
MODERN PHYSICS (LOOSELEAF)
4th Edition
ISBN: 9781119495550
Author: Krane
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 10, Problem 4Q
How would Figure 10.13 change if the temperature of the gas were increased?
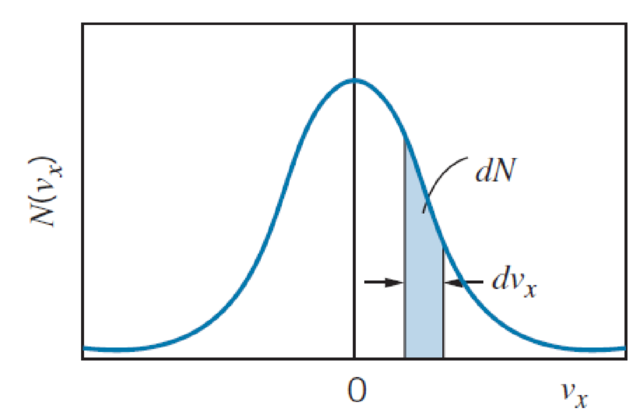
FIGURE 10.13 The Maxwell velocity distribution for gas molecules. The distribution is centered on vx = 0. The shaded strip represents the number of molecules with velocity components between vx and vx + dvx.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The combined gas law relates which of the following?
A) Volume & Temperature
B) Temperature & Pressure
C) Volume & Pressure
D) Volume, Temperature & Pressure
Describe the Ideal Gas Law?
The mercury level in the left arm of the J-shaped tube is attached to a thermostat gas-containing bulb. The left arm is 10.83 cm and the right arm is 34.71 cm above the bottom of the manometer. If the barometric pressure reads 738.4 Torr, what is the pressure of the gas? Assume that temperature-induced changes in the reading of the barometer and J tube are small enough to neglect.
Include a drawing of the diagram
Chapter 10 Solutions
MODERN PHYSICS (LOOSELEAF)
Additional Science Textbook Solutions
Find more solutions based on key concepts
The capacitance of plates.
Physics: Principles with Applications
In which extrasolar planet system(s) (A–D) is the planet closest to the star?
Lecture- Tutorials for Introductory Astronomy
An electromagnetic wave has a frequency of 12 MHz. What is its wavelength in vacuum?
University Physics Volume 2
Review Question 23.5 Where should you place an object with respect to a convex lens to have an image at exactly...
College Physics
The real part, imaginary part and the absolute value of cos(ix) .
Mathematical Methods in the Physical Sciences
49. In addition to producing images, ultrasound can be used to heat tissues of the body for therapeutic purpose...
College Physics: A Strategic Approach (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- How does the kinetic theory describe a gas?arrow_forwardWhat properties of a nonideal gas do the van der Waals constants a and b represent?arrow_forwardIf we have 35.54L of O2 gas at STP, how many moles of this gas do we have? Note: it is understood that the unit of your answer is some number of moles, however do not explicitly include units in your answer. Enter only a number. If you do enter a unit ("mol" in this case), you answer will be counted wrong. (STP means "standard temperature and pressure," which is 1 atmosphere of pressure or 101,325 Pa, and 273K).arrow_forward
- A star is formed when the gravitational attraction overcomes the pressure due to the gases inside.Thus we can write(in-picture) .... Assuming the ideal gas equation, we can write P = nkT, where n is the number of atoms/volume. Let M and m denote the total mass and the mass of each gas atom. Using the above equation, show that the condition for star formation is that the mass of the star obeys M > MJ, where : (in-picture)arrow_forwardWhat does the effective conductivity of an enclosure represent? How is the ratio of the effective conductivity to thermal conductivity related to the Nusselt number?arrow_forwardCan you please solve the below problem, showing step by step: A balloon contains mostly helium and a little nitrogen. The pressure of the helium is 425 mm Hg. What is the partial pressure of the nitrogen if the total pressure of the balloon is 67.98 kPa?arrow_forward
- The cylinder in the figure(Figure 1) has a moveable piston attached to a spring. The cylinder's cross-section area is 10 cm2, it contains 0.0055 mol of gas, and the spring constant is 1500 N/m. At 21 ∘C the spring is neither compressed nor stretched. How far is the spring compressed if the gas temperature is raised to 140 ∘C?arrow_forwardWhat is the temperature and pressure at STP?arrow_forwardWhich gas has higher specific heat capacity, a monoatomic or a diatomic gas at room temperature?arrow_forward
- As6 show the compression of hydrogen gas from 30 to 900bar on a log p diagram ?arrow_forward⦁ In a standard gas grill propane tank, there is approximately 4,579 mL of propane (C3H8). At a temperature of 55˚C, the tank has a pressure of 1,798 kPa. The tank is cooled to 25˚C and the pressure reduced to 1,025 kPa. What will the new volume be? Remember to pay close attention to the units of temperature before beginning your calculations.arrow_forwardconsidering a very small object, such as a grain of pollen, in a gas, the number of molecules striking its surface would also be relatively small. Could we expect the grain of pollen to experience any fluctuations in pressure due to statistical fluctuations in the number of gas molecules striking it in a given amount of time?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
 An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Modern Physics
Physics
ISBN:9781111794378
Author:Raymond A. Serway, Clement J. Moses, Curt A. Moyer
Publisher:Cengage Learning


An Introduction to Physical Science
Physics
ISBN:9781305079137
Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY