
Concept explainers
Draw the major product obtained when each of the following
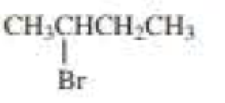
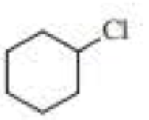
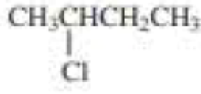
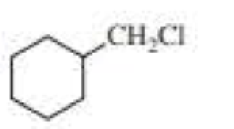
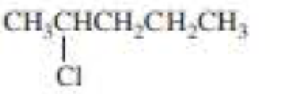
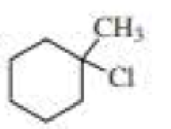
(a)
Interpretation:
Major product formed when given alkyl halides undergoes E2 reaction has to be drawn.
Concept Introduction:
An E2 reaction is a concerted, one-step reaction in which the proton and the halide ion are removed in the same step. A strong base favours an E2 reaction. A high concentration of a base favours an E2 reaction. If one of the reactants is charged, an E2 reaction will be favoured by the least polar solvent that will dissolve the reactant. If neither of the reactant is charged an E2 reaction is favoured by the protic polar solvent. The major product is a stable alkene, unless the reactants are sterically hindered.
Explanation of Solution
The product when given compound undergo E2 addition,
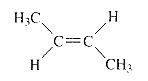
For an E2 reaction major product is the more stable alkene. The reactant is a secondary alkyl halide, undergo E2 reactions.
(b)
Interpretation:
Major product formed when given alkyl halides undergoes E2 reaction is to be identified.
Concept Introduction:
An E2 reaction is a concerted, one-step reaction in which the proton and the halide ion are removed in the same step.
Explanation of Solution
Chlorocyclohexane is a less stable conformer undergo E2 reaction as
The reactant is a secondary alkyl halide, undergo E2 reactions.
(c)
Interpretation:
Major product formed when given alkyl halides undergoes E2 reaction is to be identified.
Concept Introduction:
An E2 reaction is a concerted, one-step reaction in which the proton and the halide ion are removed in the same step. A strong base favours an E2 reaction. A high concentration of a base favours an E2 reaction. If one of the reactants is charged, an E2 reaction will be favoured by the least polar solvent that will dissolve the reactant. If neither of the reactant is charged an E2 reaction is favoured by the protic polar solvent. The major product is a stable alkene, unless the reactants are sterically hindered.
Explanation of Solution
Product of given compound when undergo E2 reaction,
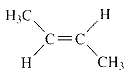
More substituted alkene is more stable therefore should be the major product.
(d)
Interpretation:
Major product formed when given alkyl halides undergoes E2 reaction is to be identified.
Concept Introduction:
An E2 reaction is a concerted, one-step reaction in which the proton and the halide ion are removed in the same step. A strong base favours an E2 reaction. A high concentration of a base favours an E2 reaction. If one of the reactants is charged, an E2 reaction will be favoured by the least polar solvent that will dissolve the reactant. If neither of the reactant is charged an E2 reaction is favoured by the protic polar solvent.
The major product is a stable alkene, unless the reactants are sterically hindered.
Explanation of Solution
Product of given compound when undergo E2 reaction,
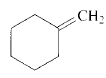
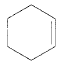
Chloromethyl cyclohexane on E2 reaction gives methylene cyclohexane. And E2 reaction is regioselective: major product is the more substituted stable alkene.
(e)
Interpretation:
Major product formed when given alkyl halides undergoes E2 reaction is to be identified.
Concept Introduction:
An E2 reaction is a concerted, one-step reaction in which the proton and the halide ion are removed in the same step. A strong base favours an E2 reaction. A high concentration of a base favours an E2 reaction. If one of the reactants is charged, an E2 reaction will be favoured by the least polar solvent that will dissolve the reactant. If neither of the reactant is charged an E2 reaction is favoured by the protic polar solvent.
The major product is a stable alkene, unless the reactants are sterically hindered.
Explanation of Solution
Product of given compound when undergo E2 reaction,
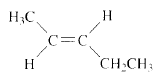
The reactant is a secondary alkyl halide, undergo E2 reactions.
(f)
Interpretation:
Major product formed when given alkyl halides undergoes E2 reaction is to be identified.
Concept Introduction:
An E2 reaction is a concerted, one-step reaction in which the proton and the halide ion are removed in the same step. A strong base favours an E2 reaction. A high concentration of a base favours an E2 reaction. If one of the reactants is charged, an E2 reaction will be favoured by the least polar solvent that will dissolve the reactant. If neither of the reactant is charged an E2 reaction is favoured by the protic polar solvent.
The major product is a stable alkene, unless the reactants are sterically hindered.
Explanation of Solution
Product of given compound when undergo E2 reaction,
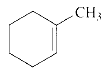
An E2 reaction is regioselective: major product is the more substituted stable alkene.
Want to see more full solutions like this?
Chapter 10 Solutions
Organic Chemistry (Package) (Looseleaf)
- Draw ALL the major product obtained when the following alkyl halide undergoes an E1 reaction. The E1 reaction below may have more than one product that forms (less substituted, more substituted, E/Z). Be sure to draw out all of the products that form.arrow_forwardWhat is the major elimination product obtained from an E2 reaction of each of the following alkyl halides with hydroxide ion?arrow_forwardDraw the product of the reaction of bromoethane with each of the following nucleophiles? - (CH3)3N -CH3CH2S−arrow_forward
- Identify the major and minor products for E2 reactions that occurs when each of the following substrates is treated with a strong base. Please explain afterwards how to get it, tricks would be good and not complicated stuffs.arrow_forwardDraw the product of each of the following Sn2 reactions: a) Chloropentane and NaSH b) (R)-3-lodohexane and NaClarrow_forwardWhat is the major product obtained when each of the following compounds undergoes an E2 reaction with methoxide ion? Show the configuration of the product. Does the product obtained depend on whether you start with the R or S enantiomer of the reactant?arrow_forward
- Identify the major and minor products for E2 reactions that occurs when each of the following substrates is treated with a strong base. Please explain afterwards how to get it, tricks would be good and not complicated stuffs. Please show the streochemistry on the drawing!arrow_forwardIdentify the major and minor products for the E2 reaction that occus when each of the following substrates is treated with a strong base: find the productsarrow_forwardDraw the major product(s) obtained from the acid-catalyzed hydration of the following alkene: CH3CH2CH2CH=CH2arrow_forward
- Be sure to answer all parts. Draw the product formed when the following alkene is treated with either [1] HBr alone and [2] HBr in the presence of peroxides. CH2CHCH2CH2CH2CH3arrow_forwardWhich of the following bases can remove a proton from a terminal alkyne in a reaction that favors products? CH3O− NH3 CH3CH2 H2C=CH F−arrow_forwardWhat products are obtained from the reaction of each of the following alkenes with OsO4 followed by aqueous H2O2?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





