
(a)
Interpretation:
The given set of solution should be identified as either acidic or basic solution, and the value of 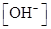 should be determined.
should be determined.
Concept Introduction:
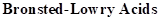 : If a species loses a proton then it is considered as
: If a species loses a proton then it is considered as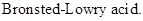
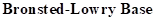 : If a species receives one proton, then it is considered as
: If a species receives one proton, then it is considered as 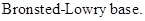
If a base receives one proton, then the formed species is a conjugate acid whereas an acid lose one proton, then the formed species is a conjugated base.
Ionic-product constant for water: It is the hydronium ion concentration times the 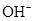 concentration present in the solution.
concentration present in the solution.
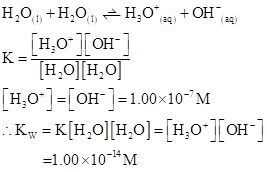
The 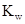 will apply to all aqueous solution.
will apply to all aqueous solution.
For acidic solution 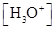 is large that is
is large that is 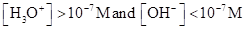
For basic solution 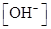 is large that is
is large that is 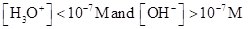
(b)
Interpretation:
The given set of solution should be identified as either acidic or basic solution and the value of 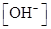 should be determined.
should be determined.
Concept introduction:
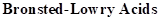 : If a species loses a proton then it is considered as
: If a species loses a proton then it is considered as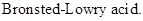
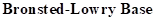 : If a species receives one proton, then it is considered as
: If a species receives one proton, then it is considered as 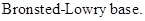
If a base receives one proton, then the formed species is a conjugate acid whereas an acid lose one proton, then the formed species is a conjugated base.
Ionic-product constant for water: It is the hydronium ion concentration times the 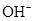 concentration present in the solution.
concentration present in the solution.
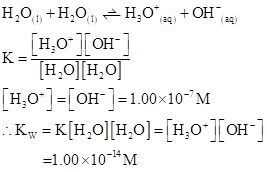
The 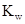 will apply to all aqueous solution.
will apply to all aqueous solution.
For acidic solution 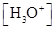 is large that is
is large that is 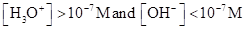
For basic solution 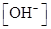 is large that is
is large that is 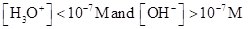
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
FUND. GEN. ORG&BIO CHEM PKG
- If an unknown solution of cobalt (II) chloride has an absorbance of 0.79, what is its concentration? Include proper units, please How did you determine this using the Beer’s Law plot?arrow_forwardHow many moles of sodium hypochlorite can be prepared from 32.5 g of NaOH?arrow_forwardgiven a stock solution of 10% dextrose, how would you make 5 mL of a 0.9% dextrose solution? find mL of stock and mL of h20arrow_forward
- Glucose-1-phosphate has a ΔG°′ value of −20.9 kJ/mol, whereas that for glucose-6-phosphate is −12.5 kJ/mol. After reviewing the molecular structures of these compounds, explain why there is such a difference in these values.arrow_forwardBalance the following equation, and tell how many moles of nickel will reactwith 9.81 mol of hydrochloric acid.arrow_forwardWhat is the pH of a 0.0025 M solution of HCl?arrow_forward
- The reaction of methoxy benzene with hydrogen iodide will yield a phenol and an alkyl halide. Which of following choices is the correct combination of the products?arrow_forwardTomato juice has a OH- concentration equal to 1 X 10 -10, what is the pH of this solution?arrow_forwardAssuming equal concentrations of conjugate base and acid, which one of the following mixtures is suitable for making a buffer solution with an optimum pH of 7.4–7.6? NaOCl / HOCl (K a = 3.2 × 10 –8) NH 3 / NH 4Cl (K a = 5.6 × 10 –10) NaNO 2 / HNO 2 (K a = 4.5 × 10 –4) NaCl / HCl CH 3COONa / CH 3COOH (K a = 1.8 × 10 –5)arrow_forward
- The following equation shows the reaction of baking soda (NaHCO3) and hydrochloric acid (HCl). NaHCO3+HCl → CO2+H2O+NaCl If you have 3.0 grams of NaHCO3, how many moles of HCl are needed for a complete relation?arrow_forwardWhat is the molar mass of diazepam (Valium), C16H13ClN2O?arrow_forwardA compound with empirical formula C2H5O was found in a separate experiment to have a molar mass of approximately 90 g. What is the molecular formula of the compound?arrow_forward

