combine to get a new orbital that has the properties of the whole molecule. The newly formed orbitals are known as molecular orbitals and only contain a maximum of two electrons. The number of newly formed molecular orbital is equal to the number of atomic orbitals involved in the bonding.
There are two types of molecular orbitals,
- a) Bonding molecular orbitals: sharing of electron density is between the nuclei and has comparatively lower energy and fills first.
- b) Antibonding molecular orbitals: Two nuclei is pulled by the electrons density in opposite direction and has higher energy comparing to bonding molecular orbital.
Molecular orbital diagram of  is given below
is given below
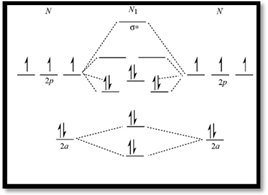
Figure 1
In the ground state of 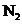 the electrons are in
the electrons are in 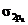 orbital when the
orbital when the 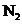 gets excited by getting energy the electron move to
gets excited by getting energy the electron move to 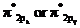 orbitals.
orbitals.
The diagram that showing transition is given below,

(b)
Interpretation:
The molecular orbital involved in transition should be identified and to sketch the transition. Bond order of 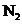 and
and 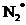 should be found and the bond length should be compared. The magnetic properties of
should be found and the bond length should be compared. The magnetic properties of 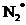 should be found out. The energy difference of the given transition should be determined
should be found out. The energy difference of the given transition should be determined
Concept Introduction:
- In molecular orbital theory, when the bonding takes place the atomic orbitals that take part combine to get a new orbital that has the properties of the whole molecule. The newly formed orbitals are known as molecular orbitals
- The bond order gives an idea about the stability of a molecule. It can be calculated using the molecular orbital theory. The stability of a molecule increase as the bond order increases.
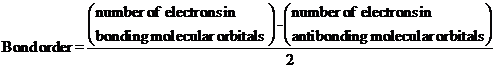
- Paramagnetic species contains at least one unpaired electrons and can be attracted towards magnetic fields. Diamagnetic species does have any unpaired electrons. That is spins of all the electrons are paired. It slightly repelled towards the magnetic fields
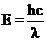

To identify: Bond of order of 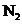 and
and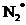 . Also to compare its bond length
. Also to compare its bond length
Answer to Problem 10.113QP
Bond order of 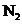 and
and 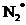 is 3 and 2 respectively. Also the bond length of
is 3 and 2 respectively. Also the bond length of 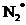 is longer than
is longer than 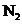 .
.
Explanation of Solution
Electronic configuration of excited nitrogen molecule 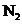 is
is 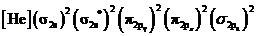
The bond order gives an idea about the stability of a molecule. It can be calculated using the molecular orbital theory. The stability of a molecule increase as the bond order increases.
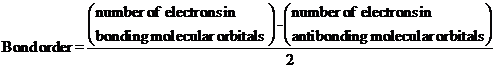
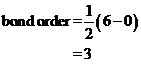
Electronic configuration of excited nitrogen molecule 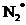 is
is 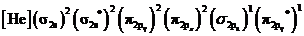
The bond order gives an idea about the stability of a molecule. It can be calculated using the molecular orbital theory. The stability of a molecule increase as the bond order increases.
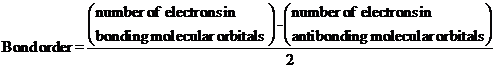
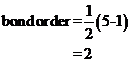
Bond order of 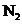 is 3 whereas
is 3 whereas 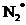 is 2.
is 2.
Therefore, the bond length of 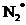 is longer than
is longer than 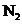 .
.
(c)
Interpretation:
The molecular orbital involved in transition should be identified and to sketch the transition. Bond order of 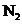 and
and 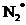 should be found and the bond length should be compared. The magnetic properties of
should be found and the bond length should be compared. The magnetic properties of 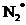 should be found out. The energy difference of the given transition should be determined
should be found out. The energy difference of the given transition should be determined
Concept Introduction:
- In molecular orbital theory, when the bonding takes place the atomic orbitals that take part combine to get a new orbital that has the properties of the whole molecule. The newly formed orbitals are known as molecular orbitals
- The bond order gives an idea about the stability of a molecule. It can be calculated using the molecular orbital theory. The stability of a molecule increase as the bond order increases.
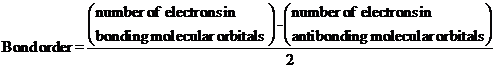
- Paramagnetic species contains at least one unpaired electrons and can be attracted towards magnetic fields. Diamagnetic species does have any unpaired electrons. That is spins of all the electrons are paired. It slightly repelled towards the magnetic fields
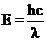

To identify: The magnetic properties of 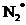
Answer to Problem 10.113QP
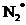 is diamagnetic
is diamagnetic
Explanation of Solution
Paramagnetic species contains at least one unpaired electrons and can be attracted towards magnetic fields. Diamagnetic species does have any unpaired electrons. That is spins of all the electrons are paired. It slightly repelled towards the magnetic fields.
Electronic configuration of excited nitrogen molecule 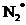 is
is 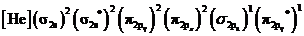
Even though there are unpaired electrons, the spin of the electrons was not change in the time of transition. All the electrons are paired so it is diamagnetic.
(d)
Interpretation:
The molecular orbital involved in transition should be identified and to sketch the transition. Bond order of 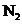 and
and 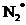 should be found and the bond length should be compared. The magnetic properties of
should be found and the bond length should be compared. The magnetic properties of 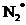 should be found out. The energy difference of the given transition should be determined
should be found out. The energy difference of the given transition should be determined
Concept Introduction:
- In molecular orbital theory, when the bonding takes place the atomic orbitals that take part combine to get a new orbital that has the properties of the whole molecule. The newly formed orbitals are known as molecular orbitals
- The bond order gives an idea about the stability of a molecule. It can be calculated using the molecular orbital theory. The stability of a molecule increase as the bond order increases.
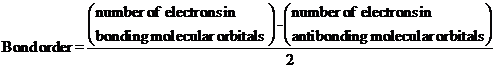
- Paramagnetic species contains at least one unpaired electrons and can be attracted towards magnetic fields. Diamagnetic species does have any unpaired electrons. That is spins of all the electrons are paired. It slightly repelled towards the magnetic fields
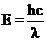

To determine: The energy difference of the given transition.
Answer to Problem 10.113QP
The energy difference of the given transition is 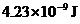
Explanation of Solution
The energy of light is calculated below.
Given,
The wavelength of light is 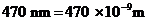 .
.
Planck’s constant is 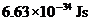
Speed of the light is 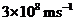
The energy of light is calculated is calculated by the equation,
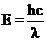

Substituting the given values in the equation,
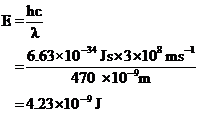
The energy difference of the given transition is
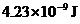
Want to see more full solutions like this?
Chapter 10 Solutions
Connect for Chemistry
- What are the relationships among bond order, bond energy, and bond length? Which of these quantities can be measured?arrow_forwardIn the molecular orbital mode l, compare and contrast bonds with bonds. What orbitals form the bonds and what orbitals form the bonds? Assume the z-axis is the internuclear axis.arrow_forwardIs the electron density consistentwith equal contributions from the tworesonance structures for O3? Explain.arrow_forward
- Write the ground-state electron configuration of O2 and calculate the bond order.arrow_forwardDraw a molecular orbital energy-level diagram for the gaseous heteronuclear diatomic molecular boron nitride, BN. How does it differ from that for C2arrow_forwardWhy does the energy ordering of the molecular orbitals of the second-period diatomic molecules change in going from N2 to O2?arrow_forward
- The wavelength at which the O2 molecule most stronglyabsorbs light is approximately 145 nm. (a) In which regionof the electromagnetic spectrum does this light fall?(b) Would a photon whose wavelength is 145 nm haveenough energy to photodissociate O2 whose bond energy is495 kJ/mol? Would it have enough energy to photoionize O2?arrow_forwardThe diatomic molecule OH exists in the gas phase. OH playsan important part in combustion reactions and is a reactiveoxidizing agent in polluted air. The bond length and bondenergy have been measured to be 97.06 pm and 424.7 kJ/mol,respectively. Assume that the OH molecule is analogous to theHF molecule discussed in the chapter and that the MOs resultfrom the overlap of a pz orbital from oxygen and the 1s orbitalof hydrogen (the OOH bond lies along the z axis).arrow_forwardSketch the molecular orbital diagram for the ground state of carbon monoxide, determine the bond order, and indicate if the molecule is paramagnetic or diamagnetic.arrow_forward
- The main component of air is N2. From the molecular orbital diagram of N2, predict its bond order and whether it is diamagnetic or paramagnetic.arrow_forwardUsing molecular orbital theory, determine the electronic structure of the first excited electronic state of N2. What differences are expected in the properties of the excited state of N2 compared with the same properties of the ground state?arrow_forwardCalculate the bond orders for O2 and BO and in each case state whether the molecule is paramagnetic or diamagnetic.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning



