
Interpretation:
The 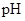 value,
value,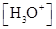 and
and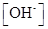 concentration for given weak acid should be determined.
concentration for given weak acid should be determined.
Concept Introduction:
Weak Acids: Acids that do not easily dissociate into ions completely which has difficulty in proton donation are considered as weak acids. Weak acid forms stronger conjugated base.
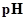 : The concentration of hydrogen ion is measured using
: The concentration of hydrogen ion is measured using 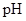 scale. The
scale. The 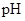 of a solution is a figure that expresses the acidity or the alkalinity of a given solution.
of a solution is a figure that expresses the acidity or the alkalinity of a given solution.
It is defined as the negative base-10 logarithm of the hydrogen or hydronium ion concentration.
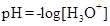
Ionic-product constant for water: It is the hydronium ion concentration times the 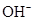 concentration present in the solution.
concentration present in the solution.
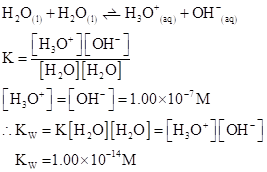
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Fundamentals Of General, Organic, And Biological Chemistry Volume 1 Second Custom Edition For Washington State University, 2/e
- The pH of black coffee is 5, and that of milk of magnesia is 10. Is the coffee twice as acidic as milk of magnesia?arrow_forwardWhat is the molarity of a NaOH solution if 48.0 mLneutralizes 35.0 mL of 0.144 M H2SO4?arrow_forwardWhat is the pH of a solution of 100 ml of 0.01 M H3PO4 and 100 ml of 0.01 M Na3PO4?arrow_forward
- The pH of a solution containing 20 mL of 0.120 N NaOH and 35 mL of 0.120 N NaOH is 1.49. True or False?arrow_forwardWhat are the molarity and the normality of a solution made by dissolving 25 g of citric acid (triprotic, C6H5O7H3) in enough water to make 800 mL of solution?arrow_forwardIf a solution has a pH of 1, how many more H+ are present than in a solution with a pH of 4?arrow_forward
- A 0.10 M solution of the deadly poison hydrogen cyanide, HCN, has a pH of 5.2. Calculate the [H3O+] of the solution. Is HCN a strong or a weak acid?arrow_forwardThe H⁺ concentration in an aqueous solution at 25 °C is 4.3 × 10⁻⁶. What is [OH⁻]?arrow_forwardGiven the following data plot of the volume of NaOH vs pH a. what does the values in 1 and 2 indicates: _____ b. Determine the molar mass of the unknown acid: _____ c. choose the identity of the acid from the choices below: _____ acetic acid, MM = 60.05 g/mole formic acid, MM = 40.03 g/mole oxalic acid = 90.03 g/molearrow_forward
- What is the pH of the following buffer mixtures? (a) 100 mL 1 M acetic acid plus 100 mL 0.5 M sodium acetate (b) 250 mL 0.3 M phosphoric acid plus 250 mL 0.8 M KH2PO4arrow_forwardThe pH of a 0.0082 M solution of HNO₃ isarrow_forwardWhat is the Molarity of 10 grams of NaCl dissolved in 3L of solvent?arrow_forward
 Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning
Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning




