
Concept explainers
Like other electrophiles, carbocations add to
undergo substitution or elimination reactions depending on the reaction conditions. With this in mind, consider the following reactions of nerol, a natural product isolated from lemon grass and other plant sources. Treatment of nerol with
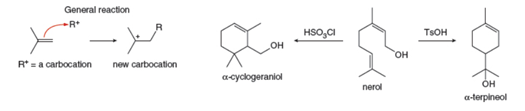
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
PKG ORGANIC CHEMISTRY
Additional Science Textbook Solutions
General Chemistry: Atoms First
Chemistry: The Central Science (14th Edition)
Chemistry (7th Edition)
- When 2-pentene is treated with Cl2 in methanol, three products are formed. Account for the formation of each product (you need not explain their relative percentages).arrow_forwardDraw the structure of each product from the reaction of benzene with 2-chloro-1-methylcyclohexane using AlCl 3 as the catalyst and Identify the major product.arrow_forward1-pentyne reacts with dicylohexylborane, followed by oxidation, to produce pentanal. The organoborane formed in step 1 is oxidized by H2O2 in the presence of OH– in the next step of the reaction. Which of the following options correctly describe this process? Choose one or more: A. –BR2 acts as a leaving group in this step. B. OH– must attack from the face of the alkene opposite the –BR2 group. C. OH replaces –BR2 in this step. D. OH replaces H in this step. E. Configuration at the carbon-bearing B is retained in the reaction.arrow_forward
- TRUE OR FALSE I. The reaction of BH3 with 2-methylbut-2-ene results in the attachment of a hydroxyl group, OH, on the third carbon since this carbon is less substituted. II. The reduction of an alkyne using a strong base such as LiNH2 gives an alkane when 2 equivalents of hydrogen gas is added. III. Hydrobromination of an alkene follows anti-Markovnikov’s rule in the presence of a peroxide.arrow_forwardDraw the structural formula for a cycloalkene with the molecular formula C6H10 that reacts withCl2 in a halogenation reaction to give the following product.arrow_forwardA chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2 CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under the same conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C. Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structures of A, B, and C; give equations for their formation; and explain the stereospecificity of these reactions.arrow_forward
- For the following alkene , write the structure of its reaction with the following 1. Reaction with H2 and a Pd catalyst 2. reaction with Br2 3. Reaction with HBrarrow_forwardname and draw the product/s formed from the reaction of 3-methylpent-2-ene: 1. with HBr 2. with Cl2 3. H2O in the presence of H2SO4 4. ethanol in the presence of H2SO4 5. Br2, H2O 6. [1] 9-BBN; [2] H2O2, OH-arrow_forwardINSTRUCTIONS: Choose from the options A-E and explain briefly by illustrations the chemistry behind each answer. Preamble: A reaction flask contains a 2-bromopentane in an ethanolic solution of sodium ethoxide at room temperature and results in the formation of two olefinic products. Predict the products that would be formed from such a reaction a)1-pentene (80%) and 2-pentene (20%)B)1-pentene (20%) and 2-pentene (80 %)C)1-pentene (50 %) and 2-pentene (50 %)D)1-pentene (0 %) and 2-pentene (100 %)E)1-pentene (100 %) and 2-pentene (0 %) What reaction pathway is followed by the reaction above?a)E2 dehydrohalogenation B)E1 dehydrohalogenationC)SN1 dehydrohalogenationD)SN2 dehydrohalogenation A mixture of E1 and E2 pathways What is responsible for the formation of different products (major and minor).A)The different activated complex involved in the mechanism. B)Bimolecular nuclephilic substitution reaction C)Bimolecular Elimination reaction D)The presence of sodium ethoxideE)The…arrow_forward
- INSTRUCTIONS: Choose from the options A-E and explain briefly by illustrations the chemistry behind each answer. Preamble: A reaction flask contains a 2-bromopentane in an ethanolic solution of sodium ethoxide at room temperature and results in the formation of two olefinic products. Predict the products that would be formed from such a reaction A. 1-pentene (80%) and 2-pentene (20%) B. 1-pentene (20%) and 2-pentene (80 %) C. 1-pentene (50 %) and 2-pentene (50 %) D. 1-pentene (0 %) and 2-pentene (100 %) E. 1-pentene (100 %) and 2-pentene (0 %) 2. What reaction pathway is followed by the reaction above? A. E1 dehydrohalogenation B. E2 dehydrohalogenation C. SN1 dehydrohalogenation D. SN2 dehydrohalogenation E. A mixture of E1 and E2 pathways 3. What is responsible for the formation of different products (major and minor). A. The different activated complex involved in the mechanism. B. Bimolecular Nucleophilic substitution reaction C. Bimolecular Elimination reaction D. The…arrow_forwardDraw structural formulas for the alkenes formed on acid-catalyzed dehydration of each alcohol. Where isomeric alkenes are possible, predict which alkene is the major product. Q. 2-Methylcyclopentanol (racemic)arrow_forwardWhen the acid-catalyzed dehydration of the compound 2-methyl-2-butanol yields two alkene products, what are the names of these two alkenes? Which of the two alkenes will be the major product? Thank youarrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

