
Concept explainers
Elemental carbon has one gas phase, one liquid phase, and two different solid phases, as shown in the phase diagram:
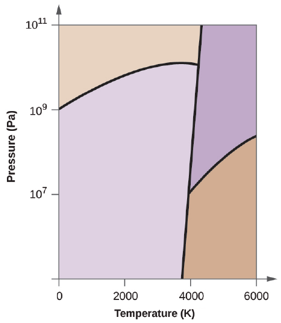
(a) On the phase diagram, label the gas and liquid regions.
(b) Graphite is the most stable phase of carbon at normal conditions. On the phase diagram, label the graphite phase.
(c) If graphite at normal conditions is heated to 2500 K while the pressure is increased to
(d) Circle each triple point on the phase diagram.
(e) In what phase does carbon exist at 5000 K and
(f) If the temperature of a sample of carbon increases from 3000 K to 5000 K at a constant pressure of
Trending nowThis is a popular solution!

Chapter 10 Solutions
Chemistry (OER)
Additional Science Textbook Solutions
College Physics
General, Organic, and Biological Chemistry (3rd Edition)
Chemistry: Structure and Properties (2nd Edition)
CHEMISTRY-TEXT
Chemistry: A Molecular Approach (4th Edition)
- The phase diagram of a hypothetical substance is (a) Estimate the normal boiling point and freezing point ofthe substance. (b) What is the physical state of the substanceunder the following conditions? (i) T = 150 K, P = 0.2 atm;(ii) T = 100 K, P = 0.8 atm; (iii) T = 300 K, P = 1.0 atm.(c) What is the triple point of the substance?arrow_forwardThe figure below is the phase diagram of a purse substance. (I), (II) and (III) are the 3 phases of the substance, and there are 3 phase boundaries. Based on this phase diagram, which of the following statement is (are) true about this substance? a) Below 0.7 atm, the liquid phase of this substance can be formed by carefully adjusting temperature. b) The substance can be transformed from liquid to solid by applying pressure. c) The normal boiling point is between 190 K and 210 K. Pressure (in atm) 1.4 1.3 1.2 1.1 1.0 0.9 0.8 (III) 0.7 0.6- 0.5- 120 140 160 180 200 220 T (in Kelvin) O a) only O b) only O c) only O a) and b) O b) and c) O c) and a) O a), b) , and c) O None of the 3 statements is true.arrow_forwardThe phase diagram for a pure substance is shown above. Use this diagram and your knowledge about changes of phase to answer the following questions. a) What does point V represent? What characteristics are specific to the system only at point V?. (b) What does each point on the curve between V and W represent? (c) Describe the changes that the system undergoes as the temperature slowly increases from X to Y to Z at 1.0 atmosphere. Note: Please briefly explain A-C. Thank you.arrow_forward
- The vapor pressure of benzene is 224 mmHg at 45 °C and 648 mmHg at 75 °C.(a) Find the enthalpy of vaporization of benzene, ∆Hvap (kJ/mol), assuming it is constant. You may also assume that ZV − ZL ≃ 1. B)arrow_forwardA metal crystallizes in the face-cente red cubic crystal structure with a unit cell edge of 4.08 x 10 -8 cm. The density of the metal is 19.3 g/cc. (a) What is the mass, in grams, of a single atom of this element? (b) What is the atomic weight of the element (g/mol). (c) What is the radius, in cm, of an atom of the element?arrow_forwardThe vapor pressure of acetone (C3H,O) at 39.5°C is 400 torr. (a) If the boiling point of acetone is 56.5°C, calculate its enthalpy of vaporization (AH,vap; in kJ/mol). (b) Using calculated (AH,vap and vapor pressure at 39.5, calculate the vapor pressure of acetone at 25°C. (R = 8.314 J/(mol.K))arrow_forward
- A 5.4-L closed vessel is filled with 0.46 g of liquid ethanol. This ethanol containing closed vessel is placed in the freezer and reaches the vapor pressure of 6.65 torr at -11 C. (i) Is there any liquid ethanol remained in the closed vessel when the container is removed and warmed to the temperature of 25 C? Justify your answer. (ii) How many grams of liquid ethanol remained in the vessel at 2.0 C? The enthalpy changes of vaporization, AH,vap of ethanol is 40.5 kJ/mol. The molar mass of ethanol is 46.07 g/mol.arrow_forwardThe molar enthalpy of vaporization of hexane (C6H14) is 28.9 kJ/mol, and its normal boiling point is 68.73 °C. What is the vapor pressure of hexane at 25.00 °C?arrow_forwardRefer to the phase diagram below, and describe the phase changes (and the temperatures at which they occur) when CO2 is heated from -80° C to -20° C at (a) a constant pressure of 3 atm; (b) a constant pressure of 6 atm; (c) a constant pressure of 9 atmarrow_forward
- The phase diagram for elemental sulfur is shown in Figure 6.18. (a) How many allotropes are shown? (b) What is the stable allotrope of sulfur under normal conditions of temperature and pressure? (c) Describe the changes to sulfur as its temperature is increased from 25?C while at 1 atm pressure.arrow_forwardName the phase transition in each of the following situationsand indicate whether it is exothermic or endothermic:(a) Bromine vapor turns to bromine liquid as it iscooled. (b) Crystals of iodine disappear from an evaporatingdish as they stand in a fume hood. (c) Rubbing alcoholin an open container slowly disappears. (d) Molten lavafrom a volcano turns into solid rock.arrow_forwardDetermine enthalpy of vaporization graphically using the Clausius-Clapeyron equation. The vapor pressure of iodomethane (CH3I) has been measured over a range of temperatures. (a) Which of the following should give a linear plot (temperatures in K)? On the x-axis: On the y-axis: (b) When the appropriate plot is made, the resulting straight line has a slope of -3.65x10° K. Use this information to determine AHvap for CH3I. kJ/mol Check & Submit Answer Show Approach Example Problem 11.2.5 Use the two-point version of the Clausius-Clapeyron equation The vapor pressure of liquid aluminum is 400. mm Hg at 2590 K. Assuming that AHvap for Al (29 change significantlv with temperature.calculate the vapor pressure of liauid Al at 2560 K. tv Ahulu Warrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning


